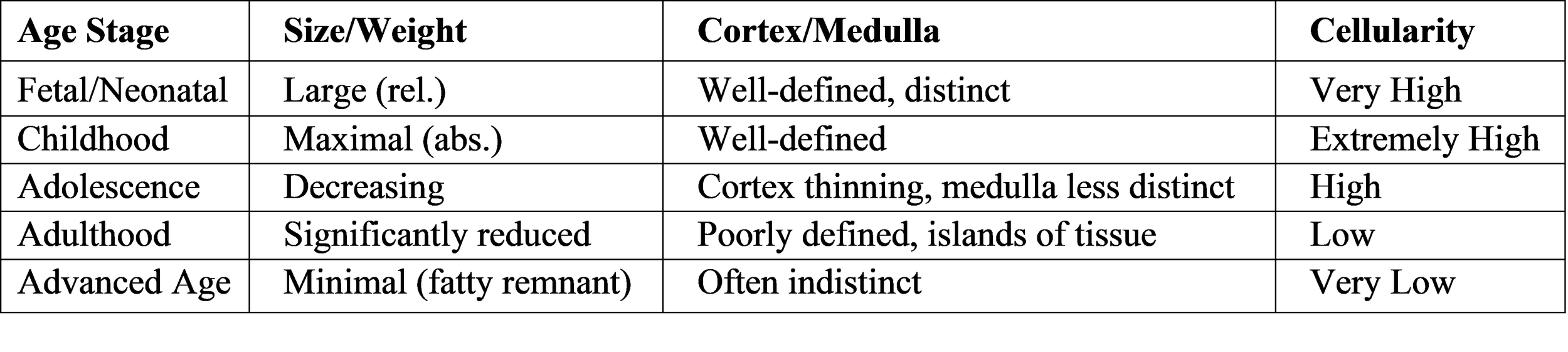Comparative Analysis of Age-Related Morphological and Functional Changes in the Human Thymus
1. Manas kyzy Uulkan
2. Kaamesh Kasipandi
Rubini Ramesh
(1. Lecturer, Dept. of Histology, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.
2. Students, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.)
Abstract
The thymus gland plays a pivotal role as the primary lymphoid organ in the maturation and selection of a functional T-cell repertoire that is capable of self-tolerance. Unlike most other organs, it begins to gradually regress and lose its function through the so-called involution process early in life. This paper presents a complete and comparative analysis of the thymus in different age categories: fetal development, infancy, childhood, pubescence, and senescence. A systematic literature reviews describes the dramatic histological, cellular, and molecular changes at each age stage. Features considered include thymic volume, cellularity, stromal microenvironment, epithelial organization, and T-cell output. Age-related thymic involution is a major contributing factor to immunosenescence, susceptibility to infections, reduced vaccine effectiveness in older adults, and the occurrence of autoimmune diseases and cancer. Research into rejuvenation therapies (hormonal, cytokine, regenerative) is also discussed. Understanding these age-specific characteristics is crucial for interventions to control immune function throughout life.
Keywords: Thymus, Involution, Immunosenescence, Thymopoiesis, T-cell development, Aging, Stroma, Rejuvenation.
INTRODUCTION
The thymus, the primary organ for T lymphocyte production and development, is a two- lobed structure in the anterior mediastinum. It is dynamic, with rapid growth and full functionality during childhood, followed by prolonged regression. The gradual replacement of thymic tissue with fat, termed thymic involution, is a major factor in immune system aging (immunosenescence) [Lynch et al., 2009].
The thymus educates T-cells to distinguish self from non-self through thymopoiesis: bone marrow progenitor cells migrate to the thymus, undergo T-cell receptor (TCR) gene rearrangement, and are selected based on TCR affinity for self-MHC molecules [Klein et al., 2014]. This selection occurs in a microenvironment of thymic epithelial cells (TECs), dendritic cells, and fibroblasts, which provide cytokines, chemokines, and cell-cell interactions.
Studying age-related thymic changes is essential for understanding immune development, age-related immune decline, clinical practices (vaccination timing, disease treatment, immune recovery), and identifying targets for rejuvenation therapies.
