Acute and Chronic Nutritional Disorders in Children
1. Nuralieva Altynai Topchubaevna
2. Aamina Khan
Mohammad Arbaz
Mohd Suhail
(1. Teacher, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.
2. Students, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.)
Abstract
In pediatric clinical practice, the terminology surrounding "eating disorders" often bridges two distinct pathophysiological domains: primary psychiatric conditions (e.g., anorexia nervosa) and profound physiological nutritional deficits, classically termed Nutritional Disorders or Protein-Energy Malnutrition (PEM). In global pediatrics, particularly within post-Soviet medical curricula, acute and chronic eating/nutritional disorders primarily refer to conditions of hypotrophy, malnutrition, and failure to thrive. This article provides an extensive review of pediatric PEM. We delineate the multifactorial etiology—spanning exogenous alimentary deficits to endogenous metabolic demands—and explore the pathogenesis of "reductive adaptation," a survival mechanism that ultimately compromises organ function. The classification distinguishes between acute (wasting) and chronic (stunting) malnutrition, guiding the clinical recognition of Marasmus and Kwashiorkor. We further detail the diagnostic reliance on WHO anthropometric Z-scores and critical laboratory evaluations. Finally, we review the structured pharmacological and nutritional rehabilitation protocols, emphasizing the World Health Organization’s (WHO) 10-step management strategy for severe acute malnutrition to prevent refeeding syndrome and reduce mortality.
Keywords: Protein-Energy Malnutrition (PEM), Severe Acute Malnutrition (SAM), Hypotrophy, Marasmus, Kwashiorkor, Refeeding Syndrome, Anthropometry.
1. Introduction
The conceptualization of "eating and nutritional disorders" in pediatric medicine requires careful semantic distinction. While in older adolescents this may evoke psychiatric conditions, in infants and young children, it predominantly refers to acute and chronic states of undernutrition, historically termed "hypotrophy" or, in modern global parlance, Protein-Energy Malnutrition (PEM) and Failure to Thrive (FTT) [1]. These disorders represent a critical failure to meet the metabolic demands of a rapidly growing organism, leading to profound developmental and physiological consequences.
Despite global advancements in public health, acute and chronic malnutrition remains a leading underlying cause of pediatric mortality, contributing to nearly 45% of deaths among children under five years of age globally [2]. The outpatient and intensive care management of these children requires a meticulous, phased approach, as their physiology is fundamentally altered. Treating a severely malnourished child is not simply a matter of providing excess food; rather, it is the delicate management of a fragile metabolic state where abrupt nutritional shifts can be fatal [3].
2. Etiology
The etiology of pediatric nutritional disorders is conventionally divided into exogenous (environmental) and endogenous (organic) factors, which frequently overlap in clinical scenarios.
2.1 Exogenous Factors
Alimentary deficiency is the most common global cause. This includes absolute caloric deprivation, improper formulation of infant formula (e.g., over-dilution), or delayed introduction of complementary foods [1]. Psychosocial factors also play a massive role; maternal depression, neglect, extreme poverty, and poor maternal-infant bonding directly correlate with inadequate caloric intake, a condition often termed "non-organic failure to thrive" [4].
2.2 Endogenous Factors
Endogenous causes encompass any organic pathology that impairs nutrient intake, absorption, or increases metabolic demand. Gastrointestinal anomalies (cleft lip/palate, pyloric stenosis), malabsorption syndromes (cystic fibrosis, celiac disease), and chronic infections (tuberculosis, pediatric HIV) are classic culprits [2]. Furthermore, congenital heart disease and chronic renal failure drastically increase the basal metabolic rate, creating a relative nutritional deficit even when caloric intake appears quantitatively normal [3].
3. Pathogenesis: The Concept of Reductive Adaptation
The human body does not passively succumb to starvation; it mounts a highly orchestrated physiological defense. This metabolic shift is known as "reductive adaptation" [5].
When a child experiences acute caloric restriction, the endocrine system rapidly alters its profile to mobilize stored energy and preserve essential organ function (brain and heart) at the expense of somatic growth. Insulin levels plummet, while counter-regulatory hormones—glucagon, cortisol, and growth hormone—surge [5]. This hormonal milieu drives glycogenolysis, lipolysis, and the catabolism of skeletal muscle to provide amino acids for hepatic gluconeogenesis [2].
