The Evolutionary Arms Race: Unraveling the Multifactorial Drivers and Global Trajectory of Antimicrobial Resistance
1. Kurmanaliev Nurlan
2. Kaveri Batwal
Krushna Jagtap
Kavita Choudhary
Aniket Mahajan
Anisha Chougale
(1. Teacher, International Medical Faculty, Osh State University, Kyrgyzstan)
(2. Students, International Medical Faculty, Osh State University, Kyrgyzstan)
Abstract
The world faces a serious health emergency because antimicrobial resistance (AMR) en- dangers all existing medical achievements which have taken decades to build and makes it harder to stop and treat all types of bacterial and parasitic and viral and fungal infections. This article aims to synthesize the current understanding of the evolutionary mechanisms, epidemiological trends, and systemic drivers of AMR, with a particular focus on bacterial pathogens.
The paper uses a narrative review method to present research results which emerged from a complete assessment of academic studies and international health organization surveillance data and important scientific studies published between 2000 and 2023.
The analysis finds that resistance stems from multiple interrelated elements which create a complicated resistance pattern. Resistance occurs through spontaneous genetic mutations and, more importantly, the horizontal gene transfer of resistance genes that occur through mobile genetic elements. Epidemiological evidence demonstrates that priority pathogens together with carbapenem-resistant Acinetobacter baumannii and fluoroquinolone-resistant
E. coli create a worldwide health crisis. The main factors that create this medical emergency result from excessive and incorrect usage of antimicrobial drugs throughout human healthcare agricultural practices and environmental settings together with the absence of new antibiotic development.
Conclusion: AMR operates as a One Health problem which requires multi sector cooper- ation between different organizations to solve. The implementation of effective antimicrobial reduction strategies must establish antimicrobial control practices together with strong in- fection control methods and worldwide monitoring systems and financial support for new treatment and diagnostic solutions. The world faces a serious risk of returning to pre- an- tibiotic times unless we implement strong measures.
Keywords: Antimicrobial Resistance, Antibiotic Resistance, One Health, Horizontal Gene Transfer, Drug Development, Public Health
INTRODUCTION
The 20th century saw the development of antibiotics which changed medical practice by making previously deadly infections treatable while creating new possibilities for surgical procedures and cancer treatments and organ transplants (Aminov 858). The medical breakthrough requires humans to overcome a biological law which governs all living things. Antimicrobial agents lead to selective pressure which benefits resistant microorganisms through their ability to survive and reproduce. The phenomenon of antimicrobial resistance (AMR) has reached a state of emergency which endangers the medical advancements of the past hundred years.
The situation requires immediate action because the available statistics demonstrate its critical nature. A landmark study published in The Lancet estimated that bacterial AMR was associated with 4.95 million deaths globally in 2019, with 1.27 million of those being directly attributable to resistance (Murray et al. 631). The figures show that AMR ranks among the top worldwide death causes which match the combined death rates of HIV/AIDS and malaria. The World Health Organization (WHO) has declared AMR one of the top ten global public health threats facing humanity (WHO, “Antimicrobial resistance”).
AMR exists as a complex biological process which exhibits multiple dynamic elements. Bac- teria can develop resistance through two primary genetic pathways which include intrinsic re- sistance and acquired resistance. Acquired resistance, which presents the most serious threat to the ongoing crisis, develops through chromosomal mutations and through horizontal gene transfer (HGT) which enables bacteria to acquire foreign resistance genes from other bacteria (Davies and Davies 348).
The drivers of AMR extend far beyond the clinical setting. The “One Health” framework recognizes that the health of people is connected to the health of animals and the shared en- vironment. The combination of livestock antibiotic misuse for growth promotion and disease prevention together with active pharmaceutical ingredient discharge from manufacturing and human waste into water systems establishes extensive resistance gene reservoirs which create selective pressure throughout ecosystems (Robinson et al. 5).
The purpose of this article is to present complete information about current AMR research. The study will investigate how resistance mechanisms work at the genetic level. The study will investigate genetic resistance mechanisms and provide current epidemiological information about major pathogens and examine various factors that contribute to the issue through the One Health framework and assess present- day developments in medical research. The research paper presents a solution to the AMR problem through implementation of an international solution which combines biological behavioral and systemic approaches to address the fundamental causes of the emergency.
