Juvenile Idiopathic Arthritis Pediatric
1. Osmonova Gulnaz Zhenishbaevna
2. Ruchitkumar Shrivas
3. Sagupata Naz
(1. Teacher, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.
2. Student, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.
3. Student, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.)
Abstract
Background/Objectives: Juvenile Idiopathic Arthritis (JIA) is the most common autoimmune rheumatic disease in children and can vary in presentation based on the properties of the JIA subtypes. Timely diagnosis and intervention are essential for maximizing quality of life, healthy growth and development, and prevention of long-term disability. This review aims to provide a clinically practical framework for the core elements important in recognition, monitoring, and management of JIA.
Methods: We performed a narrative review of the current literature, complemented by real-world clinical experience from academic rheumatology practice. The review synthesizes evidence-based knowledge with practical insights to develop an approach that can be applied in daily clinical decision-making.
Results: We propose a structured, stepwise method for evaluating suspected JIA, emphasizing the integration of pattern recognition with differential diagnosis. Our framework emphasizes two principal parameters: (1) the distribution of joint involvement (peripheral vs. axial) and (2) the presence of extra-articular manifestations, including uveitis, cutaneous findings, and gastrointestinal symptoms. This format aids in distinguishing major JIA subtypes and highlights their distinctive features. In addition, we review overarching principles for monitoring, assessing risk for uveitis, and treatment, and the importance of multidisciplinary care.
Conclusions: This structured approach is intended to support clinicians in the accurate recognition of JIA and its subtypes, facilitate early diagnosis, and provide insights on management strategies that improve patient outcomes.
Keywords: pediatric rheumatology; immunology; JIA
1. Introduction
Pediatric rheumatology is a derivative of Immunology that focuses on excessive inflammation caused by functional aberrations of leukocytes leading to an autoimmune, autoinflammatory, and immune-dysregulatory state of health. The diseases managed in this field include those affecting the musculoskeletal system, such as arthritis or myositis, as well as conditions affecting multiple organs (multisystem or systemic diseases), such as lupus, vasculitis, and scleroderma. Rheumatology is often engaged in multispecialty care in collaboration with organ-specific subspecialties. This allows the optimization of anti-inflammatory treatment of shared patients, including those with psoriasis, inflammatory bowel disease (IBD), or uveitis.
Juvenile Idiopathic Arthritis (JIA) is the most common pediatric autoimmune disease, accounting for approximately 40% of patients assessed by pediatric rheumatology [1,2]. JIA affects about 1 in 1000 children under the age of 16 years [3,4]. Although the incidence and prevalence of JIA is comparable to that of other rare pediatric diseases, early diagnosis and appropriate treatment of JIA remain challenging due, in part, to limited a pediatric rheumatology workforce globally [5]
This manuscript aims to convey the fundamental principles of JIA to primary care providers through a structured and systematic framework.
3. Arthritis, Chronic Inflammation of Synovium
In general, arthritis refers to inflammation of the synovium, a lace-like, thin membrane located in the inner lining of the fibrous sheath that encapsulates diarthrotic joints, tendons, and bursae. According to the 1997 Durban classification criteria, JIA consists of seven distinct subtypes based on clinical properties [6], i.e., systemic-onset JIA (sJIA), oligoarticular JIA (also known as pauciarticular JIA), rheumatoid factor-negative (RF−) polyarticular JIA, rheumatoid factor-positive (RF+) polyarticular JIA, enthesitis-related arthritis (ERA), psoriatic arthritis, and undifferentiated arthritis. Although it is beyond the scope of this manuscript, the readers are encouraged to review the historical aspects of JIA classification, which has continued to evolve toward a global consensus among the pediatric rheumatology community since the 1970s. It is expected that the upcoming new classification will center on immunopathogenic and genetic properties of pediatric arthritis [7].
Regardless of the taxonomy, there is a common theme among different subtypes of JIA regarding the emergence of histopathological findings. The synovium is a unique tissue composed of two layers: the intimal and subintimal layers. The intimal layer is one to two cells thick and consists of macrophage-like, phagocytic type A cells and fibroblast-like, secretory type B cells. The subintimal layer is vascularized and contains many high endothelial venules [8]. Under normal conditions, joints are immuno-privileged sites; however, a disruption in tissue homeostasis can trigger inflammatory cascades.
