Reframing Clinical Tuberculosis Treatment in the Era of Short-Course Regimens and Emerging Resistance
1. Kurmanaliev Nurlan Kambaralyevich
2. Prerna Kamble
Ishwari Kolekar
Onkar Lamdade
Sneha Mahanor
Sandeep Chauhan
(1. Teacher, International Medical Faculty, Osh State University, Kyrgyzstan)
https://orcid.org/0000-0002-5952-1463
(2. Students, International Medical Faculty, Osh State University, Kyrgyzstan)
Abstract
Background: Tuberculosis (TB) continues to be the deadliest infectious disease which affects people across the globe. The medical field has experienced a fundamental transformation during the past ten years because it now uses complete oral medications which require only short treatment periods to treat both drug-susceptible TB (DS-TB) and drug-resistant TB (DR-TB) cases. This article provides an overview of current clinical tuberculosis treatment methods which include emerging treatment protocols and ongoing resistance problems and specialized population treatment techniques.
Methods: A complete non-systematic literature review established evidence from current clinical trials together with updated clinical practice guidelines from major health organizations which include WHO and ATS/CDC/ERS/IDSA and essential meta-analyses that appeared between 2022 and 2026.
Results: Current evidence demonstrates that standard DS-TB treatment re- quires 6 months while a 4-month treatment with rifapentine and moxifloxacin shows better results. The BPaLM/BPaL regimens for DR-TB have transformed patient treatment because they achieve treatment success rates of approximately 83 percent which require only 6 months to complete. The treatment of tuberculosis infection (TBI) faces new challenges because of two factors which include treating immuno- compromised patients and the development of resistance against new drugs such as bedaquiline.
Conclusions: The field of clinical TB treatment developed new short treatment options which deliver improved safety and effectiveness. The successful implementation of these treatment protocols needs a precision medicine framework which combines quick molecular tests with drug level supervision and thorough monitoring of side effects. The future points towards even shorter regimens and pan-TB treatment approaches which need strong health systems to provide equal treatment access while preventing new resistance strains from developing.
Introduction
The clinical management of tuberculosis (TB) began with a dual system that required patients to undergo six months of first-line treatment for drug-susceptible TB (DS-TB) and to endure 18-24 months of second-line injectable treatments for multidrug-resistant TB (MDR-TB) which caused severe side effects. The treatment method achieved successful outcomes through its application to millions of patients yet it resulted in extensive patient suffering and high treatment drop-out rates and delivered poor treatment results for patients with drug-resistant tuberculosis.
The past five years, however, have ushered in a revolutionary transformation. The development of TB treatments has undergone a complete change because of a strong pipeline which includes both new drugs and repurposed drugs but especially bedaquiline and pre-tomanid together with important clinical trials. The World Health Organization (WHO) together with other major organizations now recommends all-oral treatment which re- quires less time and shorter treatment times to achieve results that match drug-sensitive tuberculosis and drug-resistant tuberculosis patients. The current period enables both patients and healthcare systems to achieve reduced burden.
The current advancements create a contradictory situation. The process of treating tuberculosis infection (TBI) needs careful decisions because of increasing immunosuppressive treatments. The management of tuberculosis infection (TBI) requires healthcare professionals to make decisions that depend on different immunosuppressive treatments. The Mycobacterium tuberculosis bacteria show multiple antimicrobial resistance (AMR) mechanisms which include resistance to recently developed drugs that now endanger existing treatment successes. The establishment of new treatment methods into various healthcare systems presents a tough obstacle which particularly affects healthcare facilities located in areas with high disease rates and limited resources.
This article provides a comprehensive review of the current state of clinical TB treatment. It aims to synthesize the latest evidence for DS-TB and DR-TB, explore the complexities of managing TBI, analyze the challenge of resistance in the context of new drugs, and finally, cast a glance toward the future of TB therapeutics.
Methods
This article is a narrative review based on a targeted literature search. The author conducted a search on PubMed and the Cochrane Library to find articles that were published between January 2022 and December 2026 in order to access the latest research developments. The search strategy combined terms related to tuberculosis (“tuberculosis” “MDR-TB” “Mycobacterium tuberculosis”) with treatment-focused terms (“treatment” “therapy” “regimens” “BPaLM” “bedaquiline” “preventive treatment”). The research team prioritized clinical practice guidelines from authoritative bodies which included ATS/CDC/ERS/IDSA and WHO together with large randomized controlled trials and systematic reviews and meta-analyses. The subsequent sections summarize the key search results to establish an evidence-based overview of the field.
