Different types of Anemia in Children
1. Endesh kyzy Gulsara
2. Masrat Mumtaz
Shahid Hameed
Tabish ur Rehman
Eysha Parvez
Laraib Mubashir
Nabia Suboohi
Saloni Sajwan
Mehak Rahanui
(1. Teacher, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.
2. Students, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.)
Abstract
Anemia represents the most common hematological disorder affecting children worldwide, with India bearing the disproportionate burden of approximately eighty million anemic children under five years of age. While iron deficiency dominates both popular perception and public health programming, the etiological landscape of childhood anemia in India encompasses a diverse array of conditions including nutritional deficiencies, hemoglobinopathies, infectious diseases, bone marrow failure syndromes, and chronic systemic disorders that demand distinct diagnostic and therapeutic approaches. This review examines the major types of anemia affecting Indian children—iron deficiency anemia, megaloblastic anemias, hemolytic anemias including thalassemia syndromes and glucose-6-phosphate dehydrogenase deficiency, aplastic and hypoplastic anemias, and anemia of chronic disease—exploring their specific epidemiology, pathophysiology, clinical presentation, and management within the Indian context. Drawing upon the National Family Health Surveys, the Comprehensive National Nutrition Survey, hospital-based hematology registries, and regional genetic studies, we synthesize current understanding of how the interplay of nutritional deprivation, genetic diversity, infectious disease burden, and health system constraints shapes the anemia experience of Indian childhood. The findings reveal that while iron deficiency remains predominant, the substantial contribution of hemoglobinopathies, particularly beta-thalassemia and hemoglobin E disorders in specific regions, and of vitamin B12 deficiency in vegetarian populations, necessitates diagnostic sophistication beyond simple iron supplementation. We discuss the clinical approach to anemia evaluation in resource-variable settings, emphasizing the importance of peripheral smear examination, reticulocyte count, and targeted investigations based on regional epidemiology and clinical phenotype. This review argues that the effective management of childhood anemia in India demands moving beyond universal iron supplementation toward differentiated diagnosis and etiology-specific treatment, with health system strengthening to enable laboratory capacity, genetic screening, and specialized hematology services that reach beyond metropolitan centers to the rural and marginalized children who bear the greatest burden.
1. Introduction
The child with pallor has been a familiar figure in Indian medical practice for generations, recognized by physicians, traditional healers, and families themselves through the characteristic waxy yellow pallor that replaces healthy complexion, the fatigue that limits play and school participation, the dyspnea on exertion that converts normal activity into distress, and the cognitive slowing that compromises educational attainment. Yet familiarity has bred neither effective control nor diagnostic precision. Anemia, despite being the most frequently encountered hematological abnormality in Indian pediatric practice, remains inadequately characterized in individual patients, with empirical iron supplementation administered to millions without confirmation of deficiency, while alternative etiologies including hemoglobinopathies, megaloblastic deficiencies, and chronic disease are missed or mismanaged. The consequences of this diagnostic imprecision extend beyond individual patient outcomes to population-level inefficiency, with iron supplementation programs consuming substantial resources while anemia prevalence remains stubbornly high, and with preventable complications of undiagnosed hemoglobinopathies including iron overload, splenic sequestration, and ineffective erythropoiesis continuing to exact their toll.
The epidemiology of childhood anemia in India is staggering in scale and complex in composition. The National Family Health Survey-5, conducted in 2019-21, documented anemia prevalence of 67.1% in children aged six to fifty-nine months, representing no improvement from previous surveys and possibly genuine worsening when methodological adjustments are considered. This prevalence, among the highest globally, translates to approximately eighty million anemic children in absolute numbers—a figure that exceeds the entire child population of most nations. The burden is not uniformly distributed, with higher prevalence in rural areas, among scheduled caste and scheduled tribe communities, in states with limited health infrastructure, and in children with concurrent undernutrition and infectious disease. Yet even among affluent urban children, prevalence exceeds forty percent, indicating that anemia is not merely a disease of poverty but a pervasive feature of Indian childhood with multiple, intersecting causes.
The traditional conceptualization of anemia as synonymous with iron deficiency, while reflecting the genuine predominance of this etiology, has obscured the substantial contribution of other conditions that demand different management. The thalassemia syndromes, with carrier frequencies approaching 3-4% nationally and exceeding 10-15% in specific communities including Sindhis, Punjabis, Gujaratis, and Bengalis, affect millions of Indian children with microcytic anemia that is iron-replete and potentially harmed by supplementation. Glucose-6-phosphate dehydrogenase deficiency, with prevalence of 2-10% varying by region and community, causes hemolytic anemia triggered by specific exposures and complicates malaria management. Vitamin B12 deficiency, common in vegetarian populations with limited dairy intake, produces megaloblastic anemia with neurological complications that may be irreversible if untreated. And the anemia of chronic disease, accompanying the high burden of tuberculosis, HIV, and chronic kidney disease in Indian children, requires management of underlying condition rather than nutritional supplementation.
