The Silent Struggle to Swallow: A Deep Dive into Achalasia
1. Dr. Kayypov Abdumazhit
2. Jaiprem Beenakumari Arshia
Anitha Devika
Kanakan Kavya
(1. Teacher, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.
2. Students, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.)
Abstract: Achalasia is a rare, primary esophageal motility disorder characterized by the selective degeneration of inhibitory neurons in the myenteric plexus, leading to failed relaxation of the lower esophageal sphincter (LES) and esophageal aperistalsis. This report examines the evolution of achalasia from its first historical descriptions to modern diagnostic frameworks, notably the Chicago Classification version 4.0. We evaluate primary therapeutic interventions, including Laparoscopic Heller Myotomy (LHM) and Per-oral Endoscopic Myotomy (POEM), the latter of which has demonstrated a long-term clinical success rate of 87.1 percent over a median follow-up of 72 months. Furthermore, the report explores the significant psychosocial burden of the disease, including high rates of anxiety and esophageal hypervigilance. Finally, we highlight recent scientific breakthroughs, such as the 2025 genome-wide association study (GWAS) that identified HLA-DQB1 risk variants, and the 2026 precision medicine trials aimed at tailoring myotomy length to individual esophageal anatomy.
Keywords: Achalasia; Dysphagia; Per-oral Endoscopic Myotomy (POEM); High-Resolution Manometry; Chicago Classification; Esophageal Motility; Regenerative Medicine; Autoimmunity.
Achalasia represents a complex, primary esophageal motor disorder of the smooth muscle, characterized fundamentally by the failure of the lower esophageal sphincter to relax in response to deglutition and the complete absence of organized peristalsis within the esophageal body.1 This condition arises from the progressive and selective degeneration of the myenteric (Auerbach’s) plexus, which serves as the primary neural coordinator for esophageal motility.4 Although first described over three centuries ago, achalasia remains a subject of intense clinical and scientific investigation, as researchers strive to unravel the precise autoimmune, viral, and genetic triggers that initiate this neurodegenerative cascade.4
Historical Context and Scientific Evolution
The medical history of achalasia began in 1674, when Sir Thomas Willis described a patient suffering from a significant "food blockage" in the esophagus.4 Willis’s primitive intervention—a whalebone with a sponge attached to manually push food into the stomach—marked the earliest recorded attempt at esophageal dilation.4 For much of the nineteenth century, the condition was referred to as "cardiospasm," reflecting a prevailing belief that the primary issue was an active, spasmodic contraction of the cardia.5
The transition to a neurogenic understanding began in 1914, when early observers suggested that the failure of the sphincter to relax might result from organic changes within the esophageal nerve plexus.4 In 1929, Sir Arthur Hurst coined the term "achalasia," deriving it from Greek roots meaning "failure of relaxation".4 This terminological shift mirrored an evolving understanding that the disease was not a positive spasm but a negative failure of the physiological inhibitory signaling required for normal bolus transit.4 Modern advancements, particularly the advent of high-resolution manometry (HRM) and the Chicago Classification, have further refined this understanding by allowing clinicians to categorize the disease into distinct phenotypes based on pressure topography.6
Neurobiological and Pathophysiological Foundations
