Brain Regions and Their Functions
1. Tashimbetova Umut Kaparovna
2. Rahul
Hemashree
Merlin
(1. Teacher, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic
2. Students, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic)
Abstract
The human brain, a structure weighing approximately 1.4 kilograms and containing an estimated 86 billion neurons, represents the most complex biological system known to science. Understanding the relationship between discrete brain regions and their specialised functions has been a central pursuit of neuroscience for over a century, progressing from early lesion studies and postmortem examinations to contemporary neuroimaging techniques such as functional magnetic resonance imaging, diffusion tensor imaging, and positron emission tomography. This review synthesises current knowledge regarding the major anatomical divisions of the human brain, from the cerebral cortex with its four distinct lobes to subcortical structures including the basal ganglia, thalamus, hypothalamus, limbic system, and the infratentorial compartments comprising the brainstem and cerebellum. Each region is examined in terms of its cytoarchitecture, principal afferent and efferent connections, and the functional deficits that arise following damage. Particular attention is paid to hemispheric lateralisation, cortical specialisation as evidenced by functional topography, and the dynamic, network-based perspective that has superseded earlier models of strict localisation. The clinical relevance of regional neuroanatomy is emphasised throughout, providing a foundation for the diagnosis and management of neurological disorders ranging from stroke and traumatic brain injury to neurodegenerative diseases. By integrating classic anatomical knowledge with recent discoveries from connectomics and in vivo tractography, this review offers a contemporary framework for understanding how the brain’s diverse regions collaborate to produce perception, action, emotion, memory, and consciousness.
1. Introduction
Hold a human brain in your hands, and you are holding the product of hundreds of millions of years of evolution. Its surface is folded like a crumpled piece of paper, with ridges and valleys that allow an enormous expanse of neural tissue to fit within the confined space of the skull. Its consistency is that of firm tofu, surprisingly fragile despite its protective casing. Yet within this unassuming organ lies everything that makes us who we are: our memories, our personalities, our capacity for language and music and mathematics, our ability to recognise a loved one’s face, to feel joy or sorrow, to plan for the future and to reflect on the past. The question of how specific brain regions contribute to these functions has occupied philosophers and scientists since antiquity, but only in the last two centuries has systematic inquiry yielded answers that are both precise and clinically useful.
Early attempts to localise function to particular brain regions were often speculative, as exemplified by phrenology, the pseudoscientific doctrine popularised by Franz Joseph Gall in the early nineteenth century, which held that specific mental faculties resided in discrete regions of the cerebral cortex and could be inferred from the contours of the skull. Despite its flawed methodology, phrenology made a critical contribution to neuroscience: it advanced the principle of cortical localisation of function, a concept that would later receive rigorous validation from clinical observations. The most famous of these observations came from Paul Broca, who in 1861 examined the brain of a patient known as Tan, so named because this was virtually the only syllable he could produce following a stroke that had damaged the left inferior frontal gyrus. Broca’s subsequent demonstration that lesions in this region consistently produced expressive language deficits provided the first compelling evidence that a specific cognitive function could be localised to a specific cortical area. Shortly thereafter, Carl Wernicke identified a second language area in the left superior temporal gyrus, responsible for language comprehension rather than production, establishing the foundation for the modern understanding of cortical specialisation.
The twentieth century witnessed an explosion of knowledge regarding brain regionalisation. The development of stereotactic atlases, most notably the work of Jean Talairach and Rolf Turner, provided standardised coordinate systems for describing neuroanatomical locations. Advances in histological staining, particularly the Nissl and Golgi methods, enabled detailed characterisation of cytoarchitecture, leading to Brodmann’s parcellation of the cerebral cortex into 52 distinct areas based on cellular composition. The advent of in vivo neuroimaging in the 1970s and 1980s transformed the field further, allowing researchers to observe brain activity in healthy, behaving individuals for the first time. Functional magnetic resonance imaging, in particular, has enabled the mapping of countless cognitive functions onto specific brain regions, while simultaneously revealing the limitations of strict localisation models: most complex behaviours involve distributed networks rather than isolated areas.
Today, the prevailing view in cognitive neuroscience is one of functional specialisation within a highly interconnected, dynamic architecture. Certain regions are indeed specialised for particular operations: the primary visual cortex processes basic visual features, the hippocampus is critical for declarative memory formation, and the amygdala evaluates emotional significance. But these regions do not work in isolation; they are nodes within larger circuits, and their functions are context-dependent, shaped by ongoing interactions with other areas. This review will navigate this intricate landscape, proceeding from the most evolutionarily recent structures to the most ancient, examining each major brain region in turn and synthesising what is currently known about its contributions to the human experience.