The diagnostic evaluation of anemia in Indian children presents challenges of resource availability, clinical expertise, and epidemiological context that shape appropriate investigation. The peripheral blood smear, a fundamental tool that can distinguish microcytic from macrocytic anemia, identify abnormal morphology suggesting hemoglobinopathy, and detect megaloblastic changes, requires skilled examination that is increasingly scarce as automated counters replace manual microscopy. The reticulocyte count, essential for distinguishing hypoproliferative from hemolytic or blood loss anemia, is unavailable in many primary care settings. Iron studies including serum ferritin, while preferable to empirical supplementation, are limited by cost and by the confounding effects of inflammation in infection-endemic populations. Hemoglobin electrophoresis, necessary for thalassemia and hemoglobinopathy diagnosis, is concentrated in tertiary centers with substantial delays and costs for referred patients. And bone marrow examination, definitive for aplastic anemia, leukemia, and marrow infiltration, requires specialized hematology services that are inaccessible to the majority of Indian children.

This review examines the major types of anemia affecting Indian children, moving beyond the iron deficiency paradigm to encompass the full spectrum of nutritional, genetic, infectious, and systemic causes. We explore the specific Indian epidemiology of each condition, the pathophysiological mechanisms that produce anemia, the clinical presentations that suggest specific etiologies, and the diagnostic and management approaches appropriate for resource-variable settings. Throughout, we maintain attention to the human dimensions of childhood anemia—the mother who recognizes pallor in her child but cannot access investigation, the child whose school performance suffers from unrecognized cognitive effects, the family bankrupted by transfusion-dependent thalassemia—and the health system and policy responses necessary for effective control. Our objective is to provide a comprehensive resource that informs clinical practice, guides diagnostic evaluation, and advocates for the differentiated, etiology-specific approach to anemia that Indian children deserve.
2. Methods
This narrative review was conducted through systematic examination of the peer-reviewed literature, national survey data, and hospital-based studies pertaining to anemia types in Indian children. Our scope encompasses children from birth through adolescence, with particular attention to the under-five age group where anemia prevalence is highest and consequences most severe.
We searched PubMed, Embase, the Cochrane Library, and Indian databases including the Indian Journal of Pediatrics, Indian Journal of Hematology and Blood Transfusion, and Journal of the Indian Academy of Pediatrics using combinations of MeSH terms and keywords including "anemia," "child," "pediatric," "India," "iron deficiency anemia," "megaloblastic anemia," "vitamin B12 deficiency," "folate deficiency," "thalassemia," "beta-thalassemia," "hemoglobin E," "sickle cell disease," "glucose-6-phosphate dehydrogenase deficiency," "G6PD," "hemolytic anemia," "aplastic anemia," "Fanconi anemia," "Diamond-Blackfan anemia," "anemia of chronic disease," "chronic kidney disease," "tuberculosis," "HIV," "malaria," "hookworm," "peripheral smear," "reticulocyte count," "hemoglobin electrophoresis," "National Family Health Survey," "NFHS," "Comprehensive National Nutrition Survey," "CNNS," and "iron supplementation."
Key data sources include the National Family Health Surveys (NFHS-3, 4, and 5) providing nationally representative anemia prevalence estimates; the Comprehensive National Nutrition Survey 2016-18 with detailed biochemical assessment including ferritin, vitamin B12, and folate; the National Rural Health Mission anemia screening data; hospital-based hematology studies from major Indian centers including All India Institute of Medical Sciences, Christian Medical College Vellore, and Tata Memorial Hospital; and regional genetic studies of thalassemia and G6PD deficiency from various states.
The synthesis integrates epidemiological data with pathophysiological understanding, clinical diagnostic approaches, and management strategies appropriate for Indian contexts. We have attempted to balance presentation of biomedical mechanisms with attention to health system realities that constrain diagnosis and treatment, and to the social and economic consequences of anemia for Indian children and families. Where evidence specific to Indian populations is limited, we have indicated the need for context-specific research while drawing upon global evidence adapted to local considerations.