METHODS
A systematic literature review was conducted using PubMed, Scopus, and Web of Science, focusing on publications from the last 30 years, with emphasis on the last decade. Extracted data included thymic morphology (size, weight, imaging), histology (cortical- medullary organization, adiposity), cellular composition (thymocyte subsets, TEC sub- sets, stromal cells), molecular markers (cytokines, transcription factors, hormones), and functional outputs (TRECs, RTEs). Data were compared across age groups: fetal/neonatal (0–2 years), childhood (2–12 years), adolescence/young adulthood (12–25 years), adult- hood (25–60 years), and advanced age (60+ years). Summary tables were created to highlight comparative features and molecular mechanisms.
RESULTS
Age-Related Comparative Histological and Morphological Features
The thymus undergoes dramatic morphological changes throughout life.
Fetal and Neonatal Period (0–2 years): The thymus originates from the third pharyngeal pouches and is fully developed at birth. Histologically, it shows distinct cortical and medullary regions. The cortex contains proliferating, immature double- positive (CD4+ CD8+) thymocytes. The medulla contains mature single-positive (CD4+ or CD8+) thymocytes and Hassall’s corpuscles (sites of Treg generation). The stroma has abundant cortical and medullary TECs (cTECs, mTECs) with minimal fat [Haynes & Hale, 1998].
Childhood (2–12 years): The thymus reaches maximum size and weight relative to body size and maintains high activity, producing naïve T-cells. Cortical and medullary layers are distinct. Early involution signs appear around puberty, with perivascular lymphocyte infiltration and small fat droplets [Bodey et al., 1997].
Adolescence and Young Adulthood (12–25 years): Active thymic involution begins. Size and cellularity decline, cortex thins, medulla becomes indistinct, and adipo- genesis progresses. The stromal epithelial network breaks down [Steinmann, 1986].
Adulthood (25–60 years): Involution progresses steadily. By middle age, thymic parenchyma is largely replaced by adipose and fibrous tissue. Remaining functional tissue forms isolated islands. Corticomedullary junctions are indistinguishable. Few atrophic TECs remain. Baseline thymopoiesis persists at low levels [Sutherland et al., 2005].
Table 1: Comparative Histological and Functional Features of the Thymus Across the Lifespan
Functional Consequences: From Thymic Output to Peripheral Immunity
Thymic activity is measured by export of new naïve T cells (Recent Thymic Emigrants, RTEs), indirectly assessed via T-cell Receptor Excision Circles (TRECs).
Output Decline: TREC levels are highest in cord blood and early childhood, decline rapidly through adolescence and young adulthood, and continue a slow decline. By age 70, thymic output is < 10% of peak capacity [Douek et al., 1998].
Peripheral T-cell Pool Composition: Reduced thymic output shrinks and di- versifies the naïve T-cell pool. Homeostatic proliferation expands existing T-cells into memory-like cells. The elderly T-cell repertoire is dominated by expanded memory and effector clones prone to senescence and exhaustion [Nikolich-Žugich, 2014].
Clinical Immunological Deficits: Manifestations include:
• Increased susceptibility to infections (e.g., severe COVID-19, herpes zoster reacti- vation)
• Poor vaccine responses due to limited high-affinity T-cell help
• Increased cancer incidence due to impaired immune surveillance
• Paradoxical autoimmunity from impaired negative selection
DISCUSSION
Thymic involution is a complex process involving histological, cellular, and molecular changes. Foxn1 downregulation is a key molecular event; forced expression rejuvenates the thymus in aged mice. Pubertal hormonal changes trigger involution, but progression beyond hormone plateauing implicates accumulated cellular damage, altered stem cell function, and inflammation.
Clinically, age-related thymic characteristics are crucial. In pediatric oncology, the robust thymus often recovers post-chemotherapy. In adult HSCT, poor thymic recovery causes prolonged immunodeficiency, opportunistic infections, and relapse. Thus, the thymus is a key therapeutic target.
Thymic Rejuvenation Strategies
Emerging research aims to boost or reverse thymic involution:
Hormonal Modulation: GH, IGF-1, or GH secretagogues. Trials show increased thymic mass and naïve T-cells in HIV patients and HSCT survivors [Napolitano et al., 2008].
Cytokine Therapy: IL-7 or IL-22 to support thymocytes and TECs.