2. Methods
This review was compiled through a comprehensive search of the peer-reviewed literature using the PubMed, Google Scholar, and Web of Science databases. Search terms included combinations of the following keywords: “brain region,” “cerebral cortex,” “frontal lobe,” “parietal lobe,” “temporal lobe,” “occipital lobe,” “insula,” “basal ganglia,” “thalamus,” “hypothalamus,” “amygdala,” “hippocampus,” “limbic system,” “brainstem,” “midbrain,” “pons,” “medulla oblongata,” “cerebellum,” “neuroanatomy,” “functional localisation,” and “connectivity.” The search was limited to articles published in English between January 2000 and December 2025, with priority given to systematic reviews, meta-analyses, and original research articles that employed contemporary neuroimaging or tractography methods. Landmark historical studies published prior to 2000 were included where they provided foundational knowledge that has not been superseded. Additionally, standard neuroanatomy textbooks including “Principles of Neural Science” by Kandel and colleagues and “The Human Nervous System” by Mai and Paxinos were consulted for anatomical reference. Data extraction focused on structural boundaries, cytoarchitectonic features, afferent and efferent connections as established by tracer studies in non-human primates and diffusion imaging in humans, and functional deficits associated with focal lesions or pharmacological inactivation. Given the narrative nature of this review, formal quality assessment using standardised instruments was not performed, but studies were excluded if they lacked sufficient methodological detail or if their findings had not been replicated by independent laboratories.
3. Results
3.1 The Cerebral Cortex: An Overview
The cerebral cortex, a thin layer of grey matter measuring approximately two to four millimetres in thickness, constitutes the outermost covering of the cerebral hemispheres. Despite its modest thickness, it contains roughly 16 billion neurons and an even larger number of glial cells, all arranged in six horizontal layers that vary in relative thickness and cellular composition across different regions. This laminated organisation is a defining feature of the neocortex, the evolutionarily newest part of the brain that is most highly developed in humans and other primates. The cortex is thrown into characteristic folds: the raised ridges are termed gyri, while the intervening grooves are called sulci. The most prominent of these sulci serve as landmarks for dividing each hemisphere into four major lobes: frontal, parietal, temporal, and occipital, to which is sometimes added the insula, a fifth lobe hidden within the lateral sulcus.
Each lobe is associated with distinct functional domains, though it is crucial to recognise that these associations are probabilistic rather than absolute. The frontal lobe, lying anterior to the central sulcus and superior to the lateral sulcus, is primarily concerned with voluntary movement, executive functions, and aspects of personality and social cognition. The parietal lobe, positioned posterior to the central sulcus and superior to the temporal lobe, integrates somatosensory information and contributes to spatial awareness and attention. The temporal lobe, located inferior to the lateral sulcus, houses the primary auditory cortex and is essential for memory formation and semantic knowledge. The occipital lobe, at the posterior pole of the hemisphere, is devoted almost entirely to vision. What follows is a detailed examination of each region and its constituent specialised areas.
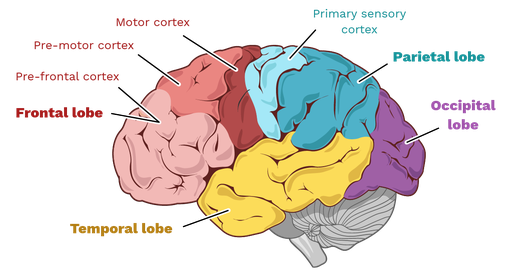
3.2 The Frontal Lobe: From Motor Execution to Executive Control
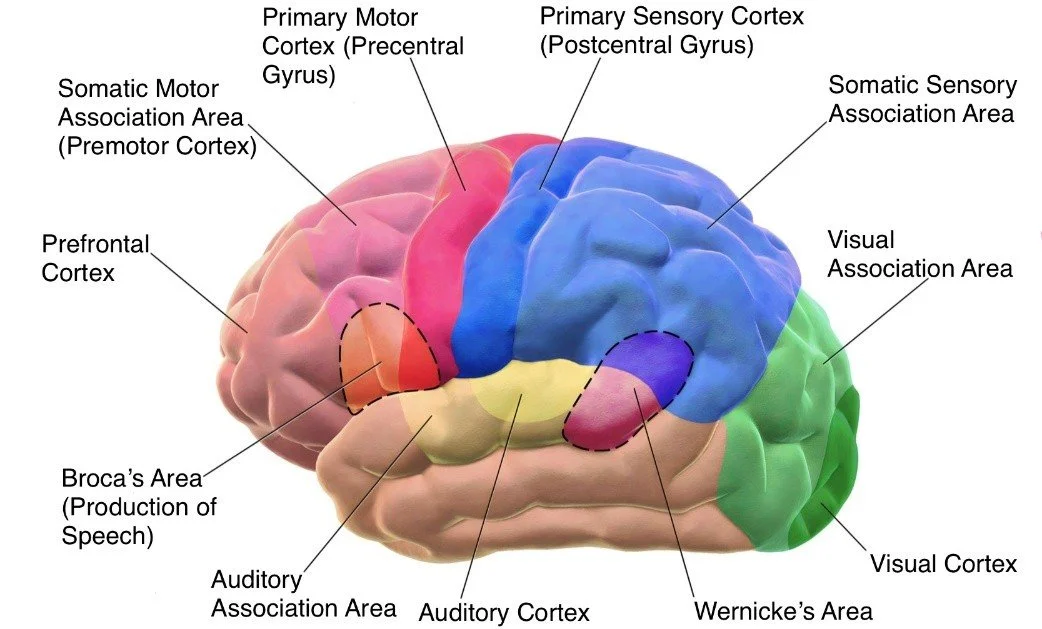
The frontal lobe is arguably the most expanded region in the human brain relative to other primates, and it is this expansion that underpins many of the cognitive abilities that distinguish our species. The lobe can be subdivided into three principal functional zones: the primary motor cortex, the premotor cortex and supplementary motor area, and the prefrontal cortex.