The pathophysiology of achalasia is rooted in a fundamental imbalance between excitatory and inhibitory neurotransmission within the enteric nervous system (ENS).1 In a healthy physiological state, swallowing triggers a wave of contraction mediated by excitatory neurotransmitters such as acetylcholine and substance P.1 Simultaneously, a wave of "deglutitive inhibition" precedes the contraction, mediated by inhibitory neurons that release nitric oxide (NO) and vasoactive intestinal peptide (VIP).1 This inhibitory wave is essential for ensuring that the lower esophageal sphincter (LES) relaxes ahead of the bolus and that the contractions of the esophageal body are sequenced in a progressive, aboral direction.2
The Role of Inhibitory Ganglion Cell Loss
Achalasia is characterized by the selective destruction of these inhibitory, non-adrenergic, non-cholinergic (NANC) ganglion cells within the myenteric plexus.1 In the initial stages of the disease, the loss of nitric oxide-producing neurons leads to an unopposed action of excitatory neurotransmitters.4 This neurochemical shift results in simultaneous, high-amplitude, non-peristaltic contractions, a stage historically termed "vigorous achalasia".4 As the neurodegeneration progresses and involves the excitatory (cholinergic) neurons, the esophageal body becomes atonic and dilated, leading to the classic presentation of the disease.1
The degree of ganglion cell loss often parallels the duration and severity of the clinical symptoms.12 In advanced cases, the myenteric plexus is replaced by collagen and fibrous tissue, resulting in an irreversible failure of both the propulsive mechanism of the esophagus and the gateway function of the LES.1

Etiological Theories: Autoimmunity, Infection, and Genetics
While the downstream neurodegeneration is well-documented, the upstream initiating events remain a subject of active research. Current evidence suggests that achalasia is a multifactorial disorder occurring in genetically predisposed individuals when an environmental trigger, likely a viral infection, initiates an aberrant autoimmune response.2
The Autoimmune Hypothesis
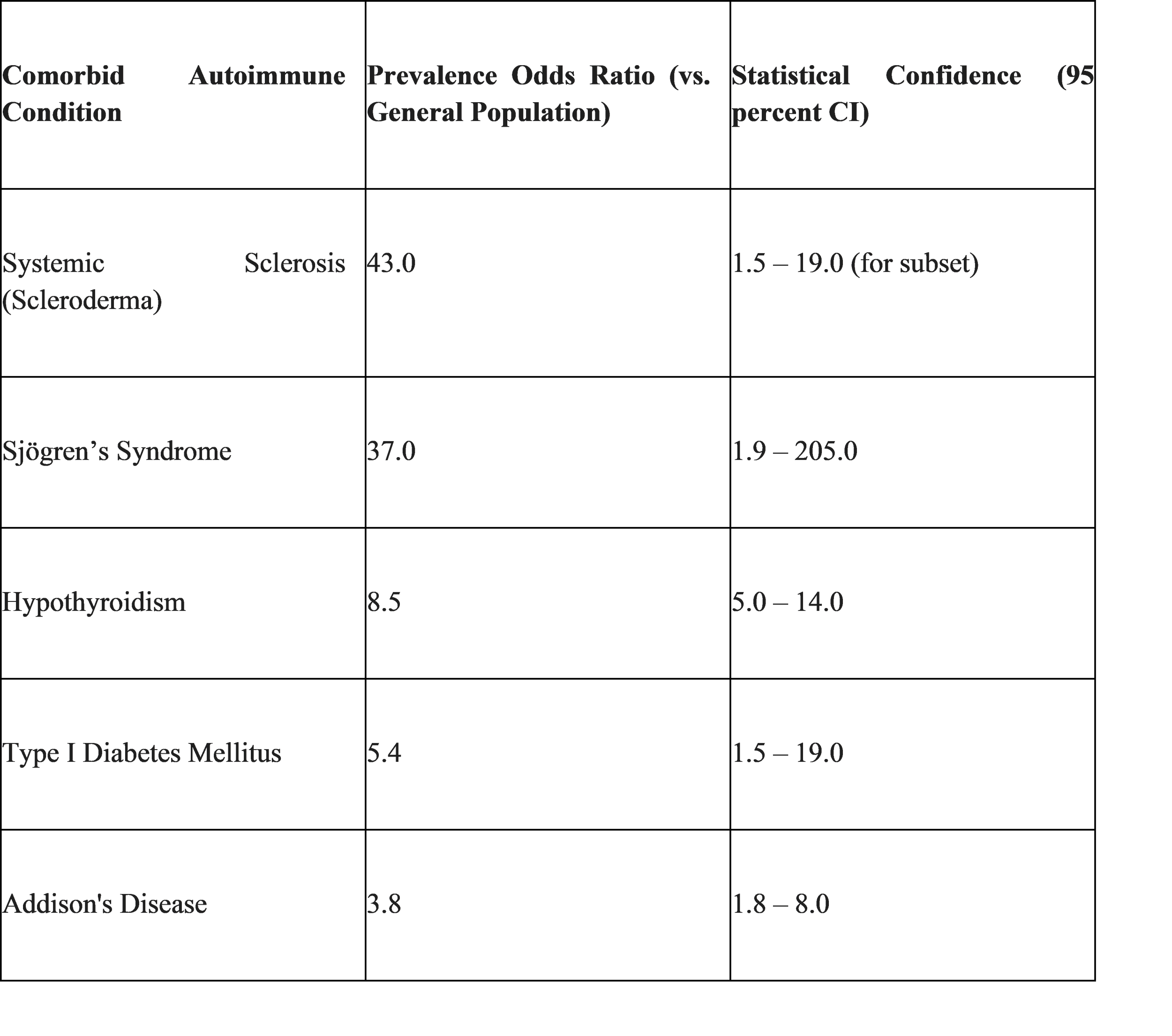
The association of achalasia with certain human leukocyte antigen (HLA) classes and the frequent presence of circulating anti-myenteric plexus autoantibodies suggest a robust autoimmune component.7 Studies have demonstrated that achalasia patients have a significantly higher prevalence of comorbid autoimmune diseases compared to the general population.16 For example, the likelihood of an achalasia patient having systemic sclerosis is 43 times higher than in the general population, while hypothyroidism is 8.5 times more common.17 This clustering of autoimmune conditions indicates a shared immunological susceptibility.16
The Viral Trigger: HSV-1 and Molecular Mimicry
The "molecular mimicry" theory posits that a viral infection triggers the immune system to attack esophageal neurons that share a similar antigenic structure with the virus.2 Herpes simplex virus type 1 (HSV-1) is a primary candidate due to its neurotropic nature and its ability to establish latency within the ganglion cells of the nervous system.18 Some studies have identified HSV-1 DNA in the esophageal muscle samples of achalasia patients, although the prevalence varies significantly by region, with some Iranian cohorts showing positivity in only 5 percent of cases.18 Other viral infections, such as Varicella zoster (VZV) and Human Papillomavirus (HPV), have also been statistically linked to increased odds of developing achalasia.15
Genetic Factors