Results
The Evolution of Drug-Susceptible TB Treatment
The 6-month treatment program that uses isoniazid and rifampin and pyrazinamide and ethambutol during its first two months (intensive phase) and subsequently uses isoniazid and rifampin for four months (continuation phase) has served as the primary treatment method for drug-susceptible tuberculosis (DS-TB) during the past 40 years. The treat- ment proves highly effective, but its lengthy nature creates difficulties that patients face when they try to follow treatment and which health facilities need to manage.
Recent studies have effectively disproven the existing belief. The pivotal study estab- lishing a non-inferior 4-month regimen was Trial 31/AIDS Clinical Trials Group A5349, which tested a regimen substituting rifapentine for rifampin and moxifloxacin for isoni- azid [7]. The American Thoracic Society and CDC and ERS and IDSA have updated their guidelines to recommend a 4-month treatment regimen that includes rifapentine and moxifloxacin and pyrazinamide and ethambutol for eligible adults and adolescents who have pulmonary drug-susceptible tuberculosis (DS-TB) [7].
Researchers are conducting studies to find new ways to shorten time frames. The phase 2C TBTC Study 38/CRUSH-TB investigates new treatment methods that use be- daquiline and moxifloxacin and pyrazinamide (BMZ) as their core treatment, while testing both with and without rifabutin or delamanid [9]. This development indicates researchers are establishing a path toward developing treatment methods that will transform how patients experience tuberculosis treatment, which will also change how tuberculosis control affects public health.
Table 1: Comparison of Standard and Novel Regimens for Drug-Susceptible Pulmonary TB
