Parkinson's Disease A Comprehensive Medical Review
1. Toychieva Zarina
(1. Teacher, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.)
Abstract
Parkinson's disease (PD) is the second most common neurodegenerative disorder globally, affecting over 10 million people worldwide. Characterized by the progressive loss of dopaminergic neurons in the substantia nigra pars compacta, PD presents with hallmark motor symptoms — bradykinesia, rigidity, resting tremor, and postural instability — alongside a constellation of non-motor features that significantly impair quality of life. This comprehensive review synthesizes current understanding of PD's epidemiology, neuropathological mechanisms, clinical presentation, diagnostic criteria, and evidence-based treatment strategies. We examine both established pharmacological interventions and emerging therapeutic frontiers, including gene therapy, neuroprotective agents, and stem cell approaches, while emphasizing the urgent need for disease-modifying treatments. The article underscores the enormous personal, societal, and economic burden of PD and advocates for continued investment in research and multidisciplinary care models.
1. Introduction
Parkinson's disease (PD) stands as one of the most formidable challenges in modern neurology. First formally described by the English physician James Parkinson in his landmark 1817 essay, "An Essay on the Shaking Palsy," the disease has since emerged from relative obscurity to become a subject of intense scientific scrutiny and a growing global health crisis. As the world's population ages, the burden of PD continues to rise at an unprecedented rate, demanding urgent attention from the medical community, policymakers, and society at large.
At its core, Parkinson's disease is a progressive neurodegenerative disorder characterized by the selective and irreversible loss of dopamine-producing neurons in a region of the brain known as the substantia nigra. Dopamine, a critical neurotransmitter, plays an indispensable role in coordinating smooth, purposeful movement. Its depletion results in the characteristic motor symptoms that define PD — the tremor that betrays itself at rest, the stiffness that tightens the body's movement, the slowness that turns simple actions into monumental tasks, and the instability that threatens every step.
Yet Parkinson's disease is far more than a movement disorder. Decades of research have revealed a complex, multisystem condition with profound non-motor manifestations — cognitive decline, psychiatric disturbances, autonomic dysfunction, sleep disorders, and sensory abnormalities — that often precede motor symptoms by years and significantly impair patients' quality of life. This broader understanding has transformed the way clinicians approach diagnosis, treatment, and patient care.
This article provides a comprehensive, up-to-date review of Parkinson's disease, covering its epidemiology, pathophysiology, clinical features, diagnostic approaches, current and emerging treatments, and the pressing challenges that lie ahead. It is written for a broad medical and scientific audience seeking a thorough yet accessible understanding of this complex condition.
2. Epidemiology: The Global Burden
Parkinson's disease affects individuals across every country, ethnicity, and socioeconomic background, though its distribution is not uniform. Understanding its epidemiology is critical for allocating healthcare resources, identifying risk factors, and projecting future care needs.