3. Results
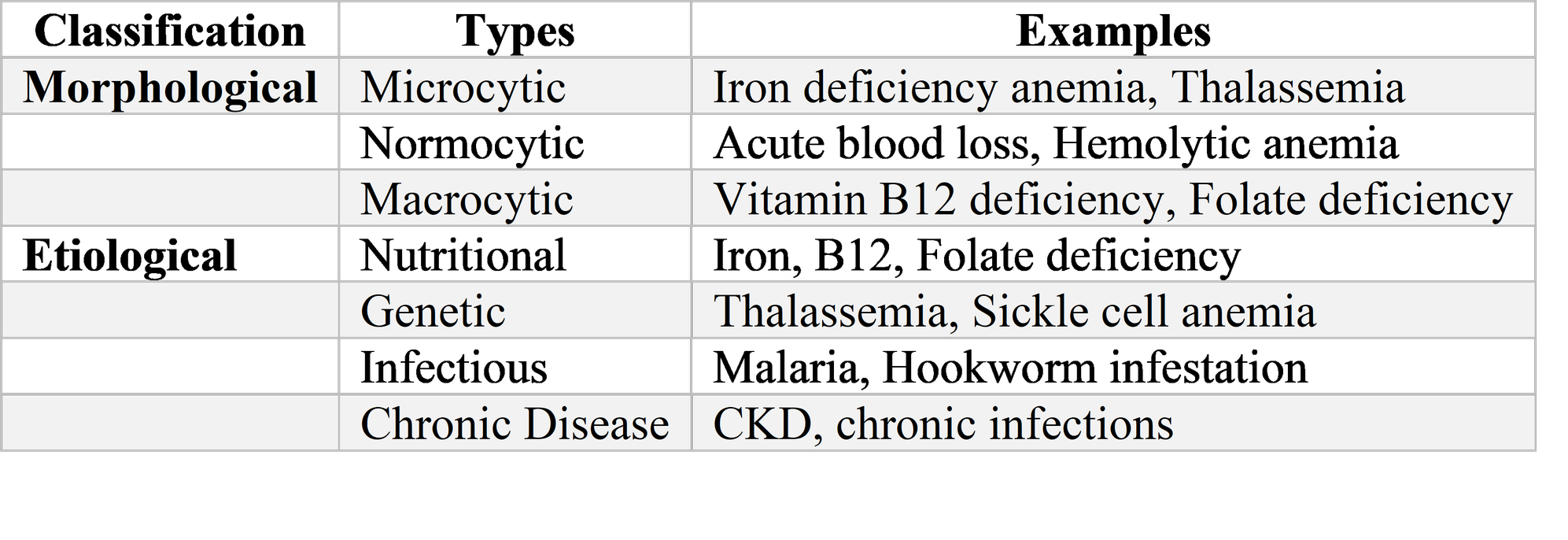
3.1 Iron Deficiency Anemia: The Predominant but Not Exclusive Cause
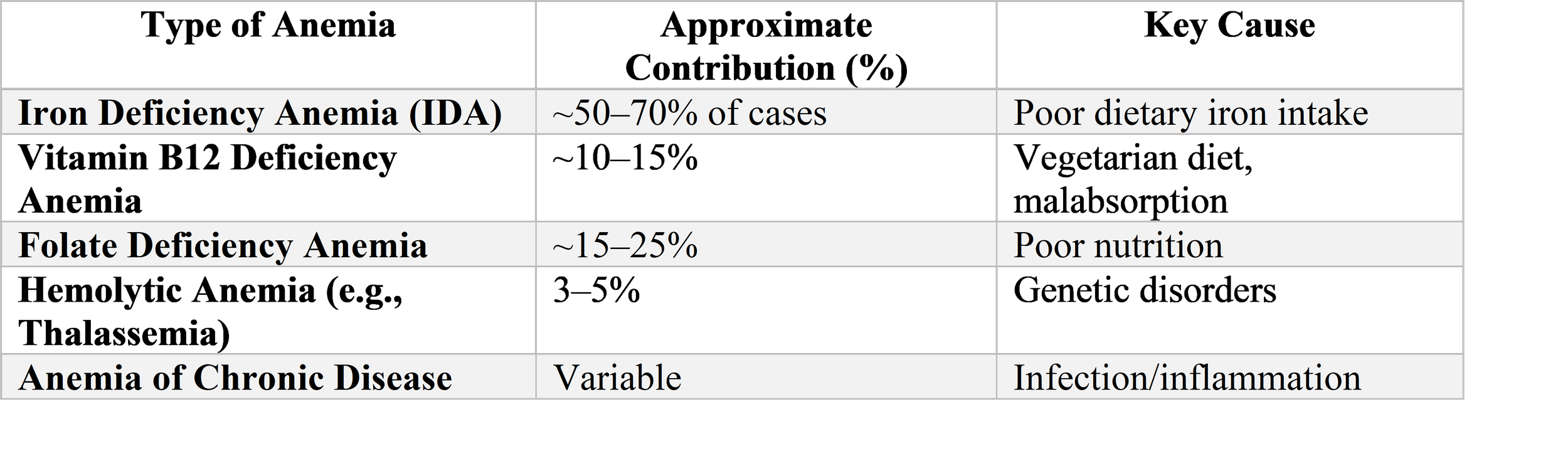
Iron deficiency anemia remains the most common type of anemia affecting Indian children, accounting for approximately fifty to sixty percent of anemia cases in population-based studies and an even higher proportion in community settings where advanced diagnostics are unavailable. The pathogenesis involves inadequate dietary iron intake, poor bioavailability of non-heme iron from cereal-based diets, increased iron requirements during rapid growth periods, and blood loss from hookworm infestation or other sources. The consequences extend beyond hematological parameters to encompass neurodevelopmental impairment, immune dysfunction, and increased infection susceptibility that may persist even after hematological correction.
The epidemiology of iron deficiency anemia in India reflects dietary patterns shaped by agricultural heritage, economic constraint, and cultural practice. The typical infant diet, dominated by breast milk with low iron concentration beyond six months and complementary foods with high phytate and low bioavailable iron, fails to meet requirements during the period of rapid growth and brain development. Early introduction of cow's milk, widespread in India due to cultural beliefs about its nutritive value, displaces iron-rich foods and causes occult intestinal blood loss. The vegetarian diet predominant in most Indian households eliminates heme iron, the most bioavailable form, while the limited consumption of vitamin C-rich fruits and vegetables impairs absorption of non-heme iron. The result is a population-level iron deficit that begins in infancy and persists through childhood, with peak prevalence in the second year of life when growth demands are highest and dietary diversification often inadequate.