The primary motor cortex, located on the precentral gyrus immediately anterior to the central sulcus, corresponds to Brodmann area 4. This region is the final cortical output station for voluntary movement. Its neurons, large pyramidal cells known as Betz cells, send axons through the corticospinal tract to synapse directly onto lower motor neurons in the spinal cord, providing the most direct route from cortical intention to muscular contraction. The organisation of the primary motor cortex is somatotopic, meaning that different body parts are represented in different parts of the cortex, arranged in a distorted map known as the motor homunculus. The face and hands, particularly the thumb, occupy disproportionately large territories relative to the trunk and legs, reflecting the fine motor control required for speech and manual dexterity. Damage to the primary motor cortex results in contralateral weakness or paralysis, with the severity depending on the extent of the lesion. Crucially, however, even complete destruction of area 4 does not abolish all movement; residual, albeit crude, movements can be generated by subcortical and brainstem motor systems, a phenomenon that underscores the hierarchical organisation of the motor system.
Directly anterior to the primary motor cortex lies the premotor cortex, Brodmann area 6. Unlike the primary motor cortex, which executes movements, the premotor cortex is involved in planning and sequencing movements, particularly those guided by external sensory cues. Neurons in the premotor cortex fire not only during movement execution but also during the observation of movements performed by others, a property that has led to the discovery of mirror neurons, cells that respond both when an individual performs an action and when they witness another individual performing the same action. This discovery, first made in the ventral premotor cortex of macaque monkeys and subsequently confirmed in humans using functional imaging, has profoundly influenced theories of action understanding, imitation, and even empathy. The supplementary motor area, also part of area 6 but located on the medial surface of the hemisphere, participates in the internal generation of movement sequences, particularly those that are bimanually coordinated or that require the retrieval of motor plans from memory.
The prefrontal cortex, occupying the most anterior portion of the frontal lobe, is the seat of what are collectively termed executive functions: working memory, cognitive flexibility, inhibitory control, planning, reasoning, and decision making. Cytoarchitectonically, the prefrontal cortex can be divided into three major subdivisions: the dorsolateral prefrontal cortex, the ventromedial prefrontal cortex, and the orbitofrontal cortex. The dorsolateral prefrontal cortex is critical for working memory, the ability to hold and manipulate information online for brief periods. When you mentally rehearse a phone number while walking to the telephone, you are engaging your dorsolateral prefrontal cortex. This region also subserves cognitive flexibility, the capacity to shift between different mental sets or strategies in response to changing task demands. Damage to the dorsolateral prefrontal cortex produces a syndrome characterised by perseveration, poor planning, and impaired abstract reasoning, while leaving basic cognitive functions such as language and memory relatively intact.
The ventromedial prefrontal cortex and orbitofrontal cortex are more closely associated with emotional regulation, reward processing, and social decision making. The case of Phineas Gage, the railroad construction foreman who in 1848 survived an iron rod passing through his frontal lobes only to undergo a dramatic personality change, has become legendary in neuroscience. Although the precise location of Gage’s lesion has been debated, modern reconstructions suggest that it primarily affected the ventromedial prefrontal cortex. Following his injury, Gage was described as irreverent, profane, impatient, and unable to plan for the future or to act in accordance with social norms, despite retaining normal intelligence, memory, and language function. Subsequent research has confirmed that the ventromedial prefrontal cortex integrates cognitive and emotional information to guide advantageous decision making. Patients with damage to this region often perform poorly on gambling tasks, persistently choosing options that yield immediate rewards despite larger long-term losses, a pattern that reflects a failure to generate anticipatory emotional signals, or somatic markers, that normally guide behaviour away from disadvantageous choices.
3.3 The Parietal Lobe: Sensory Integration and Spatial Awareness
The parietal lobe lies posterior to the central sulcus and extends backward to an imaginary line drawn between the parieto-occipital sulcus and the preoccipital notch. Its anterior portion, the postcentral gyrus, contains the primary somatosensory cortex, Brodmann areas 3, 1, and 2. This region receives inputs from the ventroposterolateral and ventroposteromedial nuclei of the thalamus, which carry tactile and proprioceptive information from the contralateral body surface. Like the primary motor cortex, the primary somatosensory cortex is organised somatotopically, forming a sensory homunculus in which the lips, tongue, and fingers are represented over disproportionately large areas. Damage to the postcentral gyrus produces contralateral deficits in tactile discrimination, including impaired ability to recognise objects by touch, to localise tactile stimuli, and to perceive the shape, size, or texture of objects placed in the hand.