Most cases of achalasia are sporadic, yet recent advancements have identified specific genetic markers. In 2025, the first genome-wide association study (GWAS) for idiopathic achalasia was published, identifying a significant risk variant in the HLA-DQB1 gene characterized by an 8-amino acid insertion. This suggests that genetic variation in class II HLA genes plays a pivotal role in the immune-mediated destruction of myenteric neurons. Historically, achalasia has also been associated with Allgrove syndrome (Triple A syndrome), an autosomal recessive disorder characterized by achalasia, alacrima, and adrenal insufficiency.14
Clinical Manifestations and the Symptom Burden
Achalasia typically presents in a bimodal age distribution, with peaks in the 20–40 and 60–70 age groups, although it can occur at any stage of life.8 The global incidence is estimated at approximately 1 in 100,000 people annually, with a prevalence of 9 to 10 in 100,000.3
The Cardinal Symptom: Progressive Dysphagia
Dysphagia, or the subjective sensation of difficulty swallowing, is the most common presenting symptom, reported by 82 percent to 100 percent of patients.22 In achalasia, dysphagia is typically progressive and involves both solids and liquids from the onset.21 This is a critical diagnostic differentiator: structural obstructions, such as malignancy or strictures, usually present first with solid-food dysphagia, only progressing to liquids as the lumen significantly narrows.13 Achalasia patients often describe the feeling of food getting "stuck" in the mid-to-lower chest, and many adopt compensatory behaviors, such as drinking large amounts of water, standing up, or arching their back to help clear the esophagus through gravitational force.9
Regurgitation and Nocturnal Aspiration
Regurgitation of undigested food and saliva is reported by up to 91 percent of patients.22 Unlike the acidic reflux seen in gastroesophageal reflux disease (GERD), the material regurgitated in achalasia is typically alkaline or neutral, consisting of food that has been stagnant in the esophagus.8 This regurgitation often occurs at night, leading to a nocturnal cough or choking fits that can wake the patient.25 If severe, it can lead to aspiration pneumonia, lung abscesses, or bronchiectasis.9
Chest Pain and Heartburn: The Risk of Misdiagnosis
Retrosternal chest pain is common, especially in younger patients and those with Type III (spastic) achalasia.6 This pain is often severe and can mimic cardiac angina.9 Furthermore, 27 percent to 42 percent of achalasia patients report heartburn, which frequently leads to a misdiagnosis of GERD.7 In achalasia, heartburn is not caused by acid reflux from the stomach but rather by the fermentation of stagnant food in the esophagus or the production of lactic acid by resident bacteria.8 This misdiagnosis often delays the correct diagnosis by several years, during which time the esophagus may undergo significant, irreversible dilation.13

Diagnostic Evaluation: A Multi-Modality Approach
Because the symptoms of achalasia overlap with more common conditions, a precise diagnosis requires a step-by-step evaluation using complementary diagnostic tests.21
Upper Endoscopy (Esophagogastroduodenoscopy)
Endoscopy is often the first test performed in patients with dysphagia to exclude mechanical obstruction or malignancy.1 However, endoscopy is not a sensitive diagnostic tool for achalasia, with up to 40 percent to 70 percent of patients showing normal findings in the early stages.21 In advanced cases, the endoscopist may observe a dilated, atonic esophagus with retained food and saliva, even after the required fasting period.1 A key diagnostic clue is the "pop" or firm resistance felt as the endoscope passes through the tightly closed LES into the stomach.1
Barium Esophagogram and Timed Barium Esophagogram (TBE)