In arthritis, the activation of endothelial cells leads to the recruitment of circulating blood leukocytes, resulting in chronic inflammation, hyperplasia, and thickening of the synovium. This process typically develops gradually over weeks, or even months, accompanied by the accumulation of a serous inflammatory milieu within the joint cavity. The inflammatory synovial fluid contains primarily neutrophils (<50,000/mm3), proteolytic enzymes, and cytokines. As a result, patients experience stiffness and dull, persistent pain in the affected joint(s), which is most noticeable in the mornings (i.e., morning stiffness). Additionally, tenderness upon palpation and pain with motion of the arthritic joint or tendon insertion sites may be present.
To minimize discomfort, children often avoid joint movement, which can lead to tendon tightness, joint contracture, and muscle atrophy. If untreated, patients with JIA may develop irreversible joint damage with bone and cartilage erosions, which can eventually lead to complete joint fusion over the years. While mortality associated with JIA is low, if the child does not receive adequate treatment, the morbidity and reduced quality of life due to long-term disability can be devastating [9,10].
4. JIA Is a Diagnosis of Exclusion—The Diamond Approach
There is no single test for JIA, and the conclusions must stem from diagnosis of exclusion. Rheumatologists are trained on pattern recognition using history and physical examination along with skillful laboratory investigations and, in some cases, imaging studies.
As shown in Figure 2, the provider may apply the “diamond rule” to rule out infectious, malignant, or orthopedic conditions before considering arthritis when a child presents with joint complaints. The infectious disease (ID) work-up is most crucial to rule out septic arthritis when there is precipitous onset of severe joint pain, particularly in a febrile child with increased C-reactive protein (CRP). This is a medical emergency requiring care coordination with orthopedics and antibiotics. The ID work-up often includes serologies for streptococcus, Epstein–Barr virus (EBV), Lyme disease, and, sometimes, parvovirus for more gradual onset of arthropathy, over days or weeks. Orthopedic concerns include, but are not limited to, trauma and soft tissue injuries that often present with an acute-onset pain that may correlate with findings on imaging studies. In addition, hypermobility and alignment concerns (pes planus, pronated feet, etc.) commonly presents with intermittent transient joint complaints upon overuse that resolve in hours to days with rest. The possibility of malignancy manifesting with joint, back, or limb pain is most concerning and needs particular attention to signs such as nighttime awakenings or subtle abnormalities in the complete blood count (CBC). In these cases, early communication with hematology/oncology specialists is essential. It is necessary to continue close follow-up and monitor patients’ laboratory results, including CBC, lactate dehydrogenase (LDH), and uric acid levels. Patients may need abdominal ultrasound or a bone scan and, if in doubt, bone marrow examination [11].
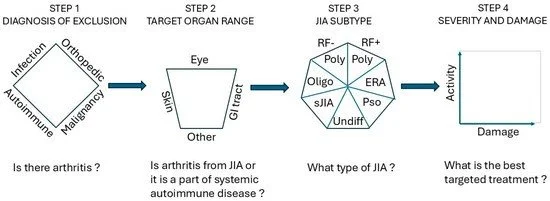
Figure 2. Clinical assessment of and conclusions on JIA require a stepwise approach built upon history and physical examination in a time continuum in conjunction with basic laboratory results and, where possible, images. The purpose of each step is given on the top line, and the decisions that must be made to move the next step are shown on the bottom line. ERA, Enthesitis-Related Arthritis; Pso, psoriatic arthritis.
Role of Primary Care Provider for a Child with JIA
Juvenile Idiopathic Arthritis (JIA) is a chronic condition that requires a multispecialty medical team to achieve the best long-term outcomes. The primary care provider serves as part of the collaborating team among specialists, including rheumatology, ophthalmology, physical and occupational therapy, and counseling services.
