Juvenile Rheumatoid Arthritis in Children: A Comprehensive Review of Epidemiology, Pathogenesis, and Modern Management Strategies
1. Endysh Kyzy Gulsara
2. Chaitnya Gondkar
Vaibhav Kale
Suraj Chavan
Vinod Choudhari
Divya Sonje
(1. Teacher, International Medical Faculty, Osh State University, Kyrgyzstan)
(2. Students, International Medical Faculty, Osh State University, Kyrgyzstan)
Abstract
Juvenile Rheumatoid Arthritis (JRA) now exists as Juvenile Idiopathic Arthritis (JIA) which stands as the most prevalent chronic rheumatic disease that affects children across the globe. The review explores the complete understanding of JRA epidemiology which includes its causes, symptoms, methods of diagnosis, and treatment methods based on scientific evidence. Recent studies on disease occurrence show that about 1 per 1,000 children worldwide have the condition although different regions and demographic groups show varying results. The disease pathogenesis develops through the intricate relationship between genetic predisposition which includes HLA alleles and non-HLA variants such as PTPN22 and STAT4 and environmental factors that lead to immune system dysfunction. Researchers have discovered new biological pathways that establish connections between T-cell receptor signaling and neutrophil extracellular traps and joint-specific microenvironments through their research on multi-omic analysis methods. The clinical system divides into seven different subtypes which follow International League of Associations for Rheumatology criteria, where each subtype has its own unique assessment of future disease progression. The development of targeted biologic disease-modifying antirheumatic drugs which include TNF inhibitors and IL-1 and IL-6 blockers has brought about significant changes to treatment methods. Current clinical guidelines recommend that doctors should implement early in- tensive treatment through target-oriented methods which will help patients reach remission while safeguarding their joints from permanent damage. The review offers a detailed summary of recent genetic research and clinical studies and population-based studies which serve as valuable resources for both medical professionals and researchers.
Keywords: juvenile idiopathic arthritis, pediatric rheumatology, autoimmunity, biologics, disease- modifying antirheumatic drugs
INTRODUCTION
The medical community has recognized Juvenile Rheumatoid Arthritis (JRA) as a distinct condition that now requires the use of Juvenile Idiopathic Arthritis (JIA) to describe the diverse types of arthritis that start in childhood because these conditions differ from both adult arthritis and each other. The term “idiopathic” acknowledges the incomplete understanding of etiological mechanisms, while the classification encompasses onset before 16 years of age with persistent arthritis of at least six weeks’ duration after exclusion of other known causes.
The importance of JIA extends its reach beyond joint inflammation. JIA stands as the primary pediatric rheumatic disease which leads to both acquired disabilities and vision loss in children. The condition requires complete treatment through multiple medical disciplines because its chronic nature and potential to cause growth problems and its additional symptoms and its psychological effects on children and their families. The last two decades have brought about major progress in understanding how JIA develops and how to treat it. The development of biologic therapies has transformed disease progression which allows many children to achieve remission who would have experienced ongoing joint destruction. Large-scale genetic studies have tracked both common and unique genetic risk factors which exist between JIA subtypes and adult-onset arthritis, thus changing our understanding of these conditions through their immunological properties instead of their age-based characteristics.
This review aims to synthesize current knowledge regarding JIA epidemiology, pathogenesis, clinical characteristics, diagnostic modalities, and treatment strategies, with particular emphasis on recent evidence from genetic studies, clinical trials, and outcomes research. The article presents a complete resource about understanding and treating this complicated pediatric condition through its combination of traditional clinical methods and modern immunogenetic research.
METHODS
The narrative review developed its findings by examining peer-reviewed studies which were located through systematic searches conducted in the PubMed and Scopus and Web of Science databases. Researchers used various search terms which contained different combinations of “juvenile idiopathic arthritis” and “juvenile rheumatoid arthritis” and “epidemiology” and “pathogenesis” and “genetics” and “treatment” and “biologics.” The research team focused on publications from 2020 to 2026 while they included important historical studies to provide con- text and foundational information about their work.
The study added epidemiological information through Global Burden of Disease Study 2021 estimates which assessed rheumatoid arthritis and JIA among people aged 0 to 19 years. The clinical classification system uses International League of Associations for Rheumatology (ILAR) criteria for its classification method. The treatment guidelines utilize recent Paediatric Society of the African League Against Rheumatism (PAFLAR) guidelines for oligoarticular disease together with research findings about biologic therapy.
