Amenorrhea
1. Aidarbek kyzy Aidanek
2. Shah Aalam
3. Sababa Sabnam
(1. Teacher, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.
2. Student, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.
3. Student, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.)
Abstract
Amenorrhea, defined as the transient or permanent absence of menstruation, serves as a critical biological indicator of dysfunction within the hypothalamic-pituitary-ovarian (HPO) axis or the anatomical integrity of the reproductive outflow tract. This report provides an exhaustive analysis of the condition, categorized into primary and secondary etiologies, while addressing the limitations of such binary classifications in clinical practice. Drawing upon verified textbook citations from Williams Gynecology, Speroff’s Clinical Gynecologic Endocrinology and Infertility, and Berek & Novak’s Gynecology, alongside recent clinical practice guidelines from the American Society for Reproductive Medicine (ASRM), the Endocrine Society, and the American College of Obstetricians and Gynecologists (ACOG), this document synthesizes the current state of reproductive endocrinology.
Key areas of focus include the molecular mechanisms of Functional Hypothalamic Amenorrhea (FHA) involving leptin and kisspeptin dysregulation; the paradigm shift in Polycystic Ovary Syndrome (PCOS) diagnosis following the 2023 International Evidence-Based Guidelines, specifically regarding Anti-Müllerian Hormone (AMH) and ultrasound thresholds; the nuanced management of hyperprolactinemia including the "hook effect" and macroprolactinemia; and the genetic and autoimmune underpinnings of Primary Ovarian Insufficiency (POI). Furthermore, the report critically evaluates diagnostic modalities, advocating for the retirement of the traditional progestin challenge test in favor of direct hormonal assessment and advanced imaging protocols, including dynamic contrast-enhanced MRI for pituitary lesions. Therapeutic strategies are reviewed with a focus on long-term health maintenance, particularly skeletal integrity mediated by the RANKL/OPG pathway and cardiovascular protection.
Keywords: Amenorrhea, Hypothalamic-Pituitary-Ovarian Axis, Polycystic Ovary Syndrome (PCOS), Primary Ovarian Insufficiency (POI), Functional Hypothalamic Amenorrhea (FHA), Hyperprolactinemia, Turner Syndrome, Müllerian Agenesis, Progestin Challenge Test, Relative Energy Deficiency in Sport (RED-S).
1. Introduction and Epidemiological Context
The menstrual cycle is a complex, rhythmic physiological process that requires the precise orchestration of neuroendocrine events. Its cessation, amenorrhea, is not a disease entity itself but rather a sign of underlying pathology ranging from benign functional adaptations to life-threatening neoplasms. In the field of Obstetrics and Gynecology, the evaluation of amenorrhea represents a fundamental competency, yet it remains one of the most intellectually demanding areas of practice due to the breadth of the differential diagnosis.
The prevalence of pathological amenorrhea—excluding physiological causes such as pregnancy, lactation, and menopause—is estimated to be approximately 3% to 4% of the reproductive-age female population.1 While seemingly low, this figure represents millions of women worldwide who are at risk for the sequelae of hypogonadism, including infertility, osteoporosis, and cardiovascular disease. The distribution of etiologies varies by population but is generally dominated by four main categories: hypothalamic dysfunction (35%), polycystic ovary syndrome (30%), pituitary disease (18%), and primary ovarian insufficiency (12%), with anatomical defects comprising the remainder.1
1.1 Verified Textbook Foundations
The foundational knowledge for this report is anchored in the three major texts of the discipline. Williams Gynecology (Hoffman et al.) provides the anatomical and surgical basis for outflow tract disorders.3 Speroff’s Clinical Gynecologic Endocrinology and Infertility (Taylor et al.) is the standard reference for the physiology of the HPO axis and the nuances of hormonal feedback loops.5 Berek & Novak’s Gynecology (Berek) offers comprehensive oncological and general gynecologic perspectives on menstrual disorders.7 These texts collectively emphasize that amenorrhea is a "vital sign" of female health, reflecting the integration of metabolic, genetic, and psychological well-being.