The barium swallow provides essential information about the anatomy and clearance function of the esophagus.29 The classic radiologic finding is the "bird's beak" appearance, characterized by a smooth, tapered narrowing of the distal esophagus at the level of the LES.1 Other findings include proximal esophageal dilation, aperistalsis, and the absence of a gastric air bubble.1
The Timed Barium Esophagogram (TBE) is a specialized technique used to quantify the rate of esophageal emptying.1 The patient drinks a standard volume of barium (100–250 mL), and spot films are taken at 1, 2, and 5 minutes.31 In healthy individuals, the esophagus clears the barium completely within 1 minute.31 In achalasia, a column of barium persists beyond 5 minutes, and the height of this column is a reliable predictor of treatment success or failure.1

High-Resolution Manometry (HRM): The Diagnostic Gold Standard
HRM is the most sensitive and specific test for diagnosing achalasia.6 Unlike traditional manometry, which used only a few pressure sensors, HRM uses catheters with up to 36 sensors spaced 1 cm apart, providing a continuous topographical representation of esophageal pressure.6
The diagnosis of achalasia on HRM is based on two primary findings:
1. Elevated Integrated Relaxation Pressure (IRP): This metric represents the average minimum pressure of the esophagogastric junction (EGJ) during a 4-second window following a swallow.6 An elevated IRP indicates that the LES fails to relax adequately.6
2. Aperistalsis: The complete absence of normal, progressive peristaltic waves in the esophageal body.6
The Chicago Classification version 4.0 (CCv4.0)
The Chicago Classification is the global standard for interpreting HRM data.10 Version 4.0, finalized by an international working group, refined the diagnostic criteria to improve accuracy and reduce inconclusive results.11
Core Metrics of CCv4.0
● Integrated Relaxation Pressure (IRP): The primary threshold for failed relaxation. The threshold varies based on the type of manometry catheter used and the patient's position during the test.34
● Distal Contractile Integral (DCI): A measure of the vigor of the esophageal contraction, calculated as the amplitude x duration x length of the contraction.36
● Distal Latency (DL): The time interval between the start of the swallow and the contractile deceleration point in the distal esophagus.34 A DL of less than 4.5 seconds indicates a premature or spastic contraction.36

Achalasia Subtypes in CCv4.0
The Chicago Classification categorizes achalasia into three types, which are critical for predicting treatment response.10

Therapeutic Management: Strategies for LES Disruption
Since the underlying neurodegeneration in achalasia is currently irreversible, the goal of all treatments is palliative: to reduce the resistance of the non-relaxing LES.1
Laparoscopic Heller Myotomy (LHM)
LHM has long been considered the surgical gold standard.29 The procedure involves performing a longitudinal incision through the circular muscle fibers of the LES and extending it onto the proximal stomach to ensure complete disruption of the high-pressure zone.29 To prevent the high incidence of postoperative acid reflux, LHM is typically paired with a partial fundoplication, such as the Dor (anterior) or Toupet (posterior) wrap.23 LHM offers high long-term success rates, especially for Type I and Type II achalasia.37
Per-oral Endoscopic Myotomy (POEM)
POEM is a revolutionary, minimally invasive endoscopic procedure introduced in 2008.8 Using an endoscope, the physician creates a submucosal tunnel in the esophageal wall, allowing them to reach the LES and perform a myotomy without external incisions.26 In 2025, meta-analyses confirmed that POEM maintains a clinical success rate of 87.1 percent over a median 72-month follow-up, though it is associated with a 24.2 percent incidence of endoscopically detectable reflux esophagitis.