RESULTS
Epidemiology and Global Burden
Prevalence and Incidence
Juvenile Idiopathic Arthritis affects approximately 1 in 1,000 children globally, positioning it as an uncommon but clinically significant pediatric condition. The study found that different regions and diagnostic criteria and research methods used three different approaches to measure the disease prevalence which resulted in a range of 1 per 1,000 to 1 per 10,000 children. The data in Table 1 compares the prevalence of various childhood conditions to each other.
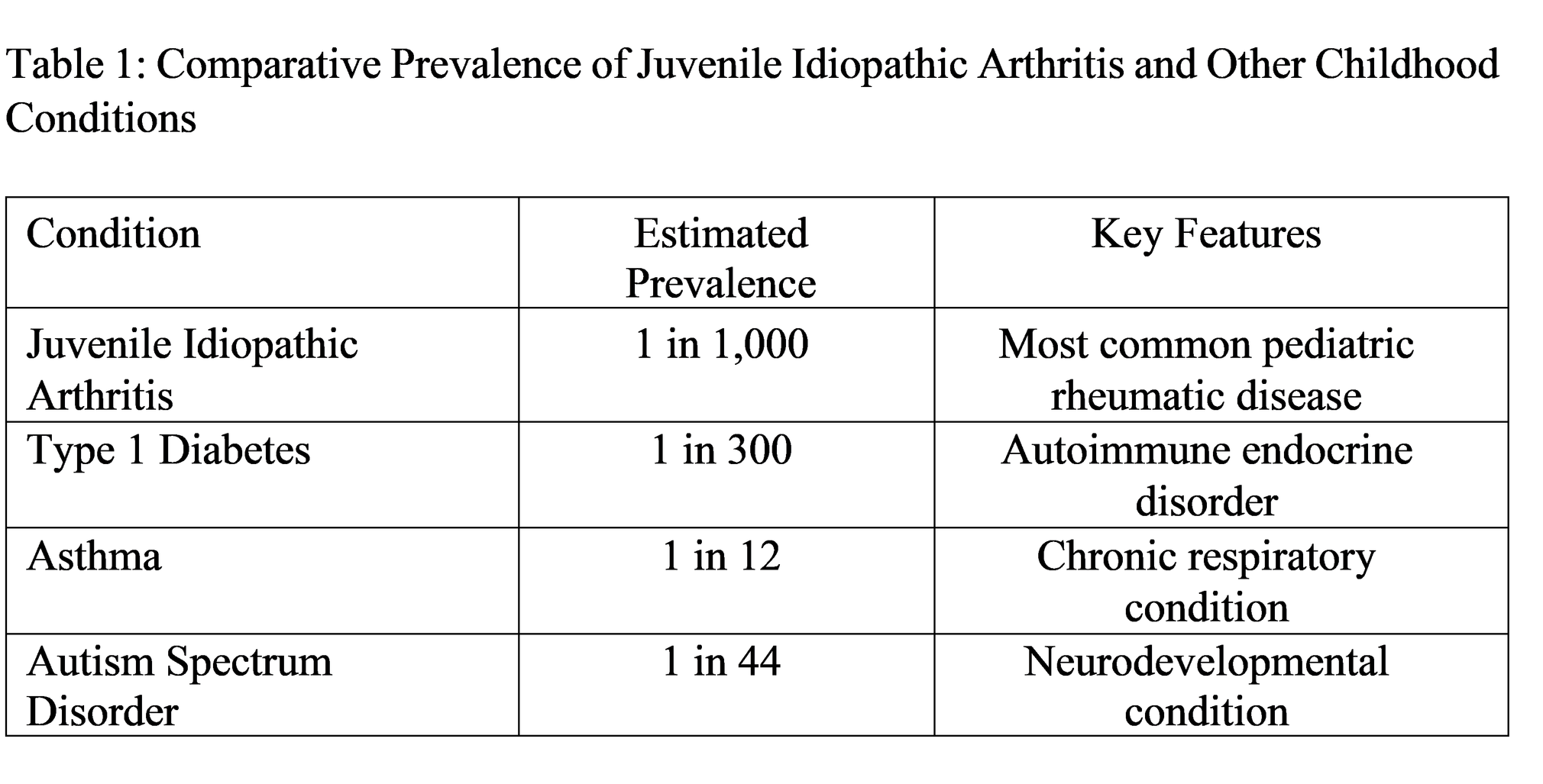
Data synthesized from epidemiological studies
Scandinavian countries exhibit higher rates of a condition compared to Asian populations because geographic differences show distinct prevalence patterns. The observed differences in susceptibility between populations arise from their genetic makeup and the way they interact with environmental factors according to the research study although population-based diagnostic methods show different results.