2.1 Prevalence and Incidence
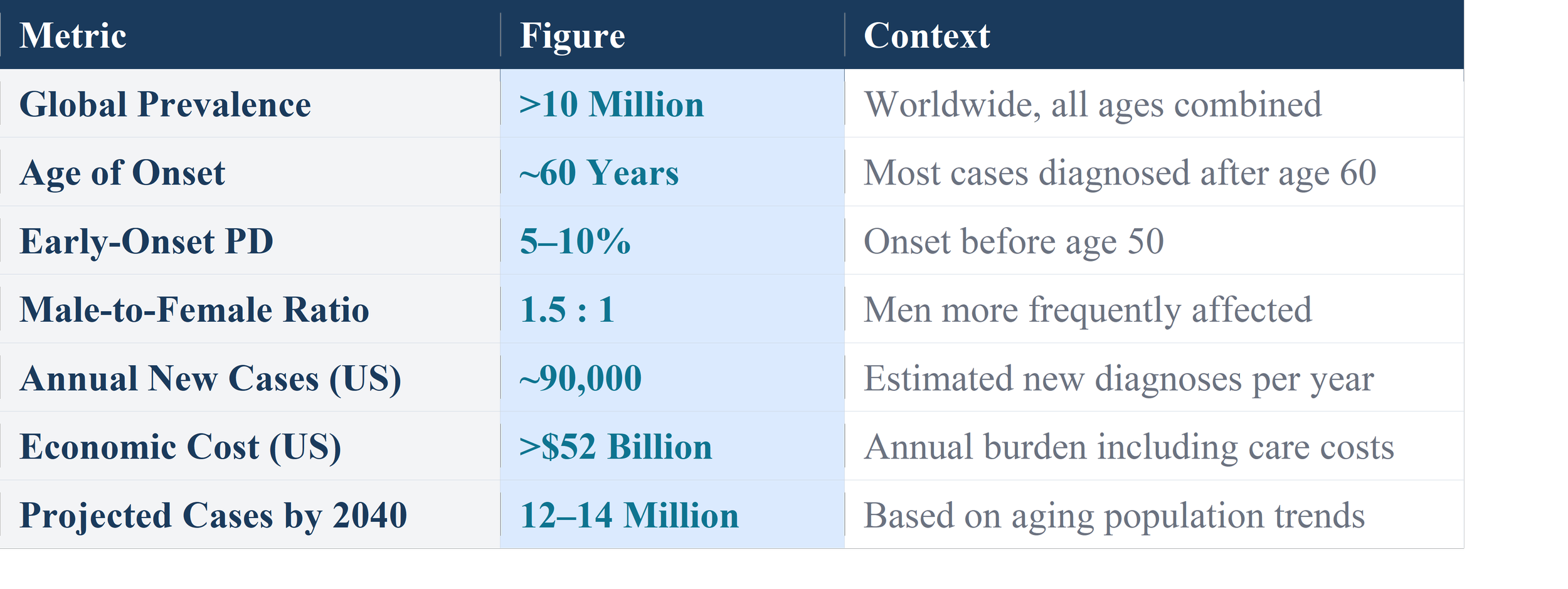
PD is the second most common neurodegenerative disorder after Alzheimer's disease. Global estimates suggest that over 10 million people currently live with Parkinson's disease. In high-income countries, prevalence ranges from approximately 100 to 200 cases per 100,000 people in the general population, rising sharply with age — reaching 1–2% among individuals over 60 years and up to 4% in those over 80 years.
The incidence of PD — the rate of new diagnoses — is estimated at 8 to 18 new cases per 100,000 persons per year in Western populations, with significant variation by geographic region, age, and sex. Men are approximately 1.5 times more likely to develop PD than women, a disparity attributed to hormonal, genetic, and occupational exposure differences.
2.2 Risk Factors
The etiology of PD is multifactorial, involving a complex interplay of genetic predisposition and environmental exposures. Several well-established risk factors have been identified:
• Age: The single most significant risk factor; prevalence increases exponentially with advancing age.
• Sex: Male sex is associated with higher risk; hormonal influences of estrogen may be neuroprotective.
• Genetics: Approximately 10–15% of PD cases have a clear genetic basis. Mutations in genes such as LRRK2, PINK1, Parkin, SNCA, and DJ-1 have been identified.
• Pesticide and herbicide exposure: Agricultural chemicals, particularly rotenone and paraquat, have been strongly linked to increased PD risk.
• Head trauma: Repeated traumatic brain injury is an established risk factor.
• Rural living and well water consumption: Epidemiological studies suggest associations, possibly related to environmental chemical exposure.
Conversely, certain factors appear to confer protection against PD. Cigarette smoking — despite its many harms — has consistently been associated with reduced PD risk in epidemiological studies, possibly due to nicotine's dopaminergic effects. Regular caffeine consumption and vigorous physical exercise have also been associated with lower PD incidence.