Looking toward 2026, the focus has shifted to "Precision Medicine in Achalasia." In April 2026, a major multicenter randomized clinical trial (PREMEDIA) commenced to evaluate whether a "short, tailored POEM" (cutting approximately 4 cm of muscle) is non-inferior to the standard 9 cm POEM while reducing long-term complications like "blown-out myotomy" and acid reflux.
Pneumatic Dilation (PD)
PD involves the endoscopic placement of a large-diameter balloon (30 mm, 35 mm, or 40 mm) across the LES.29 The balloon is inflated to forcefully stretch and tear the muscle fibers.28 While initially effective, nearly one-third of patients require repeat procedures within five years.8 PD is associated with an esophageal perforation risk of approximately 1.3 percent to 1.5 percent.8
Botulinum Toxin (Botox) Injection
Botox inhibits the release of acetylcholine from excitatory nerves, relaxing the LES.14 While highly effective in the short term, the effect typically lasts only 6 to 12 months.29 Botox is generally reserved for elderly patients or those with multiple comorbidities who are poor candidates for definitive surgery or dilation..43

Comparative Outcomes and Long-term Readmissions
A comprehensive US national study analyzing 16,610 index achalasia admissions between 2016 and 2020 revealed significant differences in 30-day readmission rates.3 Patients who underwent pneumatic dilation had a readmission rate of 12.34 percent, which was significantly higher than those who underwent POEM (4.28 percent) or LHM (3.63 percent).3

The Psychosocial Dimension of Achalasia
Achalasia is a chronic, life-altering condition that extends far beyond the physical act of swallowing.46 A major study found that female patients were up to 7.87 times more likely to experience depression and 3.1 times more likely to experience anxiety compared to age-matched controls.46
Esophageal Hypervigilance
A critical psychological factor is "esophageal hypervigilance"—a heightened, anxious focus on the sensations in the esophagus.. In a 2025 cohort study, researchers found that baseline hypervigilance and anxiety scores are better predictors of post-treatment quality of life than physiological success alone. Patients with high hypervigilance but better physiological function at baseline often report lower quality of life after treatment, as their ongoing worry continues to amplify minor sensations.
Nutritional Therapy and Lifestyle Adjustments
Nutritional therapy focuses on food texture modification and lubricant techniques.26
● Soft Textured Diet: Pureed foods, smoothies, and protein shakes are essential when solid intake is low.52
● Lubrication: Drinking warm or room-temperature liquids during meals can ease transit via gravity.52 Carbonated beverages may also help clear food blockages.53
● Foods to Avoid: Fibrous vegetables, tough meats, and foods that congeal (like white bread or fluffy rice) should be minimized.26
The Horizon: Regenerative Medicine and Immunotherapy
Research is shifting from palliative LES disruption toward restorative and anti-inflammatory therapies.7
Stem Cell Therapy and Tissue Engineering
The most promising future therapy involves the transplantation of enteric neural stem cells (ENSCs) to replace lost inhibitory neurons.19 In 2025, clinical research expanded into using patient-derived induced pluripotent stem cells (iPSCs) programmed into nNOS-specific precursors. Furthermore, bio-3D printing of esophagus-like tubular structures is being explored to restore structural integrity in end-stage "megaesophagus".