The posterior parietal cortex, comprising Brodmann areas 5 and 7, is not a unitary sensory area but rather an association cortex that integrates information from multiple modalities. This region receives inputs not only from the somatosensory cortex but also from the visual and auditory association cortices, allowing it to construct a unified representation of the body in space. The posterior parietal cortex is essential for the visual guidance of movement, including reaching, grasping, and eye movements. It also contributes to the sense of body ownership and agency, the feeling that one’s limbs belong to oneself and are under one’s voluntary control. Pioneering studies using rubber hand illusions have demonstrated that activity in the posterior parietal cortex can be modulated by manipulating multisensory congruence, and lesions in this region can produce striking disorders of body awareness. In asomatognosia, patients deny ownership of a limb, often attributing it to another person. In anosognosia, patients with hemiplegia deny that they are paralysed, a phenomenon that reveals the brain’s capacity to construct a coherent, albeit inaccurate, narrative of its own deficits.
The parietal lobe also contains specialised regions for numerical cognition and language. The intraparietal sulcus is consistently activated during tasks requiring numerical processing, including symbolic arithmetic, approximate number comparison, and mental number line estimation. This region is thought to represent magnitude in an abstract, modality-independent format, and its degree of activation correlates with individual differences in mathematical ability. Within the dominant hemisphere, typically the left, the parietal lobe includes the angular gyrus and supramarginal gyrus, areas that are critical for reading, writing, and calculation. Damage to the angular gyrus produces Gerstmann syndrome, a characteristic tetrad of finger agnosia, left-right disorientation, agraphia, and acalculia, often accompanied by an alexia without agraphia when the lesion extends to involve the adjacent visual word form area.
Perhaps the most dramatic syndrome associated with parietal lobe damage is hemispatial neglect, a condition in which patients fail to attend to stimuli on the side of space contralateral to the lesion, most commonly following right parietal strokes. A patient with left hemispatial neglect may eat only the food on the right side of their plate, shave only the right side of their face, or when asked to copy a drawing, produce a diagram that omits the left half of each feature. Remarkably, these patients are not blind to the left side of space; they can detect simple visual stimuli if presented in isolation, and the neglect can sometimes be temporarily ameliorated by vestibular or prism adaptation. Hemispatial neglect is not a sensory deficit but an attentional one, reflecting damage to a distributed network that includes the temporoparietal junction, the inferior parietal lobule, and the ventral frontal cortex. Its persistence and severity underscore the critical role of the right parietal lobe in maintaining spatial awareness across both hemispaces.
3.4 The Temporal Lobe: Audition, Memory, and Meaning
The temporal lobe lies below the lateral sulcus and extends posteriorly to merge indistinctly with the occipital and parietal lobes. Its superior temporal plane contains the primary auditory cortex, Brodmann areas 41 and 42, also known as Heschl’s gyrus. This region receives tonotopically organised input from the medial geniculate nucleus of the thalamus and is responsible for basic auditory processing, including sound localisation and frequency discrimination. Bilateral damage to the primary auditory cortex produces cortical deafness, an extremely rare condition in which individuals lose the ability to perceive sound despite intact peripheral hearing. Unilateral damage has minimal impact on hearing because the ascending auditory pathways are largely bilateral, though it may impair sound localisation in the contralateral hemifield.
Surrounding the primary auditory cortex is the auditory association cortex, which performs increasingly complex analyses of sound. This region includes the planum temporale, a structure that is larger in the left hemisphere in the majority of individuals and is critical for phonological processing. The most famous focal point in the temporal lobe is Wernicke’s area, located in the posterior portion of the superior temporal gyrus, traditionally designated Brodmann area 22. Wernicke’s area is essential for language comprehension. Patients with damage to this region produce fluent but meaningless speech, a condition termed Wernicke’s aphasia or receptive aphasia. Their sentences maintain normal prosody and grammatical structure, but they are riddled with paraphasias, substitutions of incorrect words or sounds, and neologisms, invented words that have no meaning. Comprehension of spoken language is severely impaired, and patients are often unaware of their deficits, a lack of insight that distinguishes Wernicke’s aphasia from Broca’s aphasia, in which patients are typically acutely aware of their speech difficulties.
Ventral to the superior temporal gyrus are the middle and inferior temporal gyri, which participate in higher-order visual processing, particularly the recognition of objects and faces. The ventral stream of visual processing, which courses from the occipital lobe into the inferior temporal cortex, is essential for identifying what an object is, as opposed to the dorsal stream, which projects into the parietal lobe and encodes where it is. Within the inferior temporal cortex, a specialised region known as the fusiform face area, located on the fusiform gyrus, is selectively responsive to faces. Damage to this region produces prosopagnosia, or face blindness, a condition in which patients cannot recognise familiar faces, including sometimes their own reflection, despite intact ability to recognise objects and to perceive basic facial features. Conversely, lesions in adjacent regions can produce category-specific agnosias for other stimulus classes, including places, bodies, and words, supporting the view that the ventral temporal cortex is organised into domain-specific modules specialised for processing ecologically important categories.