3. Pathophysiology: Understanding the Diseased Brain
The pathophysiology of Parkinson's disease is among the most extensively studied in all of neuroscience, yet many fundamental questions remain unanswered. What is clear is that PD involves a progressive, selective destruction of specific neuronal populations that disrupts the delicately balanced circuitry governing movement and many other functions.
3.1 The Dopaminergic System and Substantia Nigra
The substantia nigra pars compacta (SNpc), a small but critically important region in the midbrain, is the primary site of neurodegeneration in PD. The SNpc contains large populations of dopaminergic neurons — cells that synthesize and release dopamine. These neurons project their axons via the nigrostriatal pathway to the striatum (caudate nucleus and putamen), forming a critical circuit that modulates voluntary movement.
In healthy individuals, dopamine released in the striatum acts as a key modulator within the basal ganglia, a set of subcortical nuclei that regulate movement initiation, sequencing, and coordination. The basal ganglia operate through a complex balance of excitatory and inhibitory pathways — the "direct" pathway (facilitating movement) and the "indirect" pathway (suppressing unwanted movement). Dopamine acts to facilitate the direct pathway and inhibit the indirect pathway, net result being smooth, coordinated movement.
In PD, progressive loss of SNpc dopaminergic neurons — typically exceeding 60–80% loss before motor symptoms become apparent — disrupts this balance profoundly. The indirect pathway becomes over-active, while the direct pathway is under-facilitated, resulting in the characteristic motor symptoms of PD.
3.2 Alpha-Synuclein and Lewy Bodies
A defining neuropathological hallmark of PD is the presence of Lewy bodies — abnormal, intracellular protein aggregates found in degenerating neurons. These structures are composed predominantly of misfolded alpha-synuclein (alpha-Syn), a small presynaptic protein whose normal function relates to vesicle trafficking and neurotransmitter release.
In PD, alpha-synuclein misfolds and aggregates into oligomeric species and eventually into the insoluble fibrils that constitute Lewy bodies. These aggregates are toxic to neurons through multiple mechanisms — disrupting mitochondrial function, impairing the ubiquitin-proteasome system responsible for protein degradation, inducing oxidative stress, and triggering neuroinflammation.
Braak and colleagues proposed a staging hypothesis suggesting that alpha-synuclein pathology begins in the peripheral and enteric nervous systems and in the olfactory bulb before spreading in a predictable, caudo-rostral pattern through the brainstem, finally reaching the cerebral cortex in later disease stages. This "prion-like" propagation model has significant implications for understanding pre-motor symptoms and designing early intervention strategies.
3.3 Mitochondrial Dysfunction and Oxidative Stress
Mitochondrial dysfunction plays a central role in PD pathogenesis. The substantia nigra is particularly vulnerable due to its high metabolic demands, elevated dopamine turnover — which generates oxidative byproducts — and relatively limited antioxidant defenses. Complex I of the mitochondrial electron transport chain is inhibited in PD patients, leading to reduced ATP production and increased generation of reactive oxygen species (ROS).
Oxidative stress damages cellular lipids, proteins, and DNA. The resulting neuroinflammation — mediated by activated microglia — further amplifies neuronal injury, creating a vicious cycle of degeneration. Genes implicated in familial PD (PINK1, Parkin, DJ-1) all play roles in mitochondrial quality control, underscoring the centrality of mitochondrial health to PD pathogenesis.
3.4 Neuroinflammation
Activated microglia and astrocytes are prominent features of the PD brain. Neuroinflammation, driven by alpha-synuclein aggregates and other damage-associated molecular patterns, contributes to dopaminergic cell death. Pro-inflammatory cytokines including TNF-alpha, IL-1beta, and IL-6 are elevated in the PD brain and cerebrospinal fluid. Peripheral immune cells also infiltrate the brain parenchyma, amplifying the inflammatory cascade. Targeting neuroinflammation is an active area of therapeutic investigation.