Immunotherapy
Evidence suggests that a subset of achalasia cases may be driven by Type 2 (allergic) inflammation.39 Clinical trials in 2025-2026 have investigated the use of systemic corticosteroids (e.g., prednisone) for patients with early-stage variants like EGJ outflow obstruction, showing significant reductions in IRP and improvement in symptoms in allergic-type patients.39
Conclusion
From the historical whalebone dilators of 1674 to the precision medicine POEM trials of 2026, the medical community has made significant strides in managing achalasia. While POEM and LHM remain highly effective palliative tools, the identification of genetic risk markers in 2025 and the ongoing exploration of stem cell restorative therapies mark a new era aimed at curative intervention. Addressing the "silent struggle" now requires a holistic approach that integrates high-resolution diagnostics with long-term psychological and nutritional support.
References
1. Achalasia - StatPearls - NCBI Bookshelf, accessed March 22, 2026, https://www.ncbi.nlm.nih.gov/books/NBK519515/
2. Pathophysiology of achalasia - Rogers - Annals of Esophagus - AME Publishing, accessed March 22, 2026, https://aoe.amegroups.org/article/view/5440/html
3. Comparison of peroral endoscopic myotomy, laparoscopic Heller myotomy, and pneumatic dilation for patients with achalasia: a United States national experience - Clinical Endoscopy, accessed March 22, 2026, https://www.e-ce.org/journal/view.php?number=7944
4. Pathogenesis of achalasia cardia - PMC - NIH, accessed March 22, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC3386318/
5. The Pathogenesis and Management of Achalasia: Current Status and Future Directions - PMC, accessed March 22, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC4477988/
6. Impact of high-resolution manometry on achalasia diagnosis and treatment - PMC, accessed March 22, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC4290001/
7. New insights into the pathophysiology of achalasia and implications ..., accessed March 22, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC5028805/
8. Achalasia: treatment, current status and future advances - PMC, accessed March 22, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC6823561/
9. Achalasia (Cardiospasm): Symptoms, Causes, Types, Treatments - Cleveland Clinic, accessed March 22, 2026, https://my.clevelandclinic.org/health/diseases/17534-achalasia
10. Achalasia: Chicago Classification - Endoscopy Campus, accessed March 22, 2026, https://www.endoscopy-campus.com/en/classifications/achalasia-chicago-classification/
11. Esophageal Motility Disorder: Chicago Classification | BSG, accessed March 22, 2026, https://www.bsg.org.uk/clinical-resource/esophageal-motility-disorders
12. Esophageal Motility Disorders - StatPearls - NCBI Bookshelf - NIH, accessed March 22, 2026, https://www.ncbi.nlm.nih.gov/books/NBK562155/
13. Esophageal Motility Disorders - AAFP, accessed March 22, 2026, https://www.aafp.org/pubs/afp/issues/2020/0901/p291.html
14. Pathophysiology of achalasia - PubMed, accessed March 22, 2026, https://pubmed.ncbi.nlm.nih.gov/10980949/
15. Autoimmune and viral risk factors are associated with achalasia: a case-control study - PMC, accessed March 22, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC9232907/
16. Achalasia and Thyroid Disorders: A Hidden Autoimmune Overlap? Epidemiology, Mechanisms, and Clinical Relevance of an Emerging Association - MDPI, accessed March 22, 2026, https://www.mdpi.com/2624-5647/7/4/64
17. The prevalence of autoimmune disease in patients with esophageal achalasia - ICES, accessed March 22, 2026, https://www.ices.on.ca/publications/journal-articles/the-prevalence-of-autoimmune-disease-in-patients-with-esophageal-achalasia/