The clinical presentation of iron deficiency anemia in Indian children follows the expected progression from iron store depletion through functional iron deficiency to overt anemia, though many children present with advanced deficiency due to delayed recognition. Pallor, most visible in conjunctivae, tongue, and palms, is often first noticed by mothers or grandmothers who may attribute it to normal variation or seek traditional remedies before medical consultation. Fatigue and reduced playfulness, irritability, and poor appetite are nonspecific symptoms that may be misattributed to behavioral issues or other illness. The pica for non-food items including mud, chalk, and ice—particularly characteristic of iron deficiency—is frequently unrecognized or dismissed as habit. Tachycardia and cardiac flow murmurs indicate physiological compensation, with high-output cardiac failure developing in severe, prolonged cases. The cognitive and behavioral effects, including impaired attention, reduced school performance, and developmental delay, are often insidious and unrecognized until educational failure prompts evaluation.
The diagnosis of iron deficiency anemia in Indian settings typically relies upon hemoglobin concentration below age-specific thresholds—eleven grams per deciliter for children six to fifty-nine months and twelve grams per deciliter for older children—supplemented by peripheral smear examination showing microcytic, hypochromic red cells. The mean corpuscular volume and mean corpuscular hemoglobin concentration are reduced, though these indices may be normal in early deficiency or in mixed deficiencies. Serum ferritin below fifteen micrograms per liter confirms depleted stores, but this assay is frequently unavailable and is elevated by inflammation in infection-endemic populations, limiting specificity. The soluble transferrin receptor, elevated in iron deficiency but not affected by inflammation, offers improved diagnostic accuracy but remains a research tool in most Indian settings. Therapeutic trial of iron supplementation with hemoglobin rise of more than one gram per deciliter in one month provides practical confirmation when laboratory testing is limited.
The management of iron deficiency anemia follows established protocols with Indian specificities. Oral ferrous sulfate or ferrous fumarate providing three to six milligrams per kilogram of elemental iron daily or on alternate days is first-line therapy, with alternate-day dosing potentially improving absorption by avoiding hepcidin-mediated block. The duration of three to six months corrects anemia and replenishes stores, with longer courses indicated for ongoing risk. Adherence is limited by gastrointestinal side effects including constipation, abdominal pain, and nausea; by the unpleasant taste of liquid formulations for young children; and by the prolonged duration requiring sustained caregiver commitment. Intravenous iron sucrose or ferric carboxymaltose, increasingly available in urban centers, is indicated for severe anemia, malabsorption, or failed oral therapy, with caution regarding anaphylaxis risk. Blood transfusion is reserved for hemodynamic compromise or severe anemia with hemoglobin below five grams per deciliter, with volume-reduced or packed red cells preferred to avoid circulatory overload.
Prevention of iron deficiency anemia in Indian children encompasses multiple strategies with variable implementation and effectiveness. The National Iron Plus Initiative provides biweekly iron-folate supplementation for children six to fifty-nine months and weekly supplementation for older children, with coverage estimates suggesting only twenty to thirty percent of eligible children receive full courses. Dietary diversification counseling, emphasizing continued breastfeeding with timely introduction of iron-rich complementary foods including pulses, green leafy vegetables, and fruits, is integrated into maternal and child health programs but implementation is inconsistent. Iron fortification of staple foods including wheat flour and double-fortified salt is expanding but faces technical and acceptability challenges. And the control of hookworm through mass deworming programs, while reducing one cause of iron loss, has demonstrated limited impact on population anemia prevalence, indicating the predominance of dietary insufficiency over blood loss etiology.
3.2 Megaloblastic Anemias: Vitamin B12 and Folate Deficiency
Megaloblastic anemia, characterized by impaired DNA synthesis producing large, immature red cell precursors and pancytopenia in severe cases, represents a significant and underrecognized cause of anemia in Indian children, particularly in vegetarian populations and those with limited animal source food consumption. Vitamin B12 deficiency, more common than folate deficiency in the Indian context due to widespread vegetarianism and inadequate dairy intake, produces neurological complications that may be irreversible if untreated, demanding heightened clinical awareness and specific diagnostic attention.
The epidemiology of vitamin B12 deficiency in Indian children reflects dietary patterns and physiological requirements. Vitamin B12 is synthesized exclusively by microorganisms and obtained through consumption of animal products including meat, fish, eggs, and dairy. The predominantly lactovegetarian diet of most Indian families, with limited dairy consumption due to cost and availability constraints, provides inadequate B12 for growing children. Breastfed infants of B12-deficient mothers are born with depleted stores and receive inadequate B12 in breast milk, with deficiency manifesting in early infancy with failure to thrive, developmental delay, and megaloblastic anemia. The adolescent growth spurt, with increased metabolic demands, may precipitate deficiency in children with marginal status. And the increasing prevalence of pernicious anemia and other adult-onset B12 malabsorption in young adults suggests that some Indian children may have genetic or autoimmune predispositions to deficiency that manifest earlier than in other populations.