Monitoring and Laboratory Work: Regular monitoring of growth and development as well as vigilance for medication-related adverse effects is crucial. Laboratory evaluations are often needed every 2 to 3 months and typically include CBC, CMP, ESR, CRP, and urinalysis.
Specialized tests, such as tests for blood levels of therapeutics and screening for autoantibodies against therapeutics, may be required under the guidance of a rheumatologist.
Preventive measures against infection and early intervention: Children receiving immunomodulators/DMARDs for JIA are immunosuppressed and subject to susceptibility and/or escalated severity of infection. Awareness and, if there is any doubt, prompt and proper intervention with any signs and symptoms of infection are of the utmost importance in these children’s care. Families should seek urgent care for any fever and inform the medical team of their child’s progress. During an infection, it is often recommended to reduce or hold the dose of immunomodulators to allow recovery. In addition, some infections, such as Streptococcal pharyngitis, mycoplasma, shigella, salmonella, and EBV, can trigger a JIA disease flare, and post-infection follow-up may be needed.
Family and Psychosocial Support: Providing support to families is essential to address concerns related to coping, mood, and peer relationships, which significantly impact quality of life. Special attention should be given to minimizing exposure to illness and ensuring prompt evaluation and treatment of infections, especially in the presence of fever.
Immunizations: Routine immunizations and annual flu vaccine are generally encouraged among children with JIA. It is best to administer vaccines when the child is clinically stable and the underlying disease is well-controlled. Live-virus vaccines should be avoided while the patient is receiving DMARDs. The timing of vaccines should sometimes be adjusted relative to immunosuppressive dosing; as an example, for a child on a biologic with infusions/injections every 2–4 weeks, the vaccine may be given midway between doses to maximize the immune response. For patient-centered care, coordinating decisions with the treating rheumatologist will be optimal.
Communication between school and family on child’s wellbeing and community effort towards education and advocacy: Children with JIA are often bright and high achievers in school. By law, they are entitled to an Individualized Education Plan (IEP), which provides accommodations such as modified curricula and additional resources if needed. IEP should also include communication with the family to warn of any infectious outbreaks in the classroom and, when indicated, allow school absences to avoid exposure. Advocacy support by primary care teams provides extra support for patient and parent education and community engagement, which are essential for holistic healing of not only the child, but also the family. Online resources are available that can assist primary care providers to gain further insights.
Workforce and future directions: Pediatric rheumatology, a subspecialty of the American Board of Pediatrics, requires three years of fellowship following general pediatrics training. In the United States, there are approximately 600 board-certified pediatric rheumatologists and 40 accredited fellowship programs; the global workforce is estimated to be <10,000 [5]. Despite workforce limitations, the field is advancing rapidly, with the adoption of next-generation therapeutics and expansion of multicenter translational research. Clinical outcomes have improved substantially over recent decades, accomplishing near elimination of wheelchair dependence among children with severe JIA [42]. Future protocols are expected to emphasize sustained remission and the pursuit of curative therapies.
It is important to note, however, that many bDMARD and tsDMARD agents currently available on the market lack U.S. Food and Drug Administration (FDA) approval for pediatric indications. Industry efforts to expand pediatric labeling remain limited, constrained by financial considerations and a small market size. This gap represents a central challenge in the field and compels pediatric rheumatologists, in partnership with families, to seek strategies that accelerate regulatory approval pathways and ensure timely access to effective therapies [43].
Disclaimer The views expressed in this article are those of the authors and do not necessarily reflect the official policy of the Department of Defense or the U.S. Government. The identification of specific products or scientific instrumentation is considered integral to this scientific endeavor and does not constitute endorsement or implied endorsement on the part of the authors, DOD, or any component agency.
Summary
1. Juvenile Idiopathic Arthritis (JIA) is the most common pediatric rheumatologic disease, driven by chronic synovial inflammation and capable of causing long-term disability.
2. Diagnosis is clinical and by exclusion; no single test confirms JIA. Laboratory markers such as ANA, rheumatoid factor, and HLA-B27 are not diagnostic but offer prognostic value for uveitis risk, arthritis severity, and sacroiliac involvement, respectively.