18. (PDF) Assessing the prevalence of herpes simplex virus (HSV) in the esophageal muscles of achalasia patients undergoing Heller myotomy - ResearchGate, accessed March 22, 2026, https://www.researchgate.net/publication/393916204_Assessing_the_prevalence_of_herpes_simplex_virus_HSV_in_the_esophageal_muscles_of_achalasia_patients_undergoing_Heller_myotomy
19. Stem Cell Treatment for Achalasia?, accessed March 22, 2026, https://www.achalasia-action.org/wp-content/uploads/2019.01.22-Stem-Cell-Research-Conor-McCann.pdf
20. Achalasia - Symptoms and causes - Mayo Clinic, accessed March 22, 2026, https://www.mayoclinic.org/diseases-conditions/achalasia/symptoms-causes/syc-20352850
21. Esophageal Achalasia: Diagnostic Evaluation - PMC, accessed March 22, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC9174137/
22. An Overview of Achalasia and Its Subtypes - PMC, accessed March 22, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC5572971/
23. ACG Clinical Guidelines: Diagnosis and Management of Achalasia ..., accessed March 22, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC9896940/
24. Achalasia subtypes are front and center of the Chicago classification—strategies to overcome limitations in clinical applicati - AME Publishing Company, accessed March 22, 2026, https://cdn.amegroups.cn/journals/jrh/files/journals/33/articles/5407/public/5407-PB11-2549-R3.pdf?filename=aoe-03-24.pdf&t=1731243086
25. Achalasia - NHS, accessed March 22, 2026, https://www.nhs.uk/conditions/achalasia/
26. Eating Well with Acalasia, accessed March 22, 2026, https://aegendo.com.au/wp-content/uploads/2023/02/eating_well_with_achalasia.pdf
27. Achalasia | Cedars-Sinai, accessed March 22, 2026, https://www.cedars-sinai.org/health-library/diseases-and-conditions/a/achalasia.html
28. Achalasia Diagnosis and Evaluation - Liv Hospital, accessed March 22, 2026, https://int.livhospital.com/gastroenterology/achalasia/diagnosis-and-evaluation/
29. Achalasia - Diagnosis and treatment - Mayo Clinic, accessed March 22, 2026, https://www.mayoclinic.org/diseases-conditions/achalasia/diagnosis-treatment/drc-20352851
30. How to Perform and Interpret Timed Barium Esophagogram - PMC - NIH, accessed March 22, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC3644662/
31. Timed barium swallow in the assessment of esophageal emptying in patients with achalasia: How to do it and what to look for - EPOS™ - ESR, accessed March 22, 2026, https://epos.myesr.org/poster/esr/ecr2018/C-1891/background
32. The Efficacy of Peroral Endoscopic Myotomy vs Pneumatic Dilation as Treatment for Patients With Achalasia Suffering From Persistent or Recurrent Symptoms After Laparoscopic Heller Myotomy: A Randomized Clinical Trial - PubMed, accessed March 22, 2026, https://pubmed.ncbi.nlm.nih.gov/36907524/
33. Chicago classification version 4.0© technical review: Update on standard high-resolution manometry protocol for the assessment of esophageal motility - CEFIDI, accessed March 22, 2026, https://www.cefidi.com.br/wp-content/uploads/2022/10/Chicago-classification-version-4.0-technical-review-update-2021.pdf
34. ESOPHAGEAL MOTILITY DISORDERS ON HIGH RESOLUTION MANOMETRY: CHICAGO CLASSIFICATION VERSION 4.0©, accessed March 22, 2026, https://www.sth.nhs.uk/clientfiles/File/Chicago%20classification%20for%20classifying%20oesopheagl%20motility%20disorders.pdf
35. Chicago classification version 4.0© technical review: Update on standard high-resolution manometry protocol for the assessment of esophageal motility - PMC, accessed March 22, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC8268048/
36. Chicago Classification ver. 4.0: An Overview of Esophageal Motility Disorders on High-resolution Manometry - PMC, accessed March 22, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC12286510/
37. Understanding the Chicago Classification: From Tracings to Patients - Journal of Neurogastroenterology and Motility, accessed March 22, 2026, https://www.jnmjournal.org/journal/view.html?doi=10.5056/jnm17026