1.2 Clinical Definitions and Classifications
The classification of amenorrhea has historically been divided into primary and secondary types based on the timing of symptom onset relative to menarche. While this distinction aids in the initial organization of the differential diagnosis, it is essential to recognize that many etiologies, such as hyperprolactinemia or gonadal dysgenesis, can present as either primary or secondary amenorrhea depending on the severity of the defect and the time of presentation.
Primary Amenorrhea is defined by the American Society for Reproductive Medicine (ASRM) and the American College of Obstetricians and Gynecologists (ACOG) as the absence of menses by age 15 years in the presence of normal growth and secondary sexual characteristics. However, clinical evaluation is indicated earlier, at age 13 years, if there is a complete absence of secondary sexual characteristics, specifically thelarche (breast development).1 This distinction is critical because breast development serves as a bioassay for endogenous estrogen production. Its absence implies a fundamental failure of the ovary to produce estrogen or a failure of the pituitary/hypothalamus to drive the ovary.
Secondary Amenorrhea is defined as the cessation of menses for more than three months in women who previously had regular menstrual cycles, or for more than six months in women with a history of irregular cycles (oligomenorrhea).1 This definition accounts for the inherent variability in cycle length among women with ovulatory dysfunction. The prevalence of secondary amenorrhea is significantly higher than that of primary amenorrhea, and it is most frequently caused by functional hypothalamic disturbances and PCOS.
2. Physiology of the Hypothalamic-Pituitary-Ovarian (HPO) Axis
To understand the pathology of amenorrhea, one must first possess a granular understanding of the normal physiology of the HPO axis. This system functions as a closed-loop feedback mechanism involving the central nervous system, the pituitary gland, and the ovaries.
2.1 The Hypothalamic Pulse Generator
The arcuate nucleus of the hypothalamus contains neurons that secrete Gonadotropin-Releasing Hormone (GnRH). GnRH is a decapeptide that is not released continuously but rather in discrete pulses. This pulsatility is the sine qua non of reproductive function. Continuous exposure to GnRH, as seen with the administration of long-acting GnRH agonists (e.g., leuprolide), results in the downregulation of GnRH receptors on the pituitary gonadotrophs and a subsequent cessation of gonadotropin secretion.11
The frequency and amplitude of GnRH pulses determine the specific response of the pituitary. Rapid frequency pulses favor the secretion of Luteinizing Hormone (LH), while slower frequency pulses favor the secretion of Follicle-Stimulating Hormone (FSH). This frequency modulation is critical during the menstrual cycle: the follicular phase is characterized by faster pulses to drive LH, while the luteal phase, under the influence of progesterone, sees a slowing of the pulse generator.1
Recent research has elucidated the role of the KNDy neurons (Kisspeptin, Neurokinin B, and Dynorphin) in the regulation of GnRH. Kisspeptin is a potent stimulator of GnRH release, while neurokinin B and dynorphin act as fine-tuners of the pulse generator. Metabolic signals, particularly leptin (from adipose tissue) and insulin, interface with these neurons to signal energy availability. This connection explains the mechanism of Functional Hypothalamic Amenorrhea: in states of low energy availability, leptin levels fall, kisspeptin drive is reduced, and the GnRH pulse generator slows or stops, leading to anovulation and amenorrhea.11
2.2 The Anterior Pituitary and Gonadotropins
The anterior pituitary gland, located in the sella turcica, responds to GnRH by secreting the glycoprotein hormones FSH and LH. These hormones share a common alpha subunit (identical to TSH and hCG) but have distinct beta subunits that confer biological specificity.
● FSH: Targets the granulosa cells of the ovarian follicle, stimulating follicular growth and the expression of the enzyme aromatase (CYP19), which converts androgens to estrogens.