Sex Steroid Inhibition: LHRH antagonists to block sex steroid signaling, im- proving T-cell recovery post-HSCT.
Stromal Cell and Regenerative Therapy: Administering TEC progenitors or bioengineered thymic organoids.
Targeted Gene Therapy: Restoring Foxn1 expression in aged TECs.
Challenges include autoimmunity risk (from poorly selected new T-cells) and cancer pro- motion (from stimulated proliferation). The goal is to enhance residual function to clinically acceptable levels, not fully restore a juvenile thymus.
CONCLUSION
The thymus undergoes profound age-related changes from robust activity to quiescent remnant. Involution is complex, involving histological, cellular, and molecular alterations. Declining thymopoiesis due to Foxn1 downregulation, hormonal changes, and inflammation leads to a poorer peripheral T-cell repertoire, driving immunosenescence. This compromises infection and cancer defense, and reduces vaccine efficacy in the elderly. A biological trade-off exists: the high-output thymus of youth is sacrificed in old age, impacting an aging population. However, thymic plasticity offers hope. Rejuvenation strategies targeting hormones, cytokines, or epithelium itself show promise for clinical translation. Future research should focus on safe, effective therapies to enhance immune competence in the elderly, immunosuppressed patients, and those with congenital thymic defects. Modulating thymic function could significantly promote immune health across the lifespan.
References
Bodey, B., Bodey, V., Siegel, S. E., & Kaiser, H. E. (1997). Involution of the mam- malian thymus, one of the leading regulators of aging. In Vivo (Athens, Greece), 11(5), 421–440.
Bredenkamp, N., Nowell, C. S., & Blackburn, C. C. (2014). Regeneration of the aged thymus by a single transcription factor. Development (Cambridge, England), 141(8), 1627–1637.
Douek, D. C., McFarland, R. D., Keiser, P. H., Gage, E. A., Massey, J. M., Haynes,
B. F., ... & Koup, R. A. (1998). Changes in thymic function with age and during the treatment of HIV infection. Nature, 396(6712), 690–695.
Dudakov, J. A., Hanash, A. M., & van den Brink, M. R. (2015). Interleukin-22: Immuno- biology and pathology. Annual Review of Immunology, 33, 747–785.
Gruver, A. L., Hudson, L. L., & Sempowski, G. D. (2007). Immunosenescence of ageing.
The Journal of Pathology, 211(2), 144–156.
Haynes, B. F., & Hale, L. P. (1998). The human thymus. A chimeric organ comprised of central and peripheral lymphoid components. Immunologic Research, 18(2), 61–78.
Klein, L., Kyewski, B., Allen, P. M., & Hogquist, K. A. (2014). Positive and negative selection of the T cell repertoire: What thymocytes see (and don’t see). Nature Reviews Immunology, 14(6), 377–391.
Lynch, H. E., Goldberg, G. L., Chidgey, A., Van den Brink, M. R., Boyd, R., & Sem- powski, G. D. (2009). Thymic involution and immune reconstitution. Trends in Im- munology, 30(7), 366–373.
Napolitano, L. A., Schmidt, D., Gotway, M. B., Ameli, N., Filbert, E. L., Ng, M. M., ... & McCune, J. M. (2008). Growth hormone enhances thymic function in HIV-1-infected adults. The Journal of Clinical Investigation, 118(3), 1085–1098.
Nikolich-Žugich, J. (2014). Aging of the T cell compartment in mice and humans: From no naive expectations to foggy memories. Journal of Immunology (Baltimore, Md.: 1950), 193(6), 2622–2629.
Palmer, D. B. (2013). The effect of age on thymic function. Frontiers in Immunology, 4, 316.
Steinmann, G. G. (1986). Changes in the human thymus during aging. Current Topics in Pathology, 75, 43–88.
Sutherland, J. S., Goldberg, G. L., Hammett, M. V., Uldrich, A. P., Berzins, S. P., Heng,
T. S., ... & Boyd, R. L. (2005). Activation of thymic regeneration in mice and humans following androgen blockade. Journal of Immunology (Baltimore, Md.: 1950), 175(4), 2741–2753.
Taub, D. D., & Longo, D. L. (2005). Insights into thymic aging and regeneration. Im- munological Reviews, 205, 72–93.