Perhaps the most celebrated structure within the temporal lobe is the hippocampus, a curved, seahorse-shaped structure buried deep within the medial temporal cortex. The hippocampus is critical for the formation of new declarative memories, that is, memories for facts and events that can be consciously recalled. This conclusion rests on the study of patient Henry Molaison, known in the literature as H.M., who underwent surgical resection of the medial temporal lobes bilaterally in 1953 in an attempt to control intractable epilepsy. The surgery successfully reduced H.M.’s seizures but left him with a devastating and permanent anterograde amnesia: he was unable to form new memories for events occurring after the surgery, though his memory for events prior to surgery remained largely intact, and his procedural memory, the ability to learn new motor skills, was preserved. Subsequent research has refined the understanding of hippocampal function. The hippocampus is not the permanent storage site of memories but rather acts as a binding mechanism, rapidly linking together the distributed cortical representations that constitute an episode and then gradually consolidating these memories into the neocortex for long-term storage. This process of systems consolidation is thought to occur during sleep, particularly during slow-wave sleep, when the hippocampus reactivates patterns of neural activity that occurred during waking experience, a phenomenon that has been observed directly in rodents and inferred in humans through functional imaging.
Adjacent to the hippocampus lies the amygdala, an almond-shaped collection of nuclei that is essential for the processing of emotion, particularly fear. The amygdala receives sensory inputs from all modalities, either directly or via the thalamus, and projects widely to subcortical and cortical structures involved in autonomic, endocrine, and behavioural responses to emotionally salient stimuli. Classical conditioning experiments in animals and humans have demonstrated that the amygdala is necessary for the acquisition and expression of conditioned fear responses. Damage to the amygdala impairs the ability to recognise fearful facial expressions, to experience fear in threatening situations, and to learn from aversive outcomes. In the rare genetic disease Urbach-Wiethe syndrome, which causes bilateral calcification of the amygdala, patients exhibit remarkably diminished fear responses, approaching potentially dangerous animals or situations without the normal hesitation, while other aspects of emotional processing remain relatively intact.
3.5 The Occipital Lobe: The Visual Brain
The occipital lobe, occupying the posterior pole of each hemisphere, is almost exclusively devoted to visual processing. The primary visual cortex, also known as striate cortex or Brodmann area 17, lies within the calcarine sulcus on the medial surface of the occipital lobe. This region receives input from the lateral geniculate nucleus of the thalamus, which in turn receives retinal input via the optic radiations. The organisation of the primary visual cortex is retinotopic, meaning that adjacent points on the retina project to adjacent points in the cortex, preserving the spatial relationships of the visual world. Moreover, a disproportionate amount of cortical territory is devoted to the fovea, the central region of the retina responsible for high-acuity vision, a cortical magnification that reflects the behavioural importance of detailed central vision. Damage to the primary visual cortex produces a scotoma, a region of blindness in the corresponding part of the visual field. When the damage is complete in one hemisphere, the result is homonymous hemianopia, blindness in the contralateral half of the visual field of both eyes. Remarkably, some patients with damage to the primary visual cortex retain the ability to detect visual stimuli presented in their blind field, a phenomenon termed blindsight. These patients cannot consciously see the stimuli but can guess their location, movement, or even orientation at levels significantly above chance, indicating that some visual information reaches higher cortical centres via pathways that bypass the primary visual cortex, such as the retinotectal pathway projecting to the superior colliculus.
Surrounding the primary visual cortex are the secondary visual areas, including V2, V3, V4, and V5, also known as the middle temporal area. These areas perform increasingly complex analyses of visual features. The so-called ventral stream, projecting from V1 through V4 into the inferior temporal cortex, is specialised for object recognition, processing attributes such as colour, form, and texture. The dorsal stream, projecting from V1 through V5 into the parietal cortex, is specialised for spatial vision and the visual guidance of movement, processing motion, depth, and spatial relationships. Damage to area V4 produces cerebral achromatopsia, the loss of colour vision despite intact retinal colour processing, leaving the world experienced in shades of grey. Damage to area V5 produces akinetopsia, or motion blindness, a profoundly disabling condition in which moving objects appear as a series of static, discontinuous images, like viewing the world through a stroboscope. Patients with akinetopsia cannot cross a busy street, pour a cup of coffee, or follow a conversation because they cannot perceive the subtle movements of facial expressions.
3.6 The Insula: The Hidden Lobe
The insula lies buried within the lateral sulcus, hidden from surface view by the overlying frontal, parietal, and temporal opercula. This region has received increasing attention in recent decades as its role in interoception, the perception of the body’s internal state, has become appreciated. The insula receives inputs from visceral sensory pathways, including baroreceptors, chemoreceptors, and thermoreceptors, and it represents these signals in a somatotopic manner, with the anterior insula more involved in emotional awareness and the posterior insula in primary interoceptive sensation. Functional imaging studies have consistently shown insula activation during tasks requiring the detection of one’s own heartbeat, the experience of pain, the perception of temperature, and the awareness of air hunger or thirst. Moreover, the insula is activated when individuals experience emotions such as disgust, anger, fear, and happiness, and its activity correlates with subjective ratings of emotional intensity. These findings have led to the proposal that the insula constructs a moment-to-moment representation of the body’s physiological condition, the interoceptive basis of subjective feeling states.