4. Clinical Features: Recognizing Parkinson's Disease
Parkinson's disease presents with a heterogeneous constellation of motor and non-motor symptoms that evolve over the disease course. No two patients follow exactly the same trajectory, and recognizing the full spectrum of manifestations is essential for accurate diagnosis and holistic management.
4.1 Cardinal Motor Symptoms
The four cardinal motor features of PD — collectively captured by the mnemonic TRAP — form the diagnostic cornerstone:
Tremor
The most recognizable feature of PD is the resting tremor — a rhythmic, 4–6 Hz oscillation typically beginning in a hand (classically described as "pill-rolling" between thumb and forefinger) and occurring when the affected limb is at rest. Unlike action tremors, PD tremor diminishes with voluntary movement and disappears during sleep. Tremor may begin unilaterally but typically spreads to involve the contralateral side and sometimes the jaw, lips, or tongue.
Rigidity
Muscular rigidity — increased resistance to passive movement throughout the range of motion — affects the vast majority of PD patients. It may present as smooth "lead-pipe" rigidity or as "cogwheel" rigidity, where a ratchet-like quality is superimposed due to underlying tremor. Rigidity contributes to the characteristic stooped posture and masked facial expression (hypomimia) seen in PD.
Akinesia and Bradykinesia
Bradykinesia — slowness of movement — is the most disabling of the cardinal motor features and is required for the clinical diagnosis of PD. It manifests as difficulty initiating movement, slowness of execution, and decrement in amplitude with repetitive movements. Patients exhibit reduced arm swing during walking, small, cramped handwriting (micrographia), soft speech (hypophonia), reduced blinking, and difficulty with fine motor tasks.
Postural Instability
Impaired postural reflexes, typically emerging in mid-to-late disease, predispose patients to falls — a major source of morbidity in PD. Patients adopt a flexed, stooped posture and may exhibit festination (involuntary acceleration of gait with shortened shuffling steps) and freezing of gait — sudden, transient inability to initiate or continue walking.
4.2 Non-Motor Symptoms
Non-motor symptoms (NMS) are increasingly recognized as central to the PD experience, often preceding motor symptoms by years (the "pre-motor" phase) and profoundly affecting quality of life. Key non-motor manifestations include:

REM sleep behavior disorder (RBD) — in which patients physically enact their dreams due to loss of normal motor inhibition during sleep — is particularly noteworthy as a strong predictor of PD and other synucleinopathies, sometimes preceding motor diagnosis by a decade or more.
5. Diagnosis: Clinical Criteria and Investigations
The diagnosis of Parkinson's disease remains primarily clinical, based on careful history-taking and neurological examination. There is currently no definitive biomarker test that can confirm PD diagnosis in living patients with absolute certainty, though this landscape is rapidly evolving.
5.1 Diagnostic Criteria
The most widely used diagnostic framework is the Movement Disorder Society (MDS) Clinical Diagnostic Criteria for PD, updated in 2015. These criteria center on the presence of parkinsonism — defined as bradykinesia in combination with rest tremor and/or rigidity — and apply a system of supportive criteria, absolute exclusion criteria, and red flags to increase diagnostic accuracy.

5.2 Differential Diagnosis
Several conditions can mimic PD and must be carefully excluded. Atypical parkinsonian syndromes — collectively termed "Parkinson-plus" syndromes — include Progressive Supranuclear Palsy (PSP), Multiple System Atrophy (MSA), and Corticobasal Degeneration (CBD). These conditions share some features with PD but have distinct clinical profiles, respond poorly to levodopa, and carry a worse prognosis.
Secondary causes of parkinsonism — including drug-induced parkinsonism (from dopamine-blocking agents), vascular parkinsonism, and normal pressure hydrocephalus — must also be systematically excluded.
5.3 Investigations and Biomarkers
While PD is diagnosed clinically, investigations play an important role in supporting diagnosis and excluding alternatives. Key investigations include:
• Dopamine Transporter (DAT) SPECT Imaging (DaTSCAN): Demonstrates reduced dopamine transporter density in the striatum, supporting PD diagnosis. Does not differentiate PD from atypical parkinsonian syndromes.