Temporal Trends and Demographic Patterns
The Global Burden of Disease 2021 data analysis shows that between 1990 and 2021 the world- wide age-standardized rates for arthritis disability and its prevalence among people aged 0-19 increased while their disability-adjusted life years experienced minor reductions. The total burden shows that females experience higher rates in areas with advanced sociodemographic status.
Subtype Distribution
The International League of Associations for Rheumatology recognizes seven JIA subtypes:
1. Oligoarticular JIA: The most prevalent type of the disorder which affects less than four joints during its initial six months of existence occurs most frequently in young children and shows higher incidence rates in girls who also face an elevated risk of developing eye problems.
2. Polyarticular JIA (RF-negative): The condition affects five or more joints while showing RF-negative results which make up about 15 to 20 of all cases.
3. Polyarticular JIA (RF-positive): The condition affects five or more joints while showing RF-positive results which show similarities to adult rheumatoid arthritis but have a worse outcome.
4. Systemic JIA: The disease presents as arthritis which produces both daily fever and skin rash while it represents approximately 10 of all cases through its unique disease mechanisms that include autoinflammatory symptoms.
5. Enthesitis-related arthritis: The disorder causes entheses inflammation which affects mainly boys who show HLA-B27 genetic connection and who usually develop the condition during their teenage years.
6. Psoriatic arthritis: The condition causes joints to become inflamed because of psoriasis together with nail pitting and dactylitis symptoms.
7. Undifferentiated arthritis: The condition does not meet the requirements for any single category while it fulfills the criteria of multiple categories.
Etiology and Pathogenesis
Genetic Susceptibility
The genetic architecture of JIA involves multiple susceptibility loci with variable effect sizes. Family and twin studies have long supported genetic contributions, but recent genome-wide association studies and multi-omic analyses have refined understanding of specific risk variants. The strongest genetic associations for the condition exist within the HLA region according to Human Leukocyte Antigen HLA associations. Specific alleles including HLA-DR8, DR5, and DR6 confer increased susceptibility, while HLA-DR4 shows stronger association with RF-positive polyarticular disease resembling adult RA.