The anterior insula, in particular, has been implicated in empathy, moral intuition, and social emotions such as guilt and embarrassment. When individuals witness someone else experiencing pain or disgust, their anterior insula activates in a manner that mirrors the activation observed when they experience those states directly, a mechanism that may underlie empathic understanding. Damage to the insula can produce striking deficits in awareness of illness, such as anosognosia for hemiplegia, and can impair the ability to recognise emotions in others, particularly disgust. In rare cases, insular lesions have been reported to lead to the cessation of cigarette craving in addicted individuals, suggesting that the conscious desire to smoke depends on interoceptive signals represented in this region.
3.7 Subcortical Structures: The Basal Ganglia, Thalamus, and Hypothalamus
Beneath the cerebral cortex lie a collection of subcortical nuclei that are essential for movement, learning, and homeostasis. The basal ganglia, a group of interconnected structures including the caudate nucleus, putamen, globus pallidus, subthalamic nucleus, and substantia nigra, are best known for their role in motor control, but they also contribute to cognitive and emotional functions. The basal ganglia receive excitatory input from the entire cerebral cortex and send inhibitory output, via the thalamus, back to the frontal cortex. This circuit is organised into parallel loops that process motor, oculomotor, associative, and limbic information. In the motor loop, the basal ganglia facilitate desired movements by disinhibiting thalamocortical projections while suppressing competing movements. Damage to the basal ganglia produces characteristic movement disorders. In Parkinson’s disease, degeneration of dopaminergic neurons in the substantia nigra pars compacta leads to bradykinesia, rigidity, resting tremor, and postural instability. In Huntington’s disease, an autosomal dominant disorder caused by a trinucleotide repeat expansion in the huntingtin gene, degeneration of the caudate nucleus and putamen produces chorea, involuntary, dance-like movements, along with cognitive decline and psychiatric symptoms. In hemiballismus, typically caused by a stroke affecting the subthalamic nucleus, patients experience wild, flinging movements of the contralateral limbs, reflecting the loss of normal inhibitory control over motor output.
The thalamus, a paired egg-shaped structure located deep within the diencephalon, serves as the brain’s principal relay station. Nearly all sensory information except olfaction is relayed through specific thalamic nuclei before reaching the cerebral cortex. The lateral geniculate nucleus relays vision to the occipital lobe, the medial geniculate nucleus relays audition to the temporal lobe, and the ventroposterior nuclei relay somatosensation to the parietal lobe. In addition to these sensory relays, the thalamus contains association nuclei that relay information between cortical regions and intralaminar nuclei that contribute to arousal and attention. Damage to the thalamus can produce a variety of syndromes depending on the specific nuclei involved. Infarction of the paramedian thalamus can cause a triad of vertical gaze palsy, impaired consciousness, and amnesia, while damage to the ventral posterior nucleus produces contralateral loss of sensation. A curious phenomenon, thalamic pain or central post-stroke pain, can occur after thalamic lesions, in which patients experience spontaneous, intractable pain on the contralateral side of the body, even to light touch or temperature changes that are ordinarily innocuous.
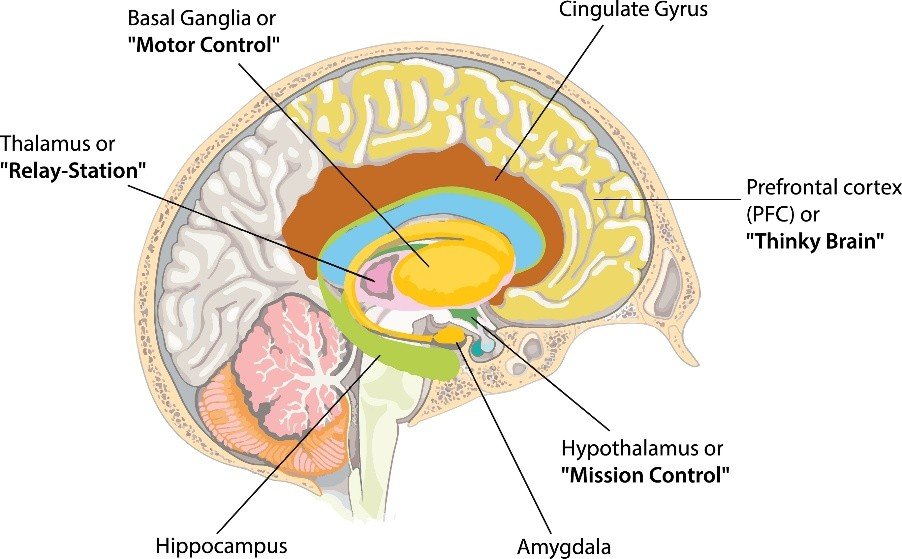
The hypothalamus, located below the thalamus and forming the floor of the third ventricle, is the master regulator of homeostasis. Despite its small size, less than four cubic centimetres in humans, the hypothalamus contains numerous distinct nuclei that control body temperature, thirst, hunger, fatigue, sleep-wake cycles, circadian rhythms, and autonomic and endocrine function. The supraoptic and paraventricular nuclei produce vasopressin and oxytocin, hormones released from the posterior pituitary that regulate water balance and social bonding, respectively. The suprachiasmatic nucleus, the brain’s master circadian pacemaker, receives direct input from the retina via the retinohypothalamic tract and synchronises peripheral clocks throughout the body to the external light-dark cycle. The arcuate nucleus contains two populations of neurons that regulate appetite: one producing the orexigenic peptides neuropeptide Y and agouti-related peptide, which stimulate hunger, and another producing the anorexigenic peptides pro-opiomelanocortin and cocaine- and amphetamine-regulated transcript, which inhibit hunger. These neurons receive inputs from peripheral hormones including leptin, ghrelin, and insulin, allowing the hypothalamus to adjust feeding behaviour according to the body’s energy stores. Lesions of the ventromedial hypothalamus produce hyperphagia and obesity, while lesions of the lateral hypothalamus produce aphagia and weight loss.