• MRI Brain: Primarily used to exclude structural causes; may show characteristic changes in MSA or PSP.
• MIBG Cardiac Scintigraphy: Demonstrates cardiac sympathetic denervation in PD, helping distinguish from MSA.
• Neuropsychological Testing: Evaluates cognitive function and helps characterize the nature and severity of cognitive impairment.
• Polysomnography: Confirms REM sleep behavior disorder when clinically suspected.
• Alpha-Synuclein Seed Amplification Assays (SAA): An emerging and highly promising biomarker test detecting pathological alpha-synuclein in cerebrospinal fluid or skin biopsies — potentially transforming future early diagnosis.
6. Treatment: A Multidisciplinary Approach
There is currently no cure for Parkinson's disease and no proven disease-modifying therapy — that is, no treatment that slows or halts underlying neurodegeneration. However, remarkable progress has been made in symptomatic management, and patients today have access to a sophisticated array of pharmacological and non-pharmacological interventions that can significantly improve function and quality of life.
6.1 Pharmacological Treatment
Levodopa — The Gold Standard
Levodopa, the metabolic precursor to dopamine, remains the most effective and widely used treatment for PD motor symptoms after more than 50 years of clinical use. Administered orally in combination with a peripheral decarboxylase inhibitor (carbidopa in the US; benserazide in Europe) to prevent peripheral conversion and reduce side effects, levodopa dramatically reduces motor symptoms in the majority of patients.
However, long-term levodopa therapy is complicated by motor fluctuations — the "wearing off" of drug effect before the next dose, leading to alternating "on" and "off" states — and levodopa-induced dyskinesias (LID), involuntary writhing movements that occur at peak drug levels. These complications develop in approximately 50% of patients after 5 years of therapy and are a major challenge in PD management.
Dopamine Agonists
Dopamine agonists (pramipexole, ropinirole, rotigotine, apomorphine) directly stimulate dopamine receptors in the striatum. They are associated with fewer motor fluctuations when used as initial therapy but carry a risk of impulse control disorders (gambling, hypersexuality, binge eating) and neuropsychiatric side effects, particularly in older patients. Apomorphine, administered subcutaneously as a continuous infusion or "rescue" injection, is particularly useful for managing severe motor fluctuations.
MAO-B Inhibitors and COMT Inhibitors
Monoamine oxidase B (MAO-B) inhibitors (selegiline, rasagiline, safinamide) reduce dopamine breakdown and extend its action, providing mild symptomatic benefit and helping to smooth motor fluctuations. Catechol-O-methyltransferase (COMT) inhibitors (entacapone, opicapone, tolcapone) block levodopa degradation in the periphery and brain, prolonging its half-life and reducing "off" time.

6.2 Surgical Therapies
Deep Brain Stimulation (DBS)
Deep Brain Stimulation (DBS) represents the most significant surgical advance in PD treatment. High-frequency electrical stimulation is delivered via implanted electrodes to specific brain targets — most commonly the subthalamic nucleus (STN) or the globus pallidus interna (GPi). DBS can dramatically reduce motor fluctuations, dyskinesias, and tremor in carefully selected patients, typically those with good levodopa responsiveness who have developed problematic motor fluctuations.
DBS does not slow disease progression and is less effective for axial symptoms (gait, balance, speech) and non-motor features. Patient selection, surgical expertise, and ongoing programming by specialized neurologists are critical determinants of outcome.
Focused Ultrasound and Lesioning
Magnetic resonance-guided focused ultrasound (MRgFUS) thalamotomy — delivering precisely targeted ultrasound energy to create a therapeutic lesion in the thalamus — has received regulatory approval for tremor-dominant PD and offers a non-invasive alternative to DBS for selected patients.