● LH: Targets the theca cells of the ovary to produce androgens (androstenedione and testosterone) and triggers the final maturation of the oocyte and ovulation. It also supports the corpus luteum.11
2.3 Ovarian Feedback Loops
The ovary responds to gonadotropins by producing sex steroids (estradiol, progesterone) and peptides (inhibin A, inhibin B).
● Negative Feedback: For the majority of the cycle, estradiol and inhibin B exert negative feedback on the hypothalamus and pituitary, suppressing FSH and LH. This prevents the recruitment of multiple dominant follicles.
● Positive Feedback: In a unique physiological paradox, once estradiol levels reach a critical threshold (sustained for >48 hours) during the late follicular phase, the feedback switches from negative to positive. This triggers the massive surge of LH that induces ovulation. In anovulatory forms of amenorrhea, such as PCOS, this surge never occurs due to the lack of a dominant follicle achieving the necessary estradiol threshold.1
2.4 The Genital Outflow Tract
For menstruation to be visible, the hormonal changes must act upon a functional end-organ: the uterus and endometrium. Estrogen causes proliferation of the endometrium (proliferative phase). Progesterone, produced after ovulation, converts this to a secretory endometrium. If pregnancy does not occur, the withdrawal of progesterone leads to enzymatic breakdown of the functional layer and menstrual shedding. Any obstruction in the outflow tract (cervix, vagina, hymen) or destruction of the endometrium (Asherman's syndrome) will result in amenorrhea despite a functioning HPO axis.2
3. Primary Amenorrhea: Genetic, Developmental, and Anatomic Etiologies
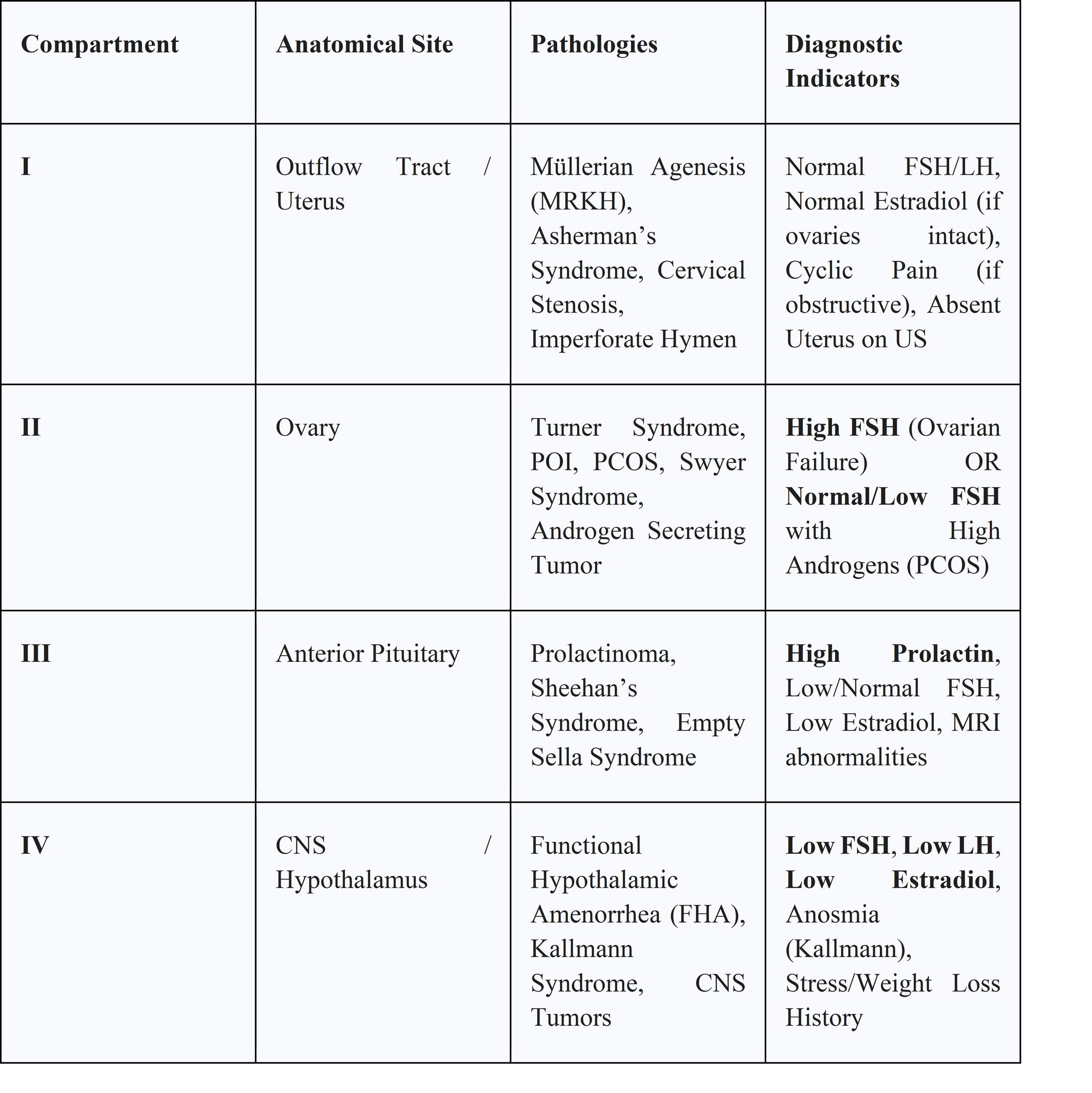
Primary amenorrhea is often the presenting symptom for significant genetic or congenital anomalies. The diagnostic approach is strictly compartmentalized based on the presence of breast development (estrogen action) and the presence of a uterus.
3.1 Hypergonadotropic Hypogonadism (Gonadal Dysgenesis)