The Table 3 polymorphisms were found to have strong links with adult RA-associated variants according to a foundational study that examined childhood-onset RA.
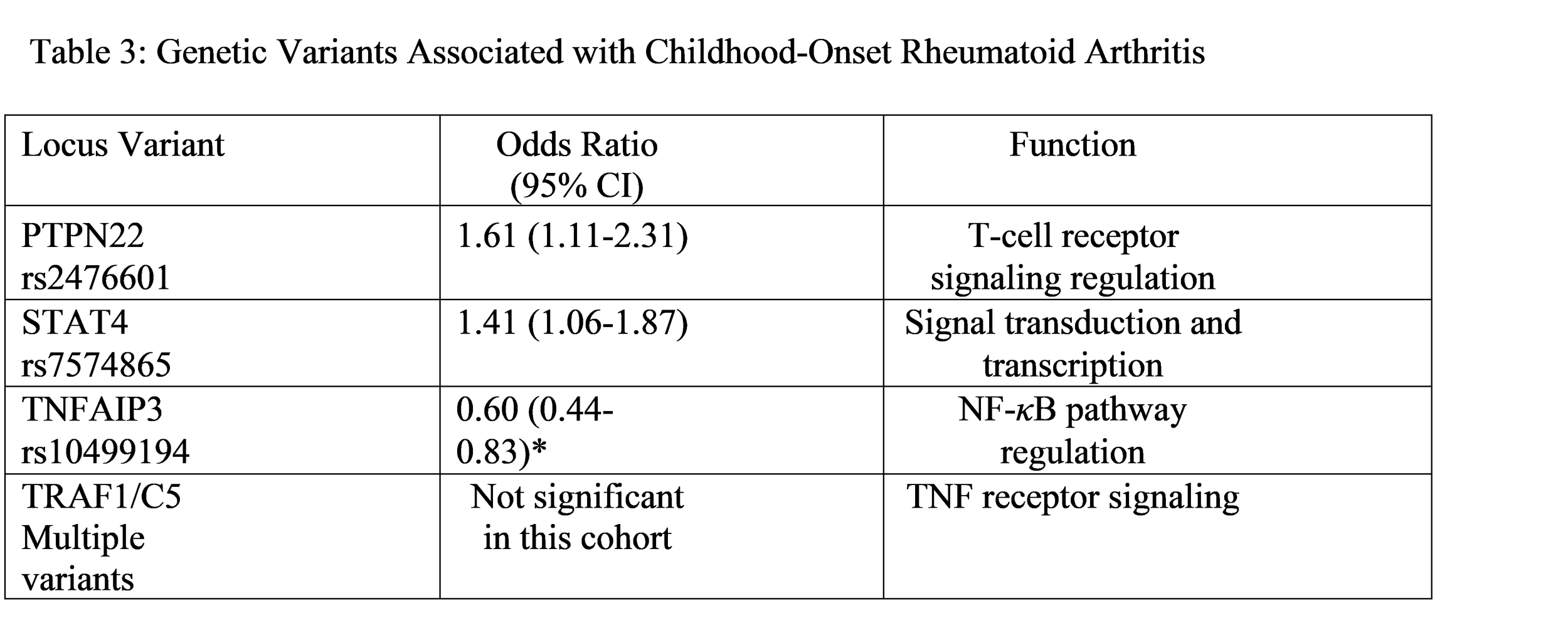
The combined effects of these variants which scientists measured through weighted genetic risk scores showed that people from higher quintiles face increased disease risk which affected especially male individuals. The polygenic inheritance patterns received support through this additive model which shows how multiple genetic factors combine to create specific traits.
In silico multi-omic analyses have discovered new pathways through their recent developments which use combined bulk-RNA and single-cell RNA sequencing datasets for research. The research identified enhanced genes and signaling pathways which control T-cell receptor signaling.
Immunopathogenesis
The autoimmune pathogenesis of JIA involves complex dysregulation of both innate and adaptive immunity. The table shows essential immunological findings which researchers discovered.
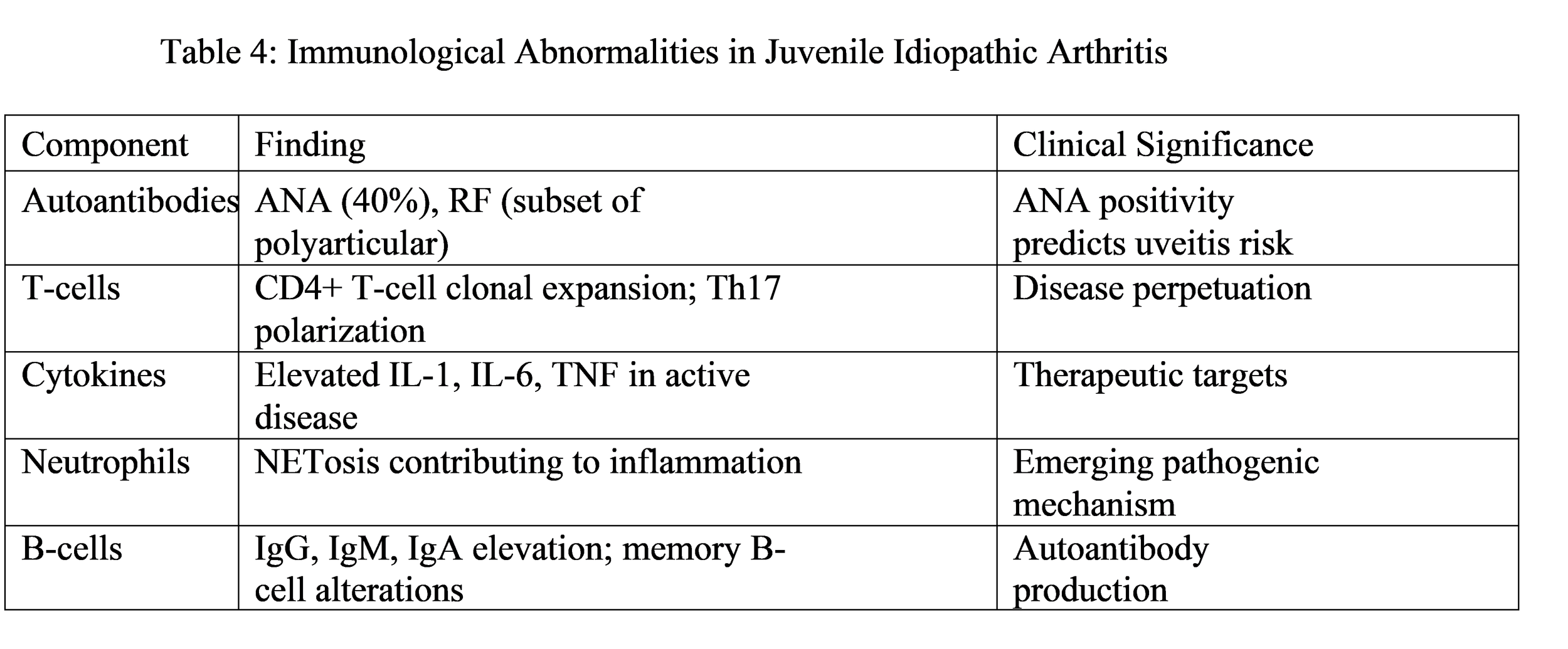
The recent discovery shows that neutrophil extracellular traps (NETs) play a crucial role in advancing disease progression. The web-like structures which consist of DNA and antimicrobial proteins are released through NETosis and they cause ongoing inflammation while they present self-antigens that disrupt immune tolerance.
The joint microenvironment stands as a vital conflict zone where immune cell interactions decide whether diseases will continue or stop. Single-cell analyses have revealed distinct populations of synovial fibroblasts, macrophages, and lymphocytes that maintain inflammatory circuits. The identification of new therapeutic targets can be achieved through studying how local interactions operate.
Environmental Triggers
The research effort has not achieved success in identifying any infectious agent as the definite cause of JIA. The researchers have proposed parvovirus B19, rubella virus, Epstein-Barr virus, and several streptococci and Yersinia and Salmonella bacteria as potential pathogens but their status remains unverified. The current model indicates that environmental factors which include potential infectious agents and other substances trigger abnormal immune reactions in children with genetic vulnerabilities which then continue to operate independently.
Clinical Manifestations