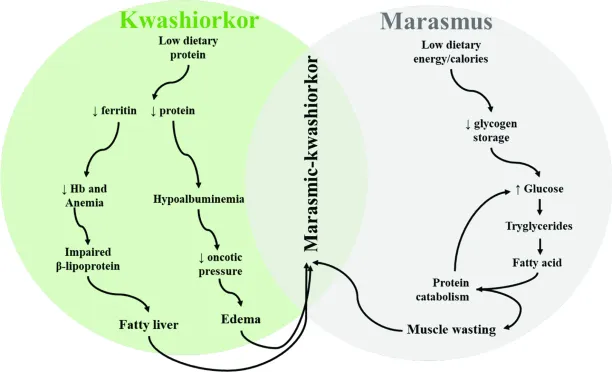
Figure 1. The pathogenesis of Protein-Energy Malnutrition. The diagram illustrates the neuroendocrine shift in starvation, highlighting the divergence into Marasmus (adapted starvation with severe muscle wasting) and Kwashiorkor (maladapted starvation with hypoalbuminemia and systemic edema).
(Source: Kliegman, R. M., et al., Nelson Textbook of Pediatrics, 21st ed., Elsevier, 2020).
However, this adaptation is a double-edged sword. As cellular energy depletes, the sodium-potassium ATPase pumps fail. Sodium and water accumulate intracellularly, while potassium and magnesium are lost in the urine. This total-body potassium depletion, masked by hemoconcentration, leaves the myocardium highly susceptible to fatal arrhythmias [4]. In Kwashiorkor, a diet severely deficient in protein but relatively adequate in carbohydrates prevents the protective rise in cortisol and epinephrine, leading to a failure to mobilize muscle proteins. Consequently, hepatic synthesis of albumin ceases, intravascular oncotic pressure drops, and profound third-spacing (edema) ensues [3].
4. Classification
The classification of nutritional disorders categorizes the chronicity and severity of the deficit, guiding both prognosis and treatment.
4.1 Acute vs. Chronic
The World Health Organization strictly delineates acute from chronic malnutrition based on anthropometric indices. Acute malnutrition (wasting) reflects a recent and severe process that leads to substantial weight loss, measured by a low Weight-for-Height Z-score (WHZ) [6]. Conversely, chronic malnutrition (stunting) is the result of prolonged nutritional deprivation leading to skeletal growth retardation, measured by a low Height-for-Age Z-score (HAZ) [1].
4.2 Severity Grading (Hypotrophy)
In many Eastern European and Central Asian schools, chronic nutritional disorders in infants are classified by the degree of "Hypotrophy":
Ø Grade I (Mild): Weight deficit of 10-20%. Subcutaneous fat is thinned only on the abdomen.
Ø Grade II (Moderate): Weight deficit of 20-30%. Fat is lost on the abdomen, torso, and limbs. The child shows delayed motor milestones and reduced immunological resistance [5].
Ø Grade III (Severe - Marasmus): Weight deficit >30%. Subcutaneous fat is completely absent, including on the face (Bichat's fat pads), giving the child an aged or "senile" appearance [2].

Figure 2. The WHO Child Growth Standards chart for Weight-for-Length Z-scores. Severe Acute Malnutrition (SAM) is defined objectively by a Z-score of <-3 standard deviations from the median, serving as the universal diagnostic threshold for intensive intervention.
(Source: World Health Organization (WHO), Child Growth Standards, 2006).
5. Clinical Presentation
The clinical manifestations of Severe Acute Malnutrition (SAM) lie on a spectrum, primarily categorized into two distinct clinical syndromes: Marasmus and Kwashiorkor.
5.1 Marasmus (Non-edematous SAM)
Marasmus represents the end-stage of chronic energy deficiency. The child is severely wasted, with an absolute loss of subcutaneous adipose tissue and skeletal muscle mass [6]. The classic presentation involves a prominent, oversized head relative to a tiny, emaciated body, and a face described as resembling an "old man" or monkey facies due to the loss of buccal fat pads. These children are typically irritable, intensely hungry, and possess a dangerously thin, "baggy pants" appearance to their gluteal skin [3].
5.2 Kwashiorkor (Edematous SAM)
Kwashiorkor presents a deceptive clinical picture because the child may not appear underweight due to massive fluid retention. The hallmark is bilateral pitting edema, initially appearing on the dorsum of the feet and progressing to generalized anasarca and ascites [4]. Clinically, these children are profoundly apathetic, anorexic, and demonstrate characteristic skin and hair changes. The "flaky paint" dermatosis (hyperpigmented plaques that desquamate to leave raw skin) and the "flag sign" (alternating bands of depigmented and normal hair reflecting periods of poor and adequate nutrition) are pathognomonic [5]. Hepatomegaly is universally present due to severe hepatic steatosis (fatty liver), as the liver cannot synthesize the apolipoproteins needed to export triglycerides [2].