6.3 Non-Pharmacological Therapies
Exercise is arguably the most evidence-supported non-pharmacological intervention in PD. Aerobic exercise, resistance training, balance training (tai chi, yoga), and dance therapy have all demonstrated benefits for motor function, balance, and quality of life, with emerging evidence suggesting potential neuroprotective effects.
• Physiotherapy: Addresses gait, balance, falls prevention, and motor function.
• Speech and Language Therapy: Manages hypophonia and dysphagia; LSVT LOUD is a validated intensive voice treatment program.
• Occupational Therapy: Optimizes activities of daily living and home adaptations.
• Neuropsychological support and cognitive rehabilitation for cognitive symptoms.
• Dietitian input: Protein redistribution to optimize levodopa absorption; nutritional support.
• Palliative and psychological support: For depression, anxiety, and end-of-life planning.
7. Emerging Therapies and Future Directions
The greatest unmet need in Parkinson's disease is a therapy that modifies the underlying course of neurodegeneration — slowing, halting, or reversing disease progression. The past decade has witnessed an explosion of research into disease-modifying strategies, driven by deeper understanding of PD pathobiology and advances in biotechnology.
7.1 Alpha-Synuclein Targeted Therapies
Given the central role of alpha-synuclein aggregation in PD pathogenesis, therapies aimed at reducing its production, aggregation, or propagation have attracted enormous interest. Approaches include:
• Immunotherapy: Active vaccines and passive immunization with monoclonal antibodies targeting extracellular alpha-synuclein have entered clinical trials. Early trials have established safety; efficacy data are awaited.
• SNCA gene silencing: Antisense oligonucleotides (ASOs) and RNA interference (RNAi) approaches to reduce alpha-synuclein production are under active investigation, with promising preclinical data.
• Small molecule aggregation inhibitors: Compounds designed to prevent alpha-synuclein from adopting toxic conformations represent another therapeutic avenue.
7.2 Gene Therapy
Viral vector-mediated gene therapy offers the potential to deliver therapeutic genes directly to the brain. Several approaches are in clinical trials, including delivery of genes encoding AADC (aromatic amino acid decarboxylase) to enhance dopamine synthesis, neurturin (a neurotrophic factor) to support surviving neurons, and GDNF to promote neuronal survival and function. Gene therapy targeting LRRK2 in mutation carriers represents a precision medicine approach.
7.3 Stem Cell Therapies
Cell replacement therapy — transplanting dopamine-producing neurons derived from human embryonic stem cells (hESCs) or induced pluripotent stem cells (iPSCs) into the striatum — aims to restore the lost dopaminergic supply. Several Phase I/II trials are underway globally, building on lessons from earlier fetal mesencephalic transplantation studies of the 1990s. The field has benefited enormously from advances in stem cell biology and our ability to generate authentic midbrain dopaminergic neurons in vitro.
7.4 Neuroprotective Strategies
Multiple candidate neuroprotective agents have been assessed in clinical trials — including coenzyme Q10, creatine, isradipine (a calcium channel blocker), and exenatide (a GLP-1 receptor agonist) — with mixed results. Exenatide and other GLP-1 agonists have generated particular excitement following a Phase II trial suggesting a possible disease-modifying effect; larger Phase III trials are ongoing.
7.5 Digital Health and Precision Medicine
Wearable sensors capable of continuously monitoring PD motor symptoms in real-world settings are transforming clinical assessment and personalized therapy optimization. Machine learning algorithms applied to sensor data, speech analysis, and gait parameters hold promise for more accurate diagnosis, disease staging, and treatment response monitoring.
The emerging field of precision medicine in PD — stratifying patients by genetic subtype, biomarker profile, and disease stage to tailor therapies — represents the future of PD research. The Michael J. Fox Foundation's PPMI (Parkinson's Progression Markers Initiative) and similar large longitudinal biomarker studies are building the foundational datasets needed to realize this vision.
8. Living with Parkinson's Disease
Beyond biology and pharmacology, Parkinson's disease is a profoundly human condition — one that reshapes every dimension of a patient's life and touches their families, caregivers, and communities. Understanding the lived experience of PD is essential for compassionate, patient-centered care.