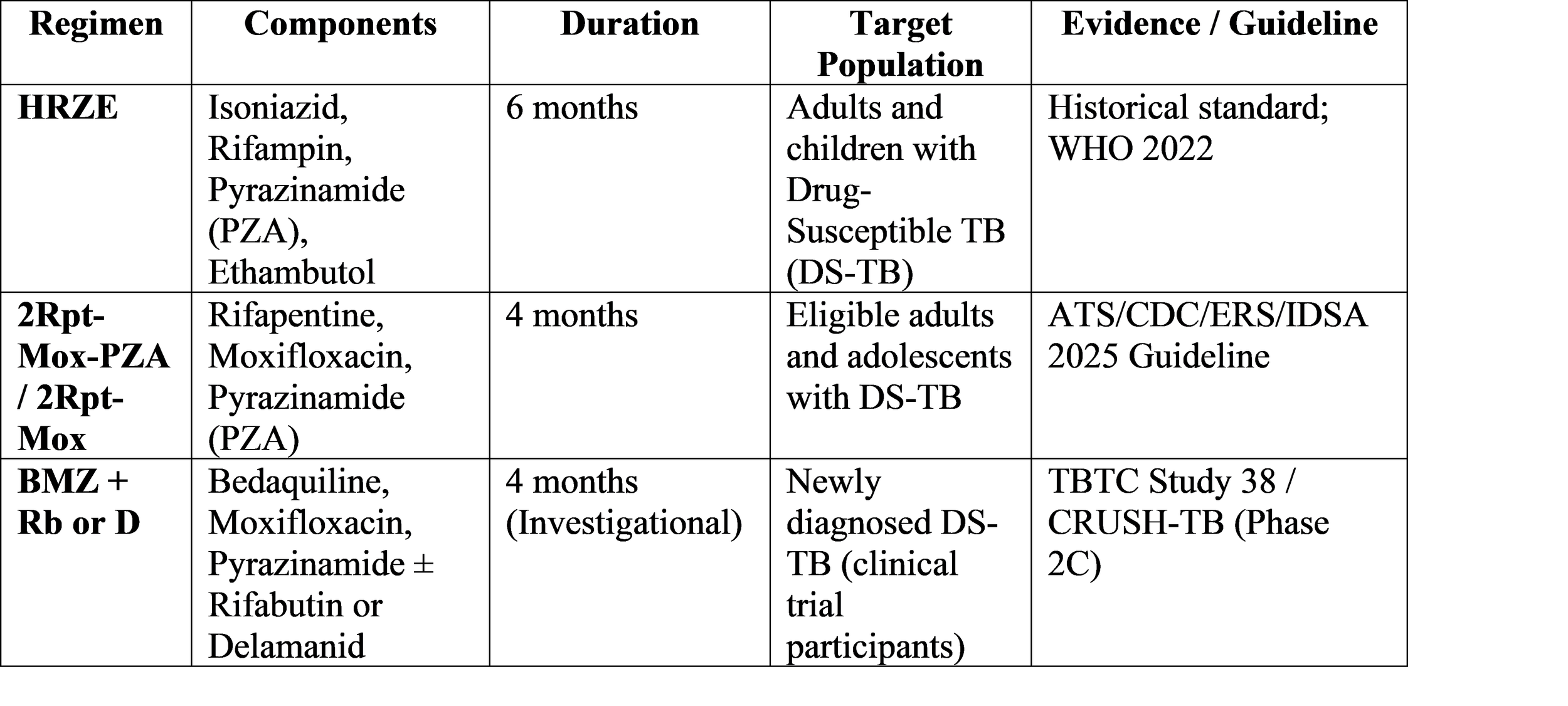
The Revolution in Drug-Resistant TB: The BPaLM Era
Drug-resistant tuberculosis treatment has experienced its most significant change yet. The previous standard of care—regimens lasting 9-24 months, often containing a toxic injectable agent and hundreds of pills—resulted in a global treatment success rate of only around 44% [8]. The introduction of bedaquiline, a first-in-class diarylquinoline, was the critical breakthrough. It serves as an essential element of every contemporary DR-TB treatment protocol.
The current standard of care for rifampicin-resistant TB (RR-TB) and MDR-TB is the 6-month, all-oral BPaLM regimen: Bedaquiline, Pretomanid, Linezolid, and Mox- ifloxacin. The regimen for patients who have developed fluoroquinolone resistance is reduced to BPaL which excludes moxifloxacin [2]. A recent systematic review and meta- analysis of all-oral bedaquiline-based regimens, including BPaLM, demonstrated a pooled treatment success rate of 83%, with mortality and loss to follow-up rates of only 5% and 4%, respectively [2]. The new treatment methods achieve almost double the success rate of previous treatment methods.
The shift is not only about efficacy but also safety. The same meta-analysis showed that shorter, bedaquiline-based regimens were associated with significantly lower rates of serious adverse events, including QT interval prolongation, compared to longer, injectable- containing regimens (Relative Risk 0.39) [2]. The successful implementation of BPaLM proves that new drug classes enable medical professionals to treat drug-resistant tuber- culosis at the same speed and efficiency as they treat drug-sensitive tuberculosis.
Table 2: Recommended All-Oral Regimens for Drug-Resistant TB