3. JIA comprises multiple subtypes defined by joint count and distribution, systemic features, and extra-articular organ involvement.
4. Systemic-onset JIA is distinguished by quotidian fever, rash, arthritis, and CBC abnormalities, and carries a risk of macrophage activation syndrome (MAS), a potentially life-threatening complication.
5. Uveitis is the most frequent extra-articular manifestation and a major cause of acquired blindness in Western countries. Often asymptomatic, it necessitates ophthalmologic screening every 3–12 months based on risk factors (age <7 years, ANA positivity).
6. Management requires rheumatology oversight. Treatment is often individualized and ranges from NSAIDs to immunomodulators; intra-articular steroid injection can benefit oligoarticular JIA, while systemic steroids are reserved for selected systemic-onset JIA and MAS. Regular clinical assessment and outcome tracking are essential.
7. Primary care providers are critical for early recognition, infection and drug toxicity monitoring, multidisciplinary coordination, school liaison, and holistic, family-centered support to optimize growth and functional outcomes.
8. These children are often immunocompromised while receiving treatment, even if their white blood cell count is normal. Therefore, live virus vaccines should be avoided. It is also of utmost importance to promptly recognize and appropriately manage any signs of fever or infection throughout the course of therapy.
References
. Ravelli, A.; Martini, A. Juvenile idiopathic arthritis. Lancet2007, 369, 767–778. [Google Scholar] [CrossRef] [PubMed]
. Sandborg, C.I.; Schulert, G.S.; Kimura, Y. Juvenile Idiopathic Arthritis. N. Engl. J. Med.2025, 393, 162–174. [Google Scholar] [CrossRef] [PubMed]
. Saurenmann, R.; Rose, J.; Tyrrell, P.; Feldman, B.; Laxer, R.; Schneider, R.; Silverman, E.D. Epidemiology of juvenile idiopathic arthritis in a multiethnic cohort: Ethnicity as a risk factor. Arthritis Rheum.2007, 56, 1974–1984. [Google Scholar] [CrossRef]
. Manners, P.J.; Bower, C. Worldwide prevalence of juvenile arthritis why does it vary so much? J. Rheumatol.2002, 29, 1520–1530. [Google Scholar] [PubMed]
. Correll, C.K.; Ditmyer, M.M.; Mehta, J.; Imundo, L.F.; Klein-Gitelman, M.S.; Monrad, S.U.; Battafarano, D.F. 2015 American College of Rheumatology Workforce Study and Demand Projections of Pediatric Rheumatology Workforce, 2015–2030. Arthritis Care Res.2022, 74, 340–348. [Google Scholar] [CrossRef] [PubMed]
. Petty, R.E.; Southwood, T.R.; Baum, J.; Bhettay, E.; Glass, D.N.; Manners, P.; Maldonado-Cocco, J.; Suarez-Almazor, M.; Orozco-Alcala, J.; Prieur, A.M. Revision of the proposed classification criteria for juvenile idiopathic arthritis: Durban, 1997. J. Rheumatol.1998, 25, 1991–1994. [Google Scholar] [PubMed]
. Nigrovic, P.A.; Colbert, R.A.; Holers, V.M.; Ozen, S.; Ruperto, N.; Thompson, S.D.; Wedderburn, L.R.; Yeung, R.S.M.; Martini, A. Biological classification of childhood arthritis: Roadmap to a molecular nomenclature. Nat. Rev. Rheumatol.2021, 17, 257–269, Erratum in Nat. Rev. Rheumatol.2021, 17, 306. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
. Kung, M.S.; Markantonis, J.; Nelson, S.D.; Campbell, P. The Synovial Lining and Synovial Fluid Properties after Joint Arthroplasty. Lubricants2015, 3, 394–412. [Google Scholar] [CrossRef]
. Kyllönen, M.S.; Kautiainen, H.; Puolakka, K.; Vähäsalo, P. The mortality rate and causes of death amongjuvenile idiopathic arthritis patients in Finland. Clin. Exp. Rheumatol.2019, 37, 508–511. [Google Scholar]