Articular Features
The primary symptom of JIA consists of arthritis that continues for more than six weeks. The condition typically shows joint swelling along with warmth and tenderness while the patient exhibits restricted ability to move their joints. The patient experiences morning stiffness which lasts beyond 30 minutes yet shows improvement through physical activity. The document presents a table which shows joint involvement patterns that different subtypes display.

Extra-Articular Manifestations
The most prevalent and dangerous extra-articular condition which affects children with JIA occurs through chronic anterior uveitis which represents the main form of uveitis. The initial stage of the condition remains without symptoms which medical professionals refer to as “silent uveitis” thus they require patients to undergo periodic eye examinations. The disease develops through four risk factors which include the oligoarticular subtype and the presence of ANA and the early onset of the disease in females. The absence of treatment for uveitis leads to disease progression which results in synechiae and cataracts and glaucoma and permanent vision impairment.
Systemic JIA shows its unique characteristics through daily fevers which rise to 39◦C or higher for two times throughout the day and then return to normal temperature and through its transient salmon-pink skin rash and its lymph node enlargement and its liver and spleen swelling and its serous membrane inflammation. This extreme physical condition presents symptoms which display both autoinflammatory disorders and innate immune system malfunctioning.
Growth Abnormalities: Chronic inflammation directly impacts growth plate development and interrupts growth hormone pathways, leading to reduced linear growth. Patients with localized growth disturbances experience two distinct conditions which include leg length inequality and micrognathia from temporomandibular joint arthritis. The use of corticosteroid treatment results in additional growth suppression effects.
Temporomandibular Joint and Cervical Spine Involvement
The temporomandibular joint (TMJ) is affected in a significant proportion of children with JIA, often bilaterally, and may lead to micrognathia, malocclusion, and difficulty with mastication. Cervical spine involvement, particularly in enthesitis-related and polyarticular subtypes, can cause neck pain and stiffness, and in severe cases atlantoaxial subluxation.