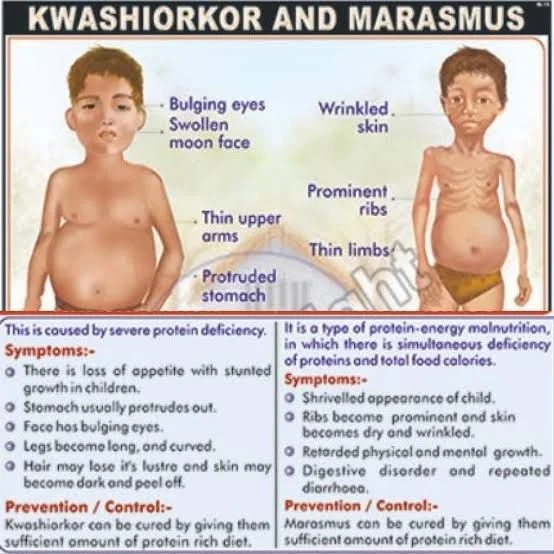
Figure 3. Clinical manifestations of Severe Acute Malnutrition. The left panel demonstrates the profound wasting and loss of subcutaneous fat characteristic of Marasmus. The right panel shows a child with Kwashiorkor, highlighting generalized edema, dermatosis, and hepatomegaly resulting from severe protein deficiency.
(Source: Manson's Tropical Diseases, 23rd ed., Elsevier, 2014).
6. Diagnosis and Laboratory Evaluation
Diagnosis relies fundamentally on rigorous anthropometry, supplemented by targeted laboratory investigations to assess the degree of systemic decompensation.
6.1 Anthropometry
The gold standard for diagnosing acute nutritional disorders is the calculation of Z-scores based on WHO growth charts. Severe Acute Malnutrition is definitively diagnosed by a WHZ score < -3, the presence of bilateral pitting edema, or a Mid-Upper Arm Circumference (MUAC) < 115 mm in children aged 6 to 59 months [6]. MUAC is highly sensitive for mortality risk and is the preferred tool for rapid outpatient screening [1].
6.2 Laboratory Diagnostics
While routine labs do not define malnutrition, they are critical for detecting hidden metabolic crises. Hypoglycemia (blood glucose < 3 mmol/L or 54 mg/dL) and severe hypokalemia are the most immediate threats to life [3]. Complete blood counts often reveal a normocytic, normochromic anemia (due to adaptive reduction in oxygen demand) or a dimorphic anemia reflecting combined iron and folate deficiencies. Total serum proteins and albumin are drastically reduced in Kwashiorkor, though they may remain deceptively normal in Marasmus due to hemoconcentration [5]. Liver function tests may show elevated transaminases, reflecting hepatocellular injury from steatosis or concurrent occult infections [2].
7. Treatment and Rehabilitation
The treatment of Severe Acute Malnutrition is a medical emergency that must be executed with extreme caution. Rapid, aggressive feeding invariably leads to Refeeding Syndrome—a fatal shift of potassium, magnesium, and phosphate into cells triggered by an insulin surge, leading to arrhythmias and cardiopulmonary collapse [4].
Therefore, the WHO dictates a strict 10-step protocol, divided into an Initial Stabilization Phase (Days 1-7) and a Rehabilitation Phase (Weeks 2-6) [6].
7.1 Stabilization Phase (Treating the Crises)
The first priority is not to make the child gain weight, but to restore cellular function and prevent death [1].
ü Hypoglycemia & Hypothermia: Treated immediately with 10% dextrose IV or oral glucose, and aggressive warming.
ü Dehydration: Managed with specialized Rehydration Solution for Malnutrition (ReSoMal), which is lower in sodium and higher in potassium than standard ORS, to prevent fatal fluid overload and heart failure [5].
ü Electrolytes: Universal supplementation of Potassium and Magnesium is initiated, while IV Sodium is strictly avoided [3].
ü Infection: Because malnutrition blunts the inflammatory response (no fever, no leukocytosis), broad-spectrum antibiotics (e.g., Ampicillin + Gentamicin) are administered empirically to all children with SAM [6].
ü Cautious Feeding: Enteral feeding is initiated using F-75 therapeutic milk (75 kcal/100mL), designed to provide low protein and low sodium to gently restart the metabolic machinery without overwhelming the liver or kidneys [2].