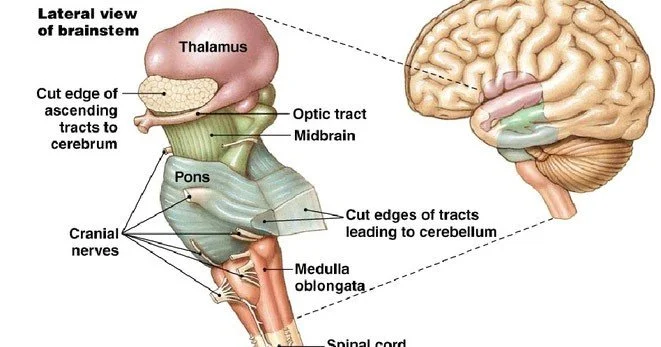
3.8 The Brainstem and Cerebellum
The brainstem, comprising the midbrain, pons, and medulla oblongata, connects the cerebrum and cerebellum to the spinal cord. Despite its relatively small size, the brainstem contains the nuclei of most cranial nerves, the reticular formation, and ascending and descending tracts that convey information between the brain and the body. The midbrain contains the superior colliculi, which coordinate eye movements and visual reflexes, and the inferior colliculi, which process auditory information. The substantia nigra, part of the basal ganglia circuit, is also located in the midbrain. The pons contains pontine nuclei that relay information from the cerebral cortex to the cerebellum and the locus coeruleus, the brain’s main source of noradrenaline, which modulates arousal, attention, and the stress response. The medulla oblongata contains autonomic centres essential for life: the dorsal respiratory group and ventral respiratory group generate the rhythm of breathing, the nucleus tractus solitarius receives visceral sensory information including blood pressure and blood chemistry, and the rostral ventrolateral medulla regulates sympathetic outflow to maintain blood pressure. Damage to the medulla is often rapidly fatal, as witnessed by the common experience of cervical spinal cord injury above the level of C3, which disrupts phrenic nerve output but spares medullary respiratory centres, versus injury to the medulla itself, which abolishes spontaneous breathing entirely.
The cerebellum, meaning little brain, is attached to the dorsal surface of the brainstem and contains more neurons than the entire cerebral cortex, approximately 69 billion of the brain’s 86 billion neurons, though these are organised into a highly regular, crystalline structure that contrasts with the laminar organisation of the cortex. The cerebellum is traditionally associated with motor coordination, particularly the smooth execution of voluntary movements, the maintenance of posture and balance, and the learning of motor skills. Individuals with cerebellar damage exhibit ataxia, a decomposition of movement into its component parts, resulting in jerky, inaccurate, and poorly timed actions. They also exhibit dysmetria, the tendency to overshoot or undershoot a target, and intention tremor, a tremor that intensifies as a goal-directed movement approaches its target. The cerebellum is, however, not exclusively motor. Functional imaging studies have consistently shown cerebellar activation during cognitive tasks involving attention, working memory, language, and mental imagery, and patients with cerebellar damage can exhibit cognitive-affective syndromes characterised by executive dysfunction, visuospatial impairments, and blunted or inappropriate affect. The cerebellum is now understood to act as a universal prediction engine, generating internal models of the sensory consequences of action that allow the brain to distinguish self-generated from externally generated sensations and to anticipate the future state of the body and the environment.
Discussion
The preceding survey of brain regions and their functions reveals a picture of extraordinary complexity, yet certain organising principles emerge. First, functional specialisation is a genuine feature of brain organisation. The primary visual cortex truly is specialised for basic visual processing, the hippocampus for declarative memory formation, and Broca’s area for language production. These specialisations are not absolute, as plasticity experiments have shown that visual cortex can be recruited for touch in congenitally blind individuals, and hippocampal circuits can compensate for some forms of medial temporal lobe damage, but the default, experience-expectant organisation of the human brain reliably allocates specific functions to specific regions. Second, these specialised regions do not operate in isolation but are embedded within large-scale, distributed networks. The default mode network, which includes the medial prefrontal cortex, posterior cingulate cortex, and angular gyrus, is deactivated during externally oriented tasks and activated during self-referential thought, mind-wandering, and autobiographical memory retrieval. The salience network, anchored in the anterior insula and dorsal anterior cingulate cortex, detects behaviourally relevant stimuli and switches between the default mode network and the central executive network. The central executive network, including the dorsolateral prefrontal cortex and posterior parietal cortex, supports goal-directed cognition and working memory. Understanding brain function requires not only knowing what each region does in isolation but also how these regions interact across time.