METHODS
The article presents itself as a complete narrative review which covers all relevant research about the subject matter. The research team conducted a systematic review of scientific literature which was not complete to obtain the information used in the study. The main databases used for research were PubMed and Google Scholar and Scopus. The search used different combinations of the following search terms “antimicrobial resistance” and “antibiotic resistance” and “horizontal gene transfer” and “mechanisms of resistance” and “ESKAPE pathogens” and “antibiotic stewardship” and “One Health” and “antibiotic pipeline” and “global burden of AMR.”
The research focused on peer- reviewed original research and meta- analyses and authorita- tive reports which were published between 2000 and 2023. The World Health Organization’s Global Antimicrobial Resistance and Use Surveillance System (GLASS) reports and the CDC’s Antibiotic Resistance Threats Reports and the Global Research on Antimicrobial Resistance (GRAM) project served as primary sources for epidemiological data. The WHO reports and Pew Charitable Trusts reports provided essential information which researchers used to study the antibiotic pipeline. The selection of studies and data was guided by their relevance, impact, and contribution to the contemporary understanding of AMR.
RESULTS
The Genetic and Molecular Mechanisms of Resistance
Bacteria show exceptional genetic flexibility through their ability to acquire resistance. Bacteria use horizontal gene transfer (HGT) as their main method for sharing resistance genes because this process enables them to transfer genetic material between different bacterial species and genera. HGT occurs through three primary pathways which include:
1. Conjugation: Bacteria transfer plasmids which are small circular DNA molecules through their pilus. This method serves as the main path for spreading multiple drug resistance.
2. Transformation: A competent bacterium takes in free DNA which exists in its environment.
3. Transduction: Bacterial DNA moves between cells when a bacteriophage virus that infects bacteria transmits it.
Mobile genetic elements contain resistance gene collections which function as integrons or transposons and enable fast development of multi- drug resistant traits. Bacteria use multiple chemical methods to destroy antimicrobial effects after they gain a resistance gene or make a mutation. The document presents a summary of these mechanisms in Table 1.
Table 1: Common Biochemical Mechanisms of Antibiotic Resistance

The Global Epidemiology of AMR: A Focus on Priority Pathogens
The ESKAPE pathogens which belong to Enterococcus faecium Staphylococcus aureus Kleb- siella pneumoniae Acinetobacter baumannii Pseudomonas aeruginosa and Enterobacter species present the greatest healthcare threat through AMR which exists as a major risk in medical fa- cilities that treat patients with serious infections. The WHO has established a list of pathogens which research and development efforts should focus on, with three priority levels (critical, high, medium) that reflect resistance patterns and public health threats (WHO, “Global priority list”). The WHO Global Antimicrobial Resistance and Use Surveillance System (GLASS) shows worldwide resistance rates which have reached dangerous levels according to its data. The treatment options become extremely limited because more than 50% of E. coli strains in multiple countries have developed resistance to third- generation cephalosporins, which are common treatment options for urinary tract and bloodstream infections. Table 2 summarizes the global burden of resistance for key pathogen- drug combinations.