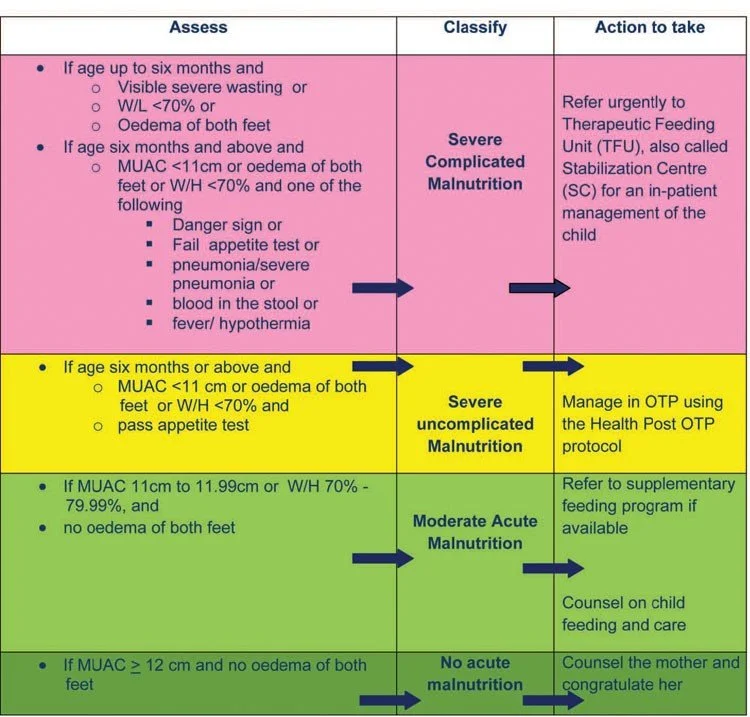
Figure 4. The WHO 10-Step Algorithm for the inpatient management of Severe Acute Malnutrition. The protocol emphasizes that the initial phase strictly avoids caloric loading, focusing instead on correcting metabolic disturbances, infections, and electrolyte shifts to prevent Refeeding Syndrome.
(Source: World Health Organization, Guideline: Updates on the management of severe acute malnutrition in infants and children, 2013).
7.2 Rehabilitation Phase
Once the child’s appetite returns and edema resolves, they enter the rehabilitation phase. The diet is transitioned to F-100 milk (100 kcal/100mL) or Ready-to-Use Therapeutic Food (RUTF) like Plumpy'Nut [6]. This phase aims for rapid catch-up growth. Crucially, emotional and sensory stimulation (play therapy) is integrated to reverse the profound cognitive and developmental delays associated with chronic malnutrition [4].
8. Conclusion
Acute and chronic nutritional disorders in pediatric practice represent a profound failure of the biological ecosystem surrounding a child. The pathophysiology extends far beyond a simple lack of calories, involving a total-body endocrine and metabolic adaptation that leaves the child exceptionally fragile. Accurate diagnosis through anthropometric Z-scores and clinical recognition of the Marasmus-Kwashiorkor spectrum is essential. Above all, clinicians must adhere to structured, phased rehabilitation protocols. Recognizing that the starving child is functionally a different physiological entity prevents the iatrogenic catastrophe of refeeding syndrome and offers the best chance at complete physiological and cognitive recovery.
9. References
[1] Kliegman, R. M., St. Geme, J. W., Blum, N. J., Shah, S. S., & Tasker, R. C. (2020). Nelson Textbook of Pediatrics (21st ed.). Elsevier. ISBN: 978-0323529501.
[2] Jameson, J. L., Fauci, A. S., Kasper, D. L., Hauser, S. L., Longo, D. L., & Loscalzo, J. (2022). Harrison's Principles of Internal Medicine (21st ed.). McGraw Hill. ISBN: 978-1264268504.
[3] Farrar, J., Hotez, P. J., Junghanss, T., Kang, G., Lalloo, D., & White, N. J. (2014). Manson's Tropical Diseases (23rd ed.). Elsevier. ISBN: 978-0702051012.
[4] Nichols, B. L., & Garza, C. (2015). History of Pediatric Malnutrition. Pediatric Clinics of North America, 62(5), 1059-1070. https://doi.org/10.1016/j.pcl.2015.05.001
[5] Waterlow, J. C. (2006). Protein-Energy Malnutrition. Edward Arnold Publishers. ISBN: 978-0340531536.
[6] World Health Organization. (2013). Guideline: Updates on the management of severe acute malnutrition in infants and children. Geneva: WHO. https://apps.who.int/iris/handle/10665/95584