Folate deficiency, while less common than B12 deficiency in most Indian settings, occurs in populations with limited consumption of green leafy vegetables and legumes, and is exacerbated by the high demand of rapid growth, chronic hemolysis in thalassemia and sickle cell disease, and antifolate medications including anticonvulsants. The neural tube defect prevention mandate for periconceptional folate supplementation has reduced deficiency in women of reproductive age but childhood deficiency persists in specific contexts.
The clinical presentation of megaloblastic anemia in Indian children includes the expected pallor and fatigue of anemia, with specific features that may suggest the diagnosis before laboratory confirmation. Glossitis, with beefy red, sore tongue, is characteristic though not universal. Neurological manifestations of B12 deficiency include developmental delay or regression, irritability, tremor, ataxia, and delayed myelination visible on neuroimaging, with potential for permanent cognitive impairment if deficiency is prolonged. The pancytopenia of severe megaloblastic anemia may present with infection or bleeding rather than isolated anemia symptoms. And the mild jaundice from ineffective erythropoiesis and intramedullary hemolysis may suggest hemolytic rather than megaloblastic etiology.
The diagnosis of megaloblastic anemia relies upon peripheral smear examination showing macrocytosis with mean corpuscular volume typically exceeding one hundred femtoliters, though mixed deficiency with iron deficiency may produce normocytic cells. The smear shows characteristic hypersegmented neutrophils with six or more nuclear lobes, and large, oval red cells. Reticulocytopenia indicates ineffective erythropoiesis. Serum vitamin B12 below two hundred picograms per milliliter confirms deficiency, with methylmalonic acid elevation providing functional confirmation when available. Serum folate and red cell folate assess folate status, with red cell folate less affected by acute dietary changes. The bone marrow, if examined, shows characteristic megaloblastic erythropoiesis with large, immature precursors and nuclear-cytoplasmic asynchrony, though marrow examination is rarely necessary for diagnosis. The management of megaloblastic anemia requires specific vitamin replacement rather than iron, with empirical iron supplementation potentially delaying diagnosis and appropriate treatment. Vitamin B12 deficiency is treated with intramuscular cyanocobalamin one thousand micrograms daily for one week, then weekly for four to eight weeks, then monthly maintenance until dietary correction or indefinitely for malabsorption. Oral high-dose B12 is effective for dietary deficiency but requires adherence monitoring. Folate deficiency is treated with oral folic acid one to five milligrams daily until hematological recovery and dietary correction. The neurological complications of B12 deficiency may improve partially with treatment but are often irreversible if prolonged, emphasizing the importance of early recognition and prevention through maternal supplementation and infant monitoring.
3.3 Hemolytic Anemias: Thalassemia Syndromes and G6PD Deficiency
The hemolytic anemias, characterized by shortened red cell survival and compensatory erythropoietic hyperplasia, represent a major cause of childhood anemia in India with distinctive regional and community patterns determined by genetic epidemiology. The thalassemia syndromes, particularly beta-thalassemia and hemoglobin E disorders, and glucose-6-phosphate dehydrogenase deficiency constitute inherited conditions with high prevalence in specific Indian populations, demanding diagnostic precision, genetic counseling, and specialized management that is often inadequately available.
Beta-thalassemia, caused by mutations in the beta-globin gene producing reduced or absent beta-chain synthesis, has carrier frequency of approximately 3-4% in India with substantial regional and community variation. The highest frequencies are observed in Sindhis, Punjabis, Gujaratis, Bengalis, and certain tribal populations, with carrier rates exceeding 10-15% in some communities. The pathophysiology involves imbalanced globin chain synthesis with alpha-chain excess, leading to ineffective erythropoiesis, hemolysis, and iron overload from transfusion and increased absorption. The clinical spectrum ranges from thalassemia minor (trait) with mild microcytic anemia, through thalassemia intermedia with variable transfusion requirement, to thalassemia major with transfusion dependence from early childhood and fatal without modern management.
Thalassemia major in Indian children typically presents between six months and two years of age as fetal hemoglobin declines and adult hemoglobin synthesis fails. Pallor, failure to thrive, and hepatosplenomegaly from extramedullary hematopoiesis are presenting features, with facial bone changes and pathological fractures developing in untreated cases. The diagnosis is suggested by severe microcytic hypochromic anemia with disproportionately low mean corpuscular volume for hemoglobin level, confirmed by hemoglobin electrophoresis showing elevated hemoglobin F and absent or severely reduced hemoglobin A. Molecular diagnosis for specific mutations is available in specialized centers and essential for prenatal diagnosis and counseling.
The management of thalassemia major has been transformed by modern transfusion and iron chelation, though access remains profoundly inequitable in India. Hypertransfusion regimens maintaining hemoglobin above nine to ten grams per deciliter suppress ineffective erythropoiesis and enable normal growth and development, but require reliable blood supply and transfusion facilities that are unavailable in many areas. Iron chelation with deferoxamine, deferiprone, or deferasirox is essential for survival beyond the second decade, preventing cardiac, hepatic, and endocrine complications of iron overload, but requires sustained adherence and monitoring that challenges health systems and families. Hematopoietic stem cell transplantation, the only curative therapy, is available in limited centers at costs prohibitive for most Indian families. The psychosocial burden of transfusion-dependent thalassemia—chronic illness, treatment demands, financial catastrophe, and family disruption—is substantial and inadequately addressed.