Diagnostic Approach
Clinical Diagnosis
The clinical diagnosis of JIA requires that patients develop arthritis which persists for six weeks in a minimum of one joint after alternative diagnoses have been excluded. The diagnosis depends on laboratory tests together with imaging studies which help to identify the condition and determine disease progression while eliminating other possible diagnoses.
Laboratory Evaluation

Imaging
Imaging plays an increasingly important role in diagnosis and disease monitoring and detection of subclinical inflammation. Conventional Radiography: Radiographs show soft tissue swelling and periarticular osteoporosis and joint space narrowing which develops as a late finding and erosions. The first stage of inflammatory processes shows no signs on radiography.
Ultrasound: Musculoskeletal ultrasound with power Doppler detects synovial hypertrophy and effusion and hyperemia while providing no radiation exposure and dynamic assessment and affordable pricing.
Magnetic Resonance Imaging: MRI provides detailed evaluation of joint structures and cartilage and bone marrow. The technique proves especially valuable for evaluating TMJ and sacroiliac joints and cervical spine because clinical examination shows low sensitivity. Contrast- enhanced MRI identifies synovitis and may detect early erosions before radiographic visibility.
Standardization efforts developed MRI scoring systems for JIA which allow objective measurement of disease activity and response to treatment.
Treatment Strategies
Therapeutic Principles
The modern treatment of JIA follows five basic principles which guide its practice. The first principle requires doctors to start treatment immediately with strong methods because they need to stop inflammation from reaching a point that will cause permanent joint destruction. The second principle requires doctors to use a treatment plan which aims to achieve complete clinical remission of their patient’s condition. The third principle requires doctors to work together with rheumatologists and ophthalmologists and physical therapists and occupational therapists and psychosocial specialists to provide patient care. The fourth principle requires doctors to work together with families to make treatment decisions. The fifth principle requires medical professionals to monitor three aspects of their patient’s health: disease activity, treatment toxicity, and quality of life.
Pharmacologic Treatment
Nonsteroidal Anti-Inflammatory Drugs (NSAIDs): The use of NSAIDs helps to relieve pain and stiffness but they do not change the progression of the disease. The treatment remains effective for patients with mild symptoms and for patients who require additional support. The most common medications for this condition include naproxen and ibuprofen and indomethacin which treats both systemic and enthesitis-related subtypes.
Disease-Modifying Antirheumatic Drugs (DMARDs):
The conventional synthetic DMARD methotrexate serves as the primary treatment for JIA across all disease subtypes. The medication requires a 6-12-week period to reach its full effect after administration of 10-15 mg/m2 via oral or subcutaneous routes on a weekly basis. The body experiences three main adverse effects which include nausea and elevated transaminases and stomatitis but these effects decrease when patients take folic acid supplements.
Leflunomide presents a viable option for patients who cannot tolerate methotrexate. Patients with enthesitis-related and polyarticular JIA can benefit from sulfasalazine treatment although some patients may experience uveitis as a side effect.
Biologic DMARDs: The introduction of biologic treatments has completely transformed the treatment results for JIA. The table 7 presents a summary of the biologic agents that are currently in use. The literature review about biologic therapy for non-systemic JIA showed that early treatment with biologic DMARDs leads to drug-free remission and reduced disease activity and improved patient outcomes. The three drugs adalimumab etanercept and tocilizumab showed similar effectiveness for treating polyarticular JIA while maintaining good tolerability.
The safety assessment shows that IL-1 and IL-6 inhibitors increase infection risk more than TNF inhibitors do. JIA patients experience serious infections and malignancies at higher rates than the general population does but TNF inhibitors show no additional risk when compared to methotrexate treatment.
Corticosteroids provide fast symptom relief but they do not protect against joint damage and their long-term use leads to serious side effects which include growth suppression and osteoporosis and cataracts and weight gain. The indications for this treatment are:
• Systemic JIA with severe features (often as pulse intravenous methylprednisolone)
• Bridge therapy while awaiting DMARD effect
• Severe flares
• Intra-articular injection for oligoarticular disease
Oligoarticular JIA: Special Considerations
The recent PAFLAR guidelines present evidence-based recommendations which guide doctors in their treatment of oligoarticular JIA through the urgent need to diagnose patients and the necessity of team-based medical treatment. The essential recommendations outline the following key points:
• The doctors should use intra-articular corticosteroid injections as their initial treatment option when patients show symptoms of active arthritis.