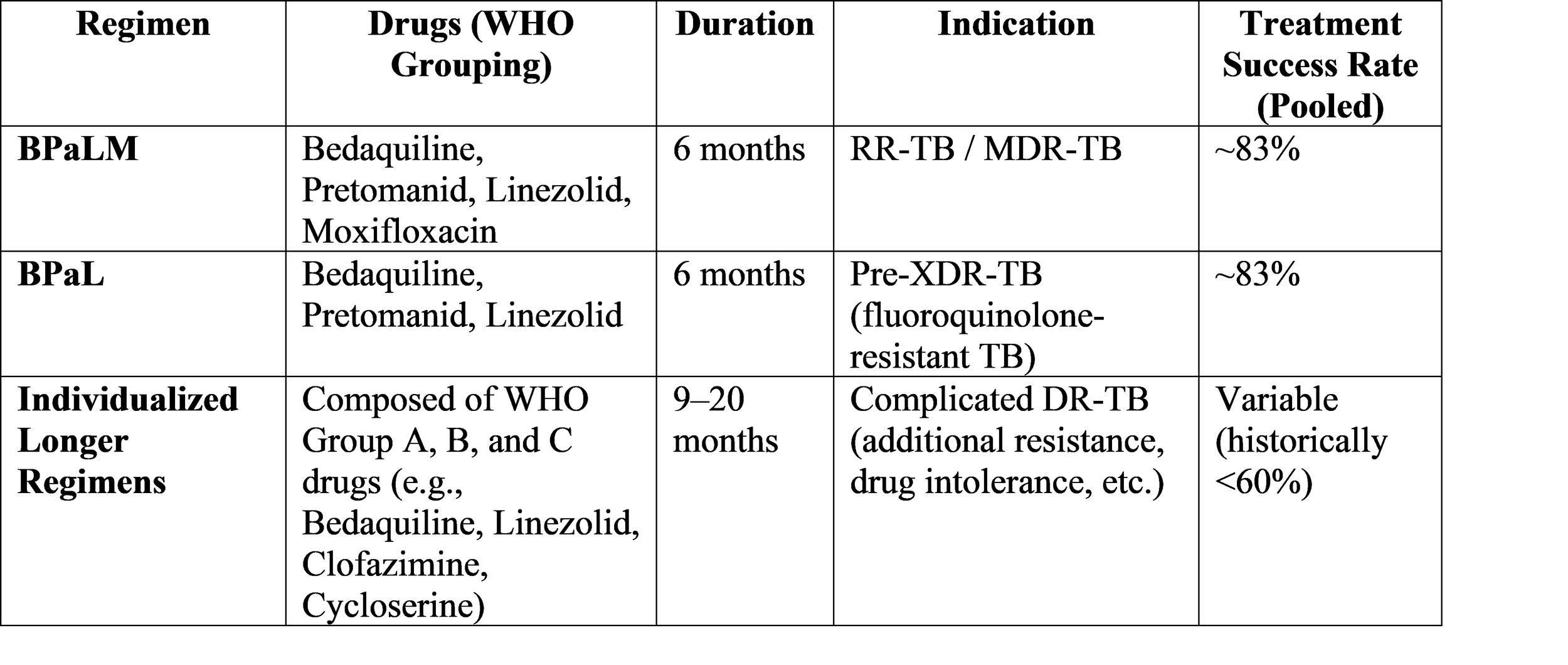
Tuberculosis Infection: Targeting High-Risk Populations
As incidence falls in many regions, the “End TB Strategy” increasingly relies on addressing the reservoir of tuberculosis infection (TBI). The goal is to identify and treat those at highest risk of progression to active disease before it occurs. Consensus statements emphasize a targeted approach, prioritizing individuals with the highest risk of reactivation [1, 4].
The following items appear on this list:
Close contacts of patients with pulmonary TB.
1. People living with HIV (PLWH), regardless of CD4 count.
2. Immunocompromised individuals who plan to start immunosuppressive therapies (e.g., TNF- inhibitors), solid organ or hematologic transplant recipients, and those on dialysis [4].
3. Patients with fibrotic lesions on chest radiographs that indicate previous TB infection.
The medical community now uses Interferon-Gamma Release Assays (IGRAs) as the primary diagnostic method for tuberculosis because these tests provide better results than the tuberculin skin test (TST) [4]. The medical field currently requires medical professionals to conduct full clinical assessments and complete chest imaging tests before beginning TBI treatment.
Medical professionals now prescribe shorter and safer preventive treatment regimens for their patients. The 3-month weekly regimen of isoniazid and rifapentine (3HP) and the 1-month daily regimen of isoniazid and rifapentine (1HP) are now preferred over the longer 6-9 months of isoniazid monotherapy, which significantly improves patient adherence to treatment [5, 6]. To achieve optimal protective effects, health professionals should begin preventive therapy at least four weeks before patients receive immunosuppressive treatment [4].