Thalassemia minor, the carrier state, produces mild microcytic anemia that is frequently misdiagnosed as iron deficiency, with inappropriate and potentially harmful iron supplementation administered. The recognition of thalassemia minor is essential for genetic counseling, with carrier screening and prenatal diagnosis available for at-risk couples to prevent birth of affected children. The expansion of screening programs, including extended family screening of identified carriers and community screening in high-prevalence areas, offers potential for prevention but requires health system investment and community engagement.
Hemoglobin E disorders, common in eastern and northeastern India including West Bengal, Assam, and tribal populations, result from a beta-globin mutation producing structurally abnormal hemoglobin with thalassemic properties. Hemoglobin E trait produces mild microcytic anemia, while hemoglobin E-beta-thalassemia compound heterozygosity produces variable severity ranging from thalassemia intermedia to major phenotype. The interaction of hemoglobin E with alpha-thalassemia and with iron deficiency further complicates the clinical spectrum, demanding diagnostic sophistication that is often unavailable.
Glucose-6-phosphate dehydrogenase deficiency, the most common enzymatic disorder affecting approximately four hundred million people worldwide, has prevalence of 2-10% in Indian populations with substantial regional and community variation. The G6PD enzyme protects red cells from oxidative stress through maintenance of reduced glutathione; deficiency produces hemolysis on exposure to oxidative triggers including fava beans (broad beans), certain drugs including primaquine and dapsone, infections, and naphthalene. The clinical presentation includes neonatal jaundice with risk of kernicterus, acute hemolytic anemia with exposure to triggers, and chronic hemolysis in severe variants. The diagnosis is suggested by hemolysis with bite cells and blister cells on smear, confirmed by quantitative G6PD assay or fluorescent spot test, with testing contraindicated during acute hemolysis when young cells with higher enzyme activity may produce false normal results. Management emphasizes trigger avoidance, with blood transfusion for severe hemolysis and phototherapy or exchange transfusion for neonatal hyperbilirubinemia. The public health importance of G6PD deficiency in India includes contraindication to primaquine for malaria radical cure, with implications for malaria elimination strategies.
3.4 Aplastic and Hypoplastic Anemias: Bone Marrow Failure
Aplastic anemia, characterized by pancytopenia and bone marrow hypocellularity, and the related hypoplastic anemias including Fanconi anemia and Diamond-Blackfan anemia, represent rare but life-threatening causes of anemia in Indian children that demand specialized diagnostic and therapeutic capabilities often concentrated in metropolitan centers.
Acquired aplastic anemia, with immune-mediated destruction of hematopoietic stem cells, presents with pallor, bleeding, and infection from pancytopenia, with severity classified by degree of cytopenias. The epidemiology in India includes idiopathic cases, drug and toxin exposure including chloramphenicol and traditional medicines, viral associations including hepatitis and parvovirus, and increasing recognition of atypical presentations of inherited bone marrow failure. The diagnosis requires bone marrow examination showing marked hypocellularity with fat replacement, with exclusion of hypoplastic myelodysplastic syndrome and acute leukemia by cytogenetics and flow cytometry. The management of severe aplastic anemia in India is constrained by limited access to matched sibling stem cell transplantation, the treatment of choice for young patients, and by cost and toxicity of immunosuppressive therapy with horse antithymocyte globulin and cyclosporine. Supportive care including transfusion, infection prophylaxis, and iron chelation is often all that is available, with outcomes poor for severe disease.
Fanconi anemia, the most common inherited bone marrow failure syndrome, has prevalence in India likely underestimated due to limited diagnostic capacity. The autosomal recessive or X-linked disorder produces progressive bone marrow failure, congenital anomalies including radial ray defects and short stature, and extreme cancer predisposition including acute myeloid leukemia and squamous cell carcinoma. The diagnosis is suggested by congenital anomalies and confirmed by chromosomal breakage study with diepoxybutane or mitomycin C, with molecular diagnosis for specific complementation groups available in research settings. The management includes androgen support for bone marrow function, hematopoietic stem cell transplantation with reduced intensity conditioning to avoid Fanconi-specific toxicity, and intensive cancer surveillance, with comprehensive care available only in specialized centers.
Diamond-Blackfan anemia, the congenital pure red cell aplasia, presents in infancy with isolated anemia and macrocytosis, elevated hemoglobin F, and characteristic physical anomalies including thumb defects and short stature. The ribosomal protein gene mutations underlying most cases have implications for cancer risk and treatment response. Corticosteroid therapy produces remission in many cases, with transfusion dependence for non-responders and stem cell transplantation for transfusion toxicity or transformation.
3.5 Anemia of Chronic Disease and Systemic Disorders
The anemia of chronic disease, now termed anemia of inflammation or anemia of chronic kidney disease depending on context, represents a common and underrecognized cause of anemia in Indian children with high burdens of infectious and chronic disease. The pathophysiology involves inflammatory cytokine-mediated upregulation of hepcidin, blocking iron mobilization from stores and absorption from diet despite adequate or increased total body iron, combined with reduced erythropoietin response and direct marrow suppression. The diagnosis is suggested by anemia with normal or elevated ferritin, low transferrin saturation, and elevated inflammatory markers, with exclusion of iron deficiency by soluble transferrin receptor or therapeutic trial.