The most common cause of primary amenorrhea is gonadal failure, characterized by elevated FSH levels (hypergonadotropic) due to the lack of ovarian negative feedback.
3.1.1 Turner Syndrome (45,X) and Mosaicism
Turner syndrome is caused by the complete or partial absence of one X chromosome. It affects approximately 1 in 2,500 live female births.
● Pathophysiology: The absence of genes on the X chromosome leads to accelerated atresia of ovarian follicles. By birth or early childhood, the ovaries have usually degenerated into fibrous streaks ("streak gonads") incapable of steroidogenesis.11
● Clinical Presentation: Patients present with short stature (due to loss of the SHOX gene), primary amenorrhea, and lack of secondary sexual characteristics. Somatic stigmata include a webbed neck (pterygium colli), shield chest, widely spaced nipples, and cubitus valgus.
● Medical Management:
○ Cardiac Screening: A transthoracic echocardiogram or cardiac MRI is mandatory to screen for coarctation of the aorta and bicuspid aortic valve, which are present in up to 30% of patients.
○ Renal Screening: Renal ultrasound to rule out horseshoe kidney or renal agenesis.
○ Growth Hormone: Initiated in childhood to optimize final adult height.
○ Pubertal Induction: Low-dose transdermal estradiol is initiated to promote breast development, followed by the addition of cyclic progestins after 12-24 months to induce menses. Delaying progestins allows for maximal breast development before the ductal system matures.10
3.1.2 Swyer Syndrome (46,XY Gonadal Dysgenesis)
Swyer syndrome represents a distinct form of gonadal dysgenesis where the individual has a male karyotype (46,XY) but develops as a phenotypic female.
● Mechanism: A mutation in the SRY gene (Sex-determining Region Y) prevents the primitive gonad from differentiating into a testis. Without testicular Sertoli cells, Anti-Müllerian Hormone (AMH) is not produced, so the Müllerian ducts (uterus, fallopian tubes) persist and develop. Without Leydig cells, testosterone is not produced, so the external genitalia do not virilize.