Third, the brain is characterised by both lateralisation and redundancy. Language functions are lateralised to the left hemisphere in the vast majority of right-handed individuals and in most left-handed individuals, as are aspects of motor planning, praxis, and mathematical calculation. The right hemisphere, in contrast, is dominant for visuospatial attention, emotional prosody, and the recognition of emotional facial expressions. Yet when a stroke damages the left hemisphere language areas, many individuals recover substantial language function through recruitment of homologous right hemisphere regions, particularly in the early recovery period. This capacity for compensation, though limited, reflects the fundamental redundancy built into brain organisation, a legacy of the evolutionary principle that critical functions should not depend on any single component.
The clinical implications of regional neuroanatomy are profound. Localisation of neurological deficits remains a cornerstone of clinical neurology. When a patient presents with acute onset of right-sided weakness and expressive aphasia, the clinician immediately suspects a left frontal lesion, most likely involving Broca’s area and the precentral gyrus, and can order appropriate imaging and treatment without delay. When a patient presents with acute vertigo, diplopia, dysarthria, and ataxia, the clinician suspects a brainstem or cerebellar lesion, and the urgency of diagnosis reflects the potential for life-threatening complications. Even in the era of advanced neuroimaging, clinical localisation remains an indispensable skill, guiding the interpretation of scans and the planning of interventions.
Several limitations should be acknowledged. The functional assignments described in this review are based largely on group-averaged neuroimaging data and lesion studies that may obscure individual variability. The precise boundaries of cytoarchitectonic areas vary across individuals, and functional activation patterns can shift substantially following brain injury or intensive training. Moreover, most functional imaging studies rely on the subtraction method, comparing activation during a task condition to activation during a baseline condition, an approach that assumes pure insertion, the questionable assumption that the task condition engages only the process of interest. Future research combining high-field functional imaging with invasive electrophysiology in surgical patients and with detailed behavioural phenotyping will continue to refine the mapping between brain and function.
Conclusion
The human brain is not a collection of isolated organs but an integrated, dynamic system in which specialised regions collaborate to produce the richness of human experience. From the frontal lobes executive command to the occipital lobe’s visual processing, from the temporal lobe’s memory systems to the parietal lobe’s spatial maps, from the basal ganglia’s movement selection to the cerebellum’s motor learning, each region contributes distinct operations that are essential for adaptive behaviour. The amygdala tells us what matters, the hippocampus records what happened, and the prefrontal cortex decides what to do about it. The insula whispers the condition of the body, the thalamus relays the state of the world, and the brainstem keeps the whole enterprise alive, breath by breath, heartbeat by heartbeat.
Understanding these regions and their functions is not merely an academic exercise. It provides the foundation for diagnosing and treating the devastating consequences of stroke, trauma, tumour, and degeneration. It illuminates the neural basis of psychiatric disorders, from the prefrontal dysfunction of depression to the limbic dysregulation of anxiety. And it addresses the deepest question of all: how does the matter of the brain give rise to the mind? That question remains incompletely answered, but the outline of an answer is now visible, written in the folds and fissures of the most remarkable structure in the known universe.
References
1. Bear, M. F., Connors, B. W., & Paradiso, M. A. (2020). Neuroscience: Exploring the Brain (5th ed.). Wolters Kluwer.
2. Brodmann, K. (1909). Vergleichende Lokalisationslehre der Grosshirnrinde. Barth.
3. Damasio, A. R. (1994). Descartes’ Error: Emotion, Reason, and the Human Brain. Putnam.
4. Kandel, E. R., Koester, J. D., Mack, S. H., & Siegelbaum, S. A. (2021). Principles of Neural Science (6th ed.). McGraw-Hill.
5. Mai, J. K., & Paxinos, G. (2012). The Human Nervous System (3rd ed.). Academic Press.
6. Mesulam, M. M. (2000). Principles of Behavioral and Cognitive Neurology (2nd ed.). Oxford University Press.
7. Penfield, W., & Rasmussen, T. (1950). The Cerebral Cortex of Man: A Clinical Study of Localization of Function. Macmillan.
8. Raichle, M. E. (2015). The brain’s default mode network. Annual Review of Neuroscience, 38, 433-447.
9. Yeo, B. T., Krienen, F. M., Sepulcre, J., Sabuncu, M. R., Lashkari, D., Hollinshead, M., … & Buckner, R. L. (2011). The organization of the human cerebral cortex estimated by intrinsic functional connectivity. Journal of Neurophysiology, 106(3), 1125-1165.