Table 2: Global Burden of Key Antimicrobial-Resistant Pathogens (Adapted from GRAM 2019)
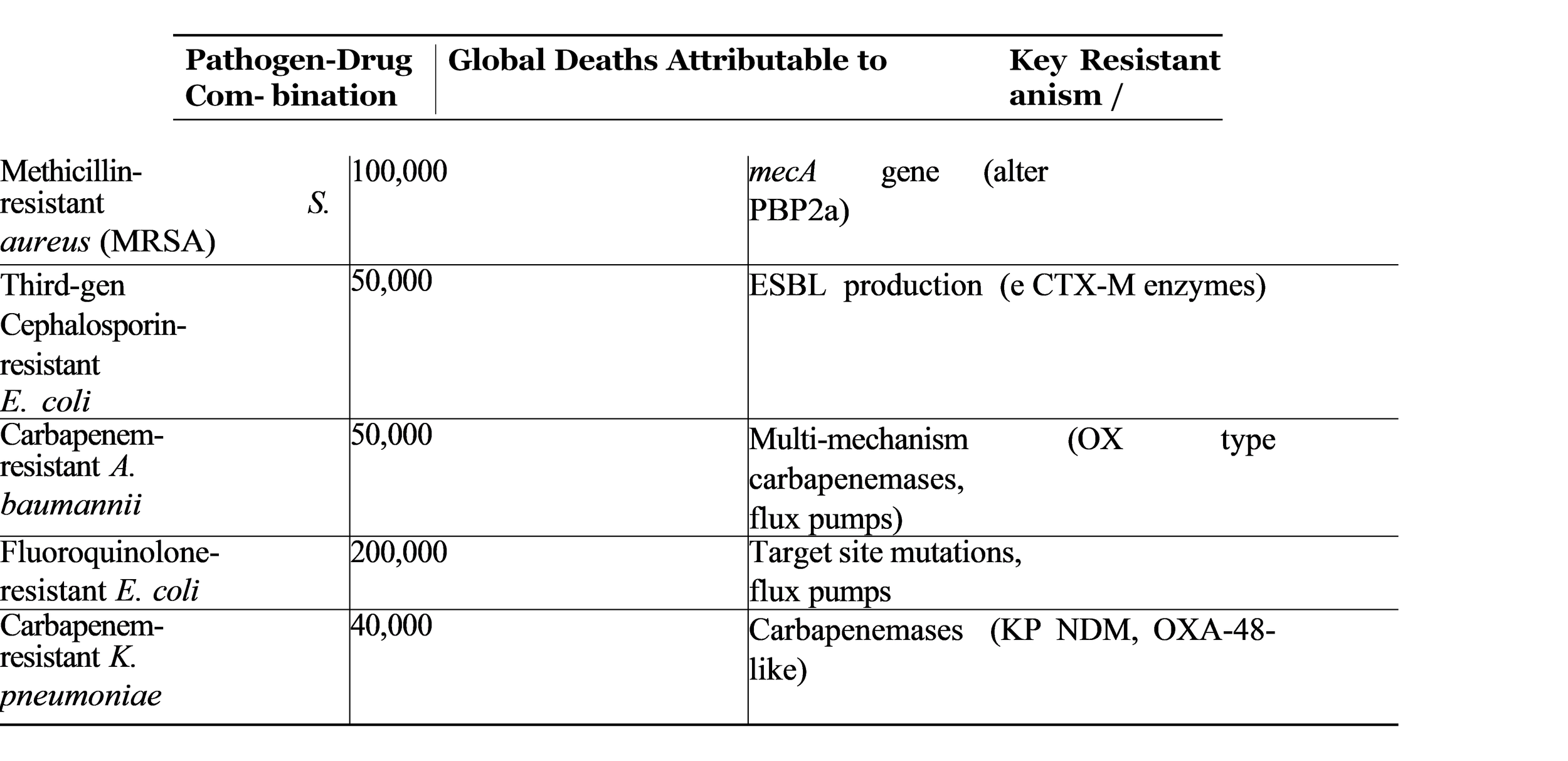
The Drivers of the AMR Crisis: A One Health Perspective
The emergence and spread of AMR requires multiple explanations to understand its develop- ment. The issue consists of three interconnected domains which include human health and animal health and agricultural systems and environmental protection efforts.
1. Human Health Sector:
The most basic cause of the problem stem from humans who misuse and overuse antibiotics in their medical treatment. This includes:
• Inappropriate Prescribing: Doctors give patients antibiotics to treat viral infections which include common cold and influenza despite the fact that these medications will not work.
• Broad-Spectrum Overuse: The medical field uses strong broad- spectrum antibiotics when doctors should select narrow- spectrum antibiotics which would achieve the same treatment results.
• Patient Non-Adherence: Patients who do not finish their antibiotic treatment will kill bacteria that are vulnerable to the medication but let resistant bacteria continue to live and grow.