Infectious causes of anemia in Indian children include malaria, with hemolysis and marrow suppression; tuberculosis, with chronic inflammation and nutritional impairment; HIV, with direct marrow effects and opportunistic infection; and chronic diarrhea and intestinal parasitism with malabsorption and blood loss. The management requires treatment of underlying infection rather than nutritional supplementation alone, with anemia often persisting despite apparent infection control due to ongoing inflammation and nutritional deficit.
Chronic kidney disease, with reduced erythropoietin production, produces normocytic normochromic anemia that is often severe and resistant to iron supplementation. Recombinant human erythropoietin is effective but expensive and rarely available for children in India. Chronic liver disease, with reduced protein synthesis and altered hormone metabolism, produces variable anemia often with hemolysis. And malignancy, with marrow infiltration, nutritional competition, and treatment toxicity, produces multifactorial anemia demanding specific diagnosis and management.
3.6 Diagnostic Approach in Indian Settings
The diagnostic evaluation of anemia in Indian children requires systematic approach adapted to available resources, clinical context, and regional epidemiology. The initial assessment includes detailed history—dietary intake, growth trajectory, bleeding symptoms, infection exposure, family history of anemia or hemoglobinopathy, and medication use—combined with careful examination for pallor, jaundice, lymphadenopathy, hepatosplenomegaly, and specific anomalies suggesting inherited syndromes.
The complete blood count with red cell indices provides essential orientation, with microcytosis suggesting iron deficiency or thalassemia, macrocytosis suggesting megaloblastic deficiency or liver disease, and normocytosis suggesting anemia of chronic disease, aplastic anemia, or early mixed deficiency. The peripheral smear examination, despite declining availability, remains invaluable for identifying morphological clues including hypochromia, target cells in thalassemia, spherocytes in hemolysis, fragmented cells in microangiopathy, and hypersegmented neutrophils in megaloblastosis. The reticulocyte count distinguishes hypoproliferative from hyperproliferative or blood loss anemia, with elevated count indicating hemolysis or response to treatment and low count indicating marrow failure or nutritional deficiency.
Targeted investigations based on initial assessment include iron studies for microcytic anemia, with ferritin and soluble transferrin receptor if available; hemoglobin electrophoresis for suspected thalassemia or hemoglobinopathy; vitamin B12 and folate levels for macrocytic anemia; G6PD assay for hemolysis with suggestive history or smear; and bone marrow examination for pancytopenia, suspected leukemia, or unexplained anemia. The integration of clinical presentation, basic hematology, and targeted testing enables etiological diagnosis in most cases, with referral to specialized hematology services for complex or refractory cases.
4. Discussion
The evidence synthesized in this review reveals childhood anemia in India as a condition of extraordinary complexity and heterogeneity, with iron deficiency representing only the most common among multiple etiologies that demand differentiated diagnosis and management. The persistence of high anemia prevalence despite decades of iron supplementation programs reflects not merely implementation failure but diagnostic imprecision, with substantial proportions of anemia attributable to hemoglobinopathies, megaloblastic deficiencies, and chronic disease that are unresponsive to or potentially harmed by iron.
The thalassemia syndromes, with carrier frequencies approaching population screening thresholds in specific communities, represent a particular challenge for Indian healthcare. The misdiagnosis of thalassemia minor as iron deficiency, with unnecessary and potentially harmful iron supplementation, reflects limited availability of hemoglobin electrophoresis and insufficient clinical awareness. The management of thalassemia major, transformed by modern transfusion and chelation in affluent settings, remains inaccessible to the majority of affected Indian children due to blood supply limitations, chelation costs, and geographic concentration of specialized care. The prevention of thalassemia births through carrier screening and prenatal diagnosis offers population-level solution but requires health system investment and community engagement that has been inadequate.
Vitamin B12 deficiency, common in vegetarian populations with limited dairy intake, produces neurological complications that may be irreversible, demanding heightened diagnostic attention in a country where vegetarianism is culturally predominant and often economically necessitated. The recognition that macrocytic anemia in an Indian child is more likely B12 than folate deficiency, contrary to patterns in other populations, should guide empirical management when specific testing is unavailable.
The diagnostic infrastructure for childhood anemia in India—peripheral smear examination, reticulocyte counting, iron studies, hemoglobin electrophoresis, and specialized hematology services—is inadequately available, particularly in rural and public sector settings where the majority of anemic children seek care. The reliance on hemoglobin measurement alone, without morphological or etiological assessment, perpetuates the iron deficiency paradigm and misses alternative diagnoses. The expansion of laboratory capacity, training in morphological diagnosis, and point-of-care testing for specific etiologies are essential health system investments.
The management of anemia must move beyond universal iron supplementation toward etiology-specific treatment, with diagnostic categorization guiding therapy. This approach demands greater initial investment in assessment but promises improved outcomes and reduced waste from inappropriate treatment. The integration of anemia diagnosis and management with broader child health services, including nutritional counseling, infection control, and chronic disease management, offers potential for comprehensive care that addresses root causes rather than merely treating biochemical deficiency.