● Clinical Presentation: These patients appear as typical females with a uterus and vagina but have streak gonads. They present with primary amenorrhea and delayed puberty.
● Malignancy Risk: The presence of Y-chromosome material in a dysgenetic gonad poses a high risk (20-30%) for the development of gonadoblastoma and dysgerminoma. Therefore, prophylactic bilateral gonadectomy is indicated immediately upon diagnosis.2
3.2 Disorders of the Outflow Tract (Müllerian Anomalies)
In patients with primary amenorrhea who do have normal breast development (indicating functional ovaries and estrogen production), the obstruction or absence of the outflow tract must be considered.
3.2.1 Müllerian Agenesis (Mayer-Rokitansky-Küster-Hauser Syndrome - MRKH)
MRKH has an incidence of 1 in 4,500 females and is the second most common cause of primary amenorrhea.
● Anatomy: It is characterized by the congenital aplasia of the uterus and the upper two-thirds of the vagina. The ovaries are embryologically distinct (originating from the genital ridge) and are typically normal in structure and function.
● Clinical Presentation: Patients present with normal thelarche and pubarche (pubic hair) but primary amenorrhea. Sexual intercourse may be difficult or impossible due to a shortened vaginal dimple.
● Associations: Skeletal anomalies (scoliosis) and renal anomalies (unilateral renal agenesis, pelvic kidney) occur in 30-40% of cases, necessitating renal ultrasound and spinal imaging.2
● Management: Treatment focuses on creating a functional vagina. Non-surgical dilation (Frank or Ingram methods) is the first-line therapy with high success rates. Surgical vaginoplasty (e.g., McIndoe procedure, Davydov procedure) is reserved for failures of dilation. Psychological counseling is essential to address the impact on identity and fertility potential (surrogacy is an option as ovaries are functional).2
3.2.2 Complete Androgen Insensitivity Syndrome (CAIS)
CAIS must be differentiated from MRKH as both present with primary amenorrhea and an absent uterus.
● Pathophysiology: Patients have a 46,XY karyotype with functioning testes that produce testosterone and AMH. However, a mutation in the androgen receptor gene makes end-organs insensitive to androgens.
● Müllerian Regression: The produced AMH causes regression of the uterus and fallopian tubes (hence, no uterus).
● Feminization: The high levels of testosterone are peripherally aromatized to estrogen, leading to breast development.
● Clinical Distinction: Unlike MRKH, patients with CAIS typically have sparse or absent pubic and axillary hair (as pubic hair growth is androgen-dependent). Testosterone levels are in the male range.
● Management: Gonads (testes) are often located in the abdomen or inguinal canal. They should be removed to prevent malignancy (seminoma), though this can be delayed until after puberty to allow for spontaneous breast development via aromatization. Hormone replacement (estrogen) is required post-gonadectomy.2
3.2.3 Imperforate Hymen and Transverse Vaginal Septum
These are obstructive lesions of the distal outflow tract.
● Pathophysiology: Failure of canalization of the vaginal plate or failure of the urogenital sinus to perforate.
● Presentation: Patients often experience cyclic pelvic pain (molimina) without visible bleeding (cryptomenorrhea). Menstrual blood accumulates in the vagina (hematocolpos) and uterus (hematometra), which can lead to retrograde menstruation and endometriosis.
● Diagnosis: Physical exam reveals a bulging, bluish membrane at the introitus (imperforate hymen) or a blind vaginal pouch (transverse septum). MRI is useful to delineate the thickness of the septum.2
4. Secondary Amenorrhea: Functional, Endocrine, and Metabolic Disorders
Secondary amenorrhea is statistically more common than primary amenorrhea. Once pregnancy is excluded—an absolute prerequisite in every evaluation regardless of sexual history claims 1—the clinician must investigate compartments ranging from the hypothalamus to the ovary.
4.1 Functional Hypothalamic Amenorrhea (FHA): A Diagnosis