• Poor Infection Prevention and Control (IPC): Healthcare facilities lack proper hand hygiene and sterilization practices which enable resistant organisms to spread between patients (Laxminarayan et al. 743).
2. Animal Health and Agriculture Sector:
The use of antibiotics in food- producing animals is a major contributor. For many years farmers have used antibiotics to treat sick animals and to make healthy animals grow faster while preventing diseases through the practice of giving subtherapeutic doses in their feed. This practice creates a massive reservoir of resistance. Bacteria which have developed resistance can spread to humans through direct animal contact and through contaminated meat in the food chain and through environmental contamination (Manyi-Loh et al. 5).
3. Environmental Sector:
The environment serves as a mixing bowl which combines all sources of resistance. The sources which create contamination include the following:
• Pharmaceutical Manufacturing Waste: Factory discharges from antibiotic produc- tion facilities contain high drug concentration which create an environment where resistant bacteria can thrive.
• Human and Animal Waste: Treated and untreated sewage from hospitals and commu- nities together with farm manure results in soil and water systems which contain antibiotics and resistant bacteria and resistance genes (Berendonk et al. 177).
The Innovation Gap: A Drying Antibiotic Pipeline
The problem of increasing resistance to existing antibiotics becomes worse because new antibi- otics no longer enter the market. The “golden era” of antibiotic discovery, which lasted from 1940 to 1960, established an “innovation gap” that now exists. The primary reasons are scientific and economic:
• Scientific Challenges: The “low-hanging fruit” of easily discovered natural products has been picked. The scientific process requires high expenses to discover new antibiotic classes which operate through different mechanisms than existing drugs.
• Economic Disincentives: Antibiotics are less profitable than drugs for chronic diseases (like diabetes or high blood pressure). The drugs have low sales volume because patients use them for brief periods and doctors reserve them as last-line treatments. The market failure has driven pharmaceutical companies to halt their antibiotic development programs.
The WHO states that current clinical pipelines fail to provide adequate solutions for the escalating threat of antimicrobial resistance. Researchers are developing new antibiotics that exist as modified versions of existing drug classes which show minimal effectiveness against the most dangerous drug- resistant bacteria (WHO, “2021 Antibacterial Agents in Clinical and Preclinical Development”).
DISCUSSION
The evidence presented demonstrates that the world currently participates in an evolutionary arms race which it is losing. The issue extends beyond biology because it originates from human conduct and economic systems and inadequate policy frameworks. The genetic promiscuity of bacteria (HGT, MDR) interacts with human activities (misuse in humans, animals, and the environment) to establish a situation which continuous creates new problems for society.
The GRAM project and GLASS data demonstrate that AMR exists as an active threat which results in millions of deaths every year. The elevated incidence of E. coli and K. pneu- moniae pathogens demonstrates that initial treatments fail while medical professionals turn to carbapenem which has become ineffective because of carbapenemase transmission. Geographic resistance rate differences demonstrate how healthcare systems and sanitary conditions and regulatory practices create unfair treatment outcomes between different regions. Areas which lack strong healthcare systems and experience high rates of infectious diseases face the greatest challenges which develop into a continuous cycle that produces both poverty and sickness.
The One Health framework is not an abstract concept but a necessary lens through which to view the entire problem. The release of antibiotics from farms and pharmaceutical plants into the environment creates selective pressure on environmental microbiomes which develops environmental “resistomes” that produce new resistance genes for human pathogens. The ex- isting interconnectivity between healthcare facilities and agricultural antibiotic usage requires implementation of intervention measures that extend beyond hospital boundaries.
The solution to this complex problem needs a global approach which includes multiple es- sential elements.
1. Antimicrobial Stewardship (AMS): Hospitals and clinics and community programs must establish procedures that require doctors to prescribe antibiotics only when their patients need them in proper amounts for precise time periods using the smallest effective dosage. The system needs advanced diagnostic technologies which can differentiate be- tween viral and bacterial infections while detecting the particular pathogen together with its resistance patterns. The medical field needs to develop point-of-care rapid diagnostic testing systems.
2. Infection Prevention and Control (IPC): The healthcare system needs enhanced IPC measures which include handwashing practices and environmental sanitation.