5. Conclusion
Childhood anemia in India represents not a single disease but a spectrum of conditions with diverse etiologies, demanding diagnostic sophistication and differentiated management that current health systems often fail to provide. The predominance of iron deficiency has obscured the substantial contribution of thalassemia, megaloblastic deficiency, hemolytic anemia, and bone marrow failure that require specific interventions and may be harmed by empirical iron supplementation.
The path forward demands diagnostic strengthening to enable etiological categorization, workforce training in morphological assessment and specialized hematology, and health system reorientation from universal supplementation toward targeted treatment based on specific diagnosis. The expansion of thalassemia screening and prevention, B12 supplementation for vegetarian populations, and management of hemolytic and aplastic anemias in specialized centers are essential components of comprehensive anemia control.
For the child with pallor in rural Bihar, the diagnostic possibilities extend beyond iron deficiency to encompass conditions that demand specific recognition and treatment. For the family of a child with thalassemia major, the promise of modern management remains unfulfilled due to geographic and economic barriers. For the vegetarian child with B12 deficiency and developmental delay, the neurological consequences may be permanent without early recognition. The effective management of childhood anemia in India requires moving beyond simple paradigms toward comprehensive, etiology-specific care that reaches all children regardless of geography or circumstance.
References
International Institute for Population Sciences (IIPS) and ICF. National Family Health Survey (NFHS-5), 2019-21: India. Mumbai: IIPS; 2022.
Ministry of Health and Family Welfare, Government of India. Comprehensive National Nutrition Survey (CNNS) 2016-2018. New Delhi: MoHFW; 2019.
Stevens GA, Finucane MM, De-Regil LM, et al. Global, regional, and national trends in haemoglobin concentration and prevalence of total and severe anaemia in children and pregnant and non-pregnant women for 1995–2011: a systematic analysis of population-representative data. Lancet Glob Health. 2013;1(1):e16-e25.
Kassebaum NJ, Jasrasaria R, Naghavi M, et al. A systematic analysis of global anemia burden from 1990 to 2010. Blood. 2014;123(5):615-624.
World Health Organization. Worldwide Prevalence of Anaemia 1993-2005: WHO Global Database on Anaemia. Geneva: WHO Press; 2008.
National Health Mission, Ministry of Health and Family Welfare. National Iron Plus Initiative: Guidelines for Control of Iron Deficiency Anaemia. New Delhi: MoHFW; 2013.
Gera T, Sachdev HS, Nestel P. Effect of iron supplementation on haemoglobin response in children: systematic review of randomised controlled trials. J Pediatr Gastroenterol Nutr. 2009;48(1):7-14.
Sachdev H, Gera T, Nestel P. Effect of iron supplementation on mental and motor development in children: systematic review of randomised controlled trials. Public Health Nutr. 2005;8(2):117-132.
Thankachan P, Muthayya S, Walczyk T, et al. Iron absorption in young Indian women: the interaction of iron status with the influence of tea and ascorbic acid. Am J Clin Nutr. 2008;87(4):881-886.
Yajnik CS, Deshpande SS, Jackson AA, et al. Vitamin B12 and folate concentrations during pregnancy and insulin resistance in the offspring: the Pune Maternal Nutrition Study. Diabetologia. 2008;51(1):29-38.
Refsum H, Yajnik CS, Gadkari M, et al. Hyperhomocysteinemia and elevated methylmalonic acid indicate a high prevalence of cobalamin deficiency in Asian Indians. Am J Clin Nutr. 2001;74(2):233-241.
Colah RB, Mukherjee MB, Martin S, Ghosh K. Sickle cell disease in tribal populations in India. Indian J Med Res. 2015;141(5):509-515.
Colah R, Gorakshakar A, Nadkarni A. Global burden, distribution and prevention of β-thalassemias and hemoglobin E disorders. Expert Rev Hematol. 2010;3(1):103-117.
Verma IC, Saxena R, Kohli S. Thalassemia screening in India: present status and the way forward. Indian J Med Res. 2019;149(5):599-601.
Colah R, Italia K, Gorakshakar A. Burden of thalassemia in India: the road map for control and management ordeal. Hemoglobin. 2017;41(4-6):233-238.
Beutler E. G6PD deficiency. Blood. 1994;84(11):3613-3636.
Nkhoma ET, Poole C, Vannappagari V, et al. The global prevalence of glucose-6-phosphate dehydrogenase deficiency: a systematic review and meta-analysis. Blood Cells Mol Dis. 2009;42(3):267-278.
Young NS, Calado RT, Scheinberg P. Current concepts in the pathophysiology and treatment of aplastic anemia. Blood. 2006;108(8):2509-2519.
Alter BP. Diagnosis, genetics, and management of inherited bone marrow failure syndromes. Hematology Am Soc Hematol Educ Program. 2007;2007:29-39.
Weiss G, Goodnough LT. Anemia of chronic disease. N Engl J Med. 2005;352(10):1011-1023.
World Health Organization. Haemoglobin Concentrations for the Diagnosis of Anaemia and Assessment of Severity. Geneva: WHO Press; 2011.