Table 3: Common Regimens for Tuberculosis Preventive Treatment (TPT)
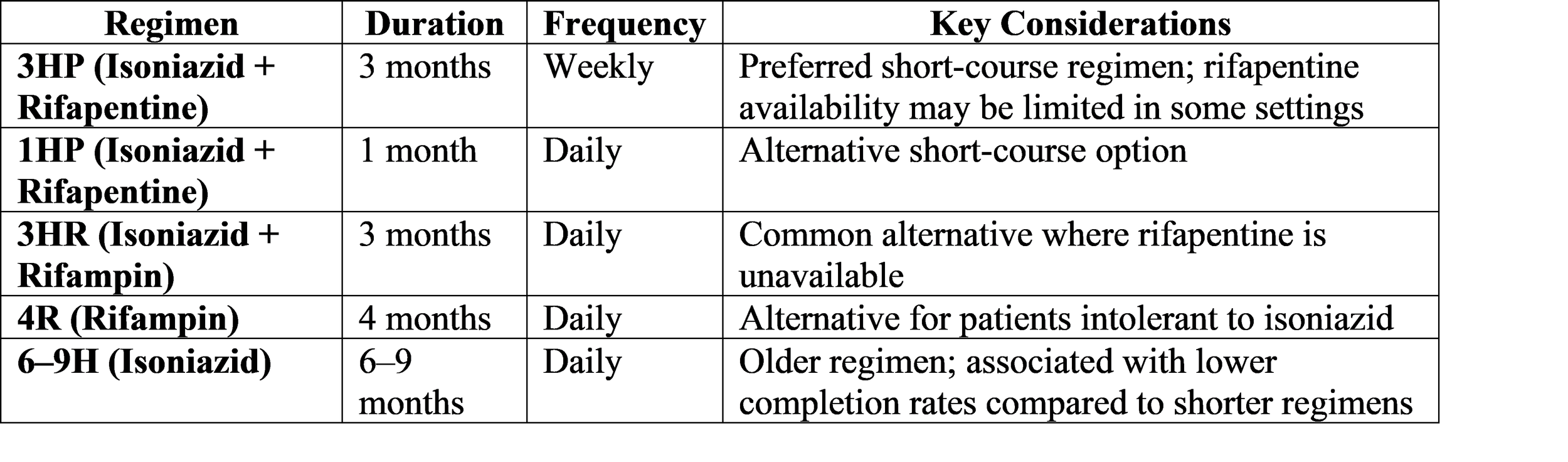
The Emerging Challenge: Resistance to Novel Drugs
The outstanding effectiveness of regimens which use bedaquiline together with premenopausal faces a major obstacle because these drugs will eventually develop resistance. The comprehensive review of AMR in M. tuberculosis describes various ways resistance develops through target gene mutations and drug efflux pump activity [3]. If bedaquiline is used alone without proper drug combinations it will create an extended period of treatment that enables resistant mutants to emerge.
There is an increasing number of reports which document bedaquiline resistance, which poses a risk to the BPaLM regimen’s ability to maintain its benefits in the future. This has led to the creation of new drugs, which now include sorfequinine (TBAJ-876), a second-generation diarylquinoline. The early-phase trials show that sorfequinine pro- vides better antimycobacterial effects, a safer profile with reduced QT prolongation, and essential effectiveness against specific bedaquiline-resistant strains. The SPaL regimen, which combines sorfequinine with pretomanid and linezolid, has progressed to Phase 3 trials to develop better methods than current resistance patterns [10].
Discussion
The review results demonstrate that TB clinical practice has experienced an essential transformation which now stands as an unchangeable fact. The treatment duration question now changes from an unknown duration to an established time frame. The transition to 4-month regimens for DS-TB and 6-month regimens for DR-TB represents a monumental victory for translational research and clinical trial science [5, 8]. The two main obstacles which prevent success in this field have received direct solutions through these innovations because they eliminate both treatment time requirements and harmful side effects. The health system requires shorter patient engagement periods which will boost patient adherence while decreasing instances of patient drop-out and reducing the spread of disease in the community.
The process of implementing these improvements requires efforts which face a fundamental contradiction because treatment becomes more straightforward while treatment contexts grow increasingly intricate. The BPaLM regimen requires baseline drug susceptibility testing (DST) to identify fluoroquinolone resistance because it depends on this testing for its success. Many high-burden areas lack sufficient access to quick molecular tests which detect second-line drugs thus increasing the chance that doctors will improperly administer these advanced treatment methods which lead to drug resistance development.
TBI management needs cross-specialty cooperation between pulmonologists and rheumatologists and HIV specialists because systems which utilize immunomodulatory treatments need this teamwork to function. The decision to test and treat must be carefully balanced against the risk of hepatotoxicity from preventive therapy and the urgency of treating the underlying immunosuppressive condition [4].
The difficulty increases because functional monotherapy together with acquired resistance presents a danger to the system. The new drug bedaquiline provides strong effectiveness but its lengthy half-life creates both advantages and disadvantages for its users. When patients do not follow their treatment plan properly or choose ineffective companion medications their chances of experiencing a relapse with resistant infections increase [3]. The evidence demonstrates that therapeutic drug monitoring (TDM) must be implemented in specific patient situations which include malabsorption and drug-drug interactions and delayed drug response to establish proper drug levels and stop resistance development. TDM implementation faces limitations because it exists as a vital link between advanced scientific discoveries and standard medical procedures.