3. Global Surveillance: The international health community needs to expand and unify its existing worldwide monitoring systems which include GLASS to monitor antibiotic resistance patterns and identify infection hotspots while assessing treatment outcomes. The system needs to monitor human populations together with animal species and environ- mental ecosystems.
4. Reducing Agricultural Use: The government needs to create and execute new rules which will eliminate the use of essential medical antibiotics in livestock for both growth and regular disease management. The key to reducing antibiotic dependencies in animal husbandry requires the adoption of superior animal management techniques.
5. Incentivizing Innovation: The current market system for new antibiotic development requires the establishment of fresh economic frameworks. The development of new antibiotics relies on two types of financial mechanisms which include “push” incentives that provide research funding for early-stage development and “pull” incentives which offer market entry rewards that enable companies to earn profits without direct connections between their revenues and actual antibiotic sales.
6. Environmental Stewardship: The organization needs to allocate funding toward water and sanitation system development projects which will decrease antibiotic and resistant bacterial environmental contamination. The government requires all organizations to fol- low established.
CONCLUSION
Antimicrobial resistance is one of the most complex and urgent challenges of the 21st century. It is a slow- moving pandemic that has been building for decades, fueled by the invisible hand of evolution and the visible failures of human stewardship. The findings of this review underscore that AMR is not merely a medical problem but a profound societal challenge, embedded in our healthcare practices, agricultural systems, and environmental waste streams.
The consequences of inaction are catastrophic. If current trends continue, common infections will once again become untreatable, routine surgeries will carry a high risk of life-threatening complications, and the gains made in maternal and child mortality will be reversed. The eco- nomic toll, in terms of lost productivity and healthcare costs, will be devastating.
However, the situation is not hopeless. The biological mechanisms of resistance are well- understood, the drivers are identifiable, and the solutions, while difficult, are clear. Averting a post- antibiotic era requires a paradigm shift in our relationship with these precious drugs. They must be treated as a shared global resource, to be conserved through stewardship, protected through prevention, and replenished through innovation. This demands a unified, global effort under the One Health banner—an effort that transcends national borders and disciplinary silos. The future of modern medicine depends on our collective ability to win this.
References
[1] Aminov, Rustam I. “A Brief History of the Antibiotic Era: Lessons Learned and Chal- lenges for the Future.” Frontiers in Microbiology, vol. 1, 2010, pp. 134-148. Frontiers, doi:10.3389/fmicb.2010.00134.
[2] Berendonk, Thomas U., et al. “Tackling Antibiotic Resistance: The Environmental Framework.” Nature Reviews Microbiology, vol. 13, no. 5, 2015, pp. 310-317. Nature, doi:10.1038/nrmicro3439.
[3] Davies, Julian, and Dorothy Davies. “Origins and Evolution of Antibiotic Resistance.” Mi- crobiology and Molecular Biology Reviews, vol. 74, no. 3, 2010, pp. 417-433. ASM Journals, doi:10.1128/MMBR.00016-10.
[4] Laxminarayan, Ramanan, et al. “Antibiotic Resistance—the Need for Global Solutions.” The Lancet Infectious Diseases, vol. 13, no. 12, 2013, pp. 1057-1098. The Lancet, doi:10.1016/S1473-3099(13)70318-9.
[5] Manyi-Loh, Christy, et al. “Antibiotic Use in Agriculture and Its Consequential Resistance in Environmental Sources: Potential Public Health Implications.” Molecules, vol. 23, no. 4, 2018, p. 795. MDPI, doi:10.3390/molecules23040795.
[6] Murray, Christopher J. L., et al. “Global Burden of Bacterial Antimicrobial Resistance in 2019: A Systematic Analysis.” The Lancet, vol. 399, no. 10325, 2022, pp. 629-655. The Lancet, doi:10.1016/S0140-6736(21)02724-0.
Rice, Louis B. “Federal Funding for the Study of Antimicrobial Resistance in Nosocomial Pathogens: No ESKAPE.” The Journal of Infectious Diseases, vol. 197, no. 8, 2008, pp. 1079-1081.