38. Comparison of outcomes of laparoscopic Heller myotomy versus per-oral endoscopic myotomy for management of achalasia - PMC, accessed March 22, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC5595380/
39. Corticosteroid Effect on Achalasia Variant EGJOO | Clinical Research Trial Listing, accessed March 22, 2026, https://www.centerwatch.com/clinical-trials/listings/NCT06588348/corticosteroid-effect-on-achalasia-variant-egjoo
40. Timed barium esophagography to predict recurrent achalasia after peroral endoscopic myotomy: a retrospective study in Thailand - PMC, accessed March 22, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC11474463/
41. Current therapies for achalasia: comparison and efficacy - PubMed, accessed March 22, 2026, https://pubmed.ncbi.nlm.nih.gov/9706766/
42. Treatment of achalasia: the short-term response to botulinum toxin injection seems to be independent of any kind of pretreatment - PMC, accessed March 22, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC122056/
43. Role of botulinum toxin injection in treatment of achalasia - Heddle - Annals of Esophagus, accessed March 22, 2026, https://aoe.amegroups.org/article/view/5438/html
44. Effect of Peroral Endoscopic Myotomy vs Pneumatic Dilation on Symptom Severity and Treatment Outcomes Among Treatment-Naive Patients With Achalasia: A Randomized Clinical Trial - PMC, accessed March 22, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC6618792/
45. Comparison of peroral endoscopic myotomy, laparoscopic Heller myotomy, and pneumatic dilation for patients with achalasia: a United States national experience - ResearchGate, accessed March 22, 2026, https://www.researchgate.net/publication/387367505_Comparison_of_peroral_endoscopic_myotomy_laparoscopic_Heller_myotomy_and_pneumatic_dilation_for_patients_with_achalasia_a_United_States_national_experience
46. Integrating psychological assessment in achalasia management ..., accessed March 22, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC11487623/
47. Anxiety and Hypervigilance Predict Poor Outcomes in Achalasia - Northwestern Medicine Breakthroughs for Physicians, accessed March 22, 2026, https://breakthroughsforphysicians.nm.org/gastroenterology-research-article-hypervigilance-treatment-outcomes-in-esophageal-disorder.html
48. Psychological burden of achalasia: Patients' screening rates of depression and anxiety and sex differences - PMC, accessed March 22, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC10174570/
49. Esophageal tissue engineering: Current status and perspectives - ResearchGate, accessed March 22, 2026, https://www.researchgate.net/publication/288823502_Esophageal_tissue_engineering_Current_status_and_perspectives
50. Stem cell therapy for the treatment of achalasia and gastroparesis - Research - The University of Melbourne, accessed March 22, 2026, https://research.unimelb.edu.au/strengths/initiatives/international-training-groups/ku-leuven-melbourne-joint-phd/joint-phd-opportunities/stem-cell-therapy-for-the-treatment-of-achalasia-and-gastroparesis
51. Botox Injections for Esophageal Motor Disorders - BINASSS, accessed March 22, 2026, https://www.binasss.sa.cr/jul25/38.pdf
52. Achalasia: nutrition therapy - AGA GI Patient Center, accessed March 22, 2026, https://patient.gastro.org/achalasia-nutrition-therapy/
53. Living With Achalasia, accessed March 22, 2026, https://www.achalasia-action.org/living-with-achalasia/
54. Living With - Achalasia Action, accessed March 22, 2026, https://www.achalasia-action.org/wp-content/uploads/living-with-achalasia.pdf
55. A Pilot Study of Corticosteroid Treatment for Achalasia-Variant Esophagogastric Junction Outflow Obstruction - PMC, accessed March 22, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC12964023/
Achalasia with dense eosinophilic infiltrate responds to steroid therapy - PubMed, accessed March 22, 2026, https://pubmed.ncbi.nlm.nih.gov/21839711/