The review faces its main restriction because it follows a non-systematic approach which brings about selection bias problems. The study presents current information about a field that evolves quickly because upcoming Phase 3 SPaL trial and CRUSH-TB trial results will require modifications to clinical guidelines within the next few years.
The ultimate objective for future development establishes a single treatment method that delivers safe and efficient results for all tuberculosis strains, including those which show resistance. The study of the BMZ backbone in DS-TB combined with the development of sorfequinine represents progress towards this objective. The achievement of this objective requires ongoing drug development work together with health systems investment to deliver diagnostic tools and medications and specialized knowledge needed for dealing with remaining difficulties.
Conclusion
The field of clinical tuberculosis treatment has introduced an innovative treatment approach. The previous treatment methods which required extended use of hazardous medications have been abandoned in favor of short all-oral treatment programs that medical research has validated as effective. The medical community now uses four-month treatment programs to treat drug-susceptible tuberculosis. The BPaLM regimen has established new treatment standards for drug-resistant tuberculosis by achieving success rates which were impossible to achieve ten years ago. The management of tuberculosis infection is becoming more targeted and patient-friendly, focusing on those at highest risk of progression.
The achievement exists in a state of weakness. The emergence of resistance to novel drugs, the complexities of implementation in resource-limited settings, and the nuanced needs of immunocompromised populations serve as powerful reminders that a “magic bullet” remains elusive. The future of TB treatment depends on developing new drugs but requires implementation of a precision medicine framework. The framework requires integration of three components which include rapid comprehensive drug susceptibility testing and vigilant adverse event monitoring and therapeutic drug monitoring to achieve optimal results for all patients. The implementation of TB elimination requires us to combine our development of new drugs with our establishment of solid and fair health systems.
References
[1] Caminero, José Antonio, et al. “Recommendations on the Diagnosis and Treatment of Tuberculosis Infection: SEPAR/SEIMC/Spanish Ministry of Health Consensus Statement.” Archivos De Bronconeumologia, Jan. 2026. PubMed.
[2] Fekadu, Ginénus, et al. “Impact of All-Oral Bedaquiline-Based Shorter Regimens in the Treatment of Drug-Resistant Tuberculosis: A Systematic Review and Meta- Analysis.” BMJ Global Health, vol. 10, no. 4, Apr. 2025, p. e018220. PubMed Central.
[3] Gopalaswamy, Radha, et al. “The Power of Resistance: Mechanisms of Antimicrobial Resistance in Mycobacterium Tuberculosis and Its Impact on Tuberculosis Management.” Clinical Microbiology Reviews, Jan. 2026, p. e0019425. PubMed.
[4] Horn-Magar, Carola, et al. “[Positive Interferon-Gamma Release Assay and Immuno- suppression: Guideline-Based Diagnosis and Treatment of Tuberculosis Infection].” Innere Medizin (Heidelberg, Germany), vol. 67, no. 2, Feb. 2026, pp. 181-93. PubMed.
[5] Lopes, Sofia R., et al. “Update in Tuberculosis Treatment: A Scoping Review of Current Practices.” Breathe (Sheffield, England), Mar. 2025. PubMed.
[6] “New Clinical Trial Shows Promise for Shorter TB Treatment.” News-Medical, 19 Nov. 2025.
[7] Saukkonen, Jussi J., et al. “Updates on the Treatment of Drug-Susceptible and Drug- Resistant Tuberculosis: An Official ATS/CDC/ERS/IDSA Clinical Practice Guide- line.” American Journal of Respiratory and Critical Care Medicine, vol. 211, no. 1, Jan. 2025, pp. 15-33. PubMed.“Shorter All-Oral TB Regimens Boost Success in Drug-Resistant Cases.” EMJ Re- views, 26 Apr. 2025.
[8] TB Trials Consortium. “Phase 2C Clinical Trial of Novel Short-Course Regimens for the Treatment of Pulmonary Tuberculosis: TBTC Study 38/CRUSH-TB Design.” Contemporary Clinical Trials, vol. 158, Sept. 2025, p. 108075. PubMed Central.
[9] Zhao, Chun-Yan, et al. “A New Era in Tuberculosis Prevention and Treatment: Breakthroughs in Drug Development and Future Prospects.” Infectious Diseases and Therapy, Oct. 2025.