8.1 Psychosocial Impact
The diagnosis of a progressive, incurable neurological condition carries an enormous psychological burden. Depression and anxiety are among the most common non-motor symptoms, affecting approximately 40% and 30% of PD patients respectively, and are independently associated with reduced quality of life. Apathy — a loss of motivation and goal-directed behavior distinct from depression — is particularly challenging to treat and is strongly associated with cognitive impairment.
Social isolation, relationship strain, loss of independence, and changes in occupational and recreational roles compound the psychological impact. Stigma — both self-imposed and societal — remains a significant barrier to help-seeking and social participation.
8.2 Caregiver Burden
The vast majority of people with PD rely substantially on informal caregivers — most often spouses, partners, or children — for daily support. As PD progresses, caregiver demands escalate, encompassing personal care, medication management, emotional support, and advocacy. Caregiver burnout, depression, and physical ill-health are highly prevalent and represent a major public health concern that receives insufficient attention.
8.3 Multidisciplinary Care
The complexity of PD demands a multidisciplinary approach. Expert consensus and growing evidence support the superiority of specialized, coordinated PD care — involving neurologists, specialist nurses, physiotherapists, occupational therapists, speech therapists, neuropsychologists, dietitians, social workers, and palliative care teams — over conventional neurology-only management.

9. Conclusion
Parkinson's disease represents one of the defining medical challenges of our time. Its rising prevalence, complex multisystem pathology, devastating impact on individuals and families, and enormous economic cost demand unwavering scientific commitment and policy attention. The past two decades have brought transformative advances in our understanding of PD biology, dramatic improvements in symptomatic treatment, and the emergence of a rich pipeline of potential disease-modifying therapies. Yet the fundamental goal — a treatment that arrests the neurodegenerative process — remains elusive.
The path forward requires integration across multiple fronts: continued investment in fundamental neuroscience; rigorous and innovative clinical trial design; development and validation of sensitive biomarkers for early detection and trial stratification; global collaboration to build the large, diverse cohorts necessary for precision medicine; and unwavering commitment to the well-being of patients and caregivers through comprehensive, multidisciplinary care.
Most critically, the voices, experiences, and priorities of people living with Parkinson's disease must remain at the center of the research enterprise and healthcare system. They are not merely the subjects of scientific inquiry but the driving moral force behind it — and ultimately the beneficiaries of every advance that brings us closer to a world free of Parkinson's disease.
References:
1. Parkinson J. An Essay on the Shaking Palsy. London: Sherwood, Neely, and Jones; 1817.
2. Dorsey ER, Bloem BR. The Parkinson Pandemic — A Call to Action. JAMA Neurology. 2018;75(1):9–10.
3. Postuma RB, et al. MDS clinical diagnostic criteria for Parkinson's disease. Movement Disorders. 2015;30(12):1591–1601.
4. Obeso JA, et al. Past, present, and future of Parkinson's disease: A special essay on the 200th Anniversary of the Shaking Palsy. Movement Disorders. 2017;32(9):1264–1310.
5. Braak H, et al. Staging of brain pathology related to sporadic Parkinson's disease. Neurobiology of Aging. 2003;24(2):197–211.
6. Bloem BR, Okun MS, Klein C. Parkinson's disease. The Lancet. 2021;397(10291):2284–2303.
7. Fox SH, et al. International Parkinson and Movement Disorder Society Evidence-Based Medicine Review: Update on treatments for the motor symptoms of Parkinson's disease. Movement Disorders. 2018;33(8):1248–1266.
8. Bhidayasiri R, Tarsy D. Movement Disorders: A Video Atlas. Springer; 2012.
9. Kalia LV, Lang AE. Parkinson's disease. The Lancet. 2015;386(9996):896–912.
10. Poewe W, et al. Parkinson disease. Nature Reviews Disease Primers. 2017;3:17013.