• Doctors should prescribe methotrexate to children who do not respond to their current treatment while experiencing multiple medical emergencies.
• Patients who belong to high-risk groups should receive ophthalmologic screening tests which take place at three to four-month intervals.
• Doctors should use TNF inhibitors to treat patients who experience severe arthritis symptoms or uveitis which does not respond to other treatments.
• The clinical target treatment program requires operators to achieve total clinical remission.
The guidelines achieved 85-95% consensus among international experts, reflecting broad agreement on management principles.
Non-Pharmacologic Management
The definition of physical therapy shows that it helps maintain joint movement while building strength in muscles around joints and it stops patients from developing contractures. The main focus of occupational therapy is to help people with their daily activities which include school work and methods to protect their joints.
The development of chronic illness during childhood creates psychological problems for affected individuals. The combination of anxiety and depression leads to social withdrawal which requires complete mental health treatment.
The JIA treatment guidelines state that there is no specific diet which can treat this condition yet the Mediterranean dietary pattern which includes anti-inflammatory foods can work together with medical treatments. The combination of age-appropriate physical activity and sufficient sleep and effective stress management results in better overall health.
DISCUSSION
Integration of Findings
Current knowledge about JIA has developed from its initial understanding as a single disease called “juvenile rheumatoid arthritis” into a comprehension of multiple different conditions that exhibit distinct genetic patterns and disease development routes and clinical manifestation path- ways. The existing epidemiological data together with genetic information and immunological research results demonstrate that pediatric arthritis shares common disease pathways with adult RF-positive polyarticular arthritis while also exhibiting distinct characteristics that are unique to children.
The global increase in childhood arthritis cases which the GBD 2021 study measured occurs because of three factors: better detection methods and higher disease recognition rates and actual case numbers which have risen. The higher disease frequency in high-SDI areas and female predominance indicate that both genetic factors and environmental factors and healthcare system elements interact in complex ways.
JIA genetic research has revealed both shared and distinct characteristics that separate JIA from adult RA. The PTPN22, STAT4, and TNFAIP3 variant associations with childhood- onset RA demonstrate shared autoimmune pathways, whereas oligoarticular JIA shows distinct pediatric characteristics through different HLA associations. The cumulative risk model using weighted genetic risk scores demonstrates that multiple variants contribute additively, consistent with polygenic inheritance.
Researchers have developed new immunopathogenetic knowledge which shows that T-cell and B-cell systems interact with innate immunity elements and joint tissue components in their native environment. Researchers study neutrophil extracellular traps to understand how basic biological processes lead to new treatment methods.
Clinical Implications
The treat-to-target method which uses biological treatments has created better results for JIA treatment. Remission which used to be uncommon has become possible for many children today. The following problems continue to exist:
• Treatment access: Biologic therapies remain expensive and may be unavailable in resource- limited settings.
• Uveitis management: Ocular disease may progress independently of arthritis which needs separate monitoring and treatment algorithms.
• Transition to adult care: Adolescents need structured transition programs for maintaining disease control throughout their adult lives.
• Long-term safety: The safety profile of biologic agents needs ongoing surveillance because it requires monitoring from the beginning of their use in childhood.
The PAFLAR guidelines demonstrate how management needs to be adapted according to different regional conditions because they show that the best medical treatment needs to use both scientific evidence and the available local resources and existing limitations.
Research Gaps and Future Directions
The field has made significant advancements, yet important knowledge deficiencies continue to exist.
Predictive biomarkers: Reliable biomarkers, which can forecast disease progression and treatment outcomes and complication risks, will enable doctors to deliver customized treatment. Current candidates include autoantibodies, cytokine profiles, and genetic signatures, but none of these markers have demonstrated practical value in clinical settings.
Treatment optimization: The medical community still needs to determine which treatment sequence should start with methotrexate before moving to biologics and which biologic should be used as the primary treatment and at what point to reduce treatment intensity. The field requires comparative effectiveness research and randomized trials to achieve its goals.
The established genetic links to the disease require further research to determine how risk variants interact with environmental factors. Single-cell technologies and multiomic integration will uncover the pathways that cause the disease.
Prevention: Can JIA be prevented in high-risk individuals? This question, which used to be theoretical, will become answerable through advances in risk prediction technology.
Long-term outcomes: The first generation of children who received biologic treatment will reach adulthood, and their functional outcomes, psychosocial development, and health results will provide insights for their future health management.
Limitations
This review has multiple restrictions which limit its findings. First, the use of different terms (JRA and JIA) throughout historical and modern scientific writing creates difficulties for re- searchers who want to combine information from different sources. The rapidly changing nature of the field requires current research to include recent data, yet some findings will only remain relevant until upcoming information becomes available. The geographical and demographic differences faced by researchers create challenges to their ability to apply certain epidemiological findings to different population groups.
CONCLUSION
The condition of Juvenile Idiopathic Arthritis includes various forms of chronic inflammatory arthritis which affect children with a prevalence rate of 1 out of 1000 while causing major disruptions to their physical abilities, growth development, eyesight, and overall life satisfaction. The disease develops through a combination of genetic factors which include HLA and non-HLA genetic variants together with environmental elements that lead to immune system disorders which impact both types of immune defense mechanisms. Multiomic studies have discovered new biological pathways which involve T-cell receptor signaling together with neutrophil extra- cellular traps and specific joint microenvironments that will become the basis for developing new treatments.
The medical community needs to use seven ILAR subtypes for patient classification be- cause these subtypes provide essential information for both patient prognosis and treatment decision-making though new research shows that immunological classification will become a valuable addition to existing phenotype-based methods. The introduction of biologic DMARDs which target specific inflammatory pathways has transformed treatment approaches because these drugs help many children achieve remission when doctors use treat-to-target strategies together with early aggressive treatment. The latest guidelines recommend doctors to provide multidisciplinary treatment while they should conduct regular checks for uveitis and create a planned process to move patients into adult healthcare services.
The global increase in childhood arthritis cases shown by epidemiological research results demonstrates that researchers must continue to study the condition while treatment accessibility requires improvement and researchers must create predictive biomarkers which will help customize patient treatment. The advance of scientific understanding about JIA disease mechanisms together with the growth of treatment options brings us closer to the goal of achieving disease prevention and finding a cure. The current forecast for JIA children shows better out- comes than previous times, but society requires to work toward accessibility of treatment for all children across different regions.
References
[1] “How Common Is Juvenile Rheumatoid Arthritis?” Advance Study, 2026. [Online article]
[2] “(Juvenile Idiopathic Arthritis).” , People’s Medical Publishing House. [Clinical reference]
[3] “Juvenile Rheumatoid Arthritis: Causes, Symptoms, Diagnosis, and Treatment.” Sancheti Hospital, 2026. [Online article]
[4] Fung A, Yue X, Wigle RR, Guo JJ. “Literature analysis and implication of biologic therapy for children with non-systemic juvenile idiopathic arthritis in real-world settings.” In- tractable Rare Dis Res, 2025;14(3):162-174. [Peer-reviewed article]
[5] Prahalad S, Conneely KN, Jiang Y, et al. “Susceptibility to childhood-onset rheumatoid arthritis: Investigation of a weighted genetic risk score that integrates cumulative effects of variants at five genetic loci.” Arthritis Rheum, 2013;65(6):1663-1667. [Peer-reviewed article]
[6] Rosenbaum D, et al. “Juvenile Idiopathic Arthritis and Spondylarthritis.” Semin Musculoskelet Radiol, 2025;29(2):249-266. [Peer-reviewed article]
[7] Hassan W, et al. “PAFLAR guidelines for oligoarticular juvenile idiopathic arthritis.” Clin Rheumatol, 2026;45(2):1323-1336. [Practice guideline]
[8] Simonds MM, et al. “Pathogenesis of juvenile idiopathic arthritis.” Curr Opin Rheumatol, 2025;37(5):321-326. [Peer-reviewed article]
[9] “Global burden and genetic insights of RA and JIA in ages 0-19 years: GBD 2021 and MR analysis.” Front Immunol, 2026. [Peer-reviewed article]
[10] “Juvenile Idiopathic Arthritis.” Arthritis Foundation, 2026. [Online resource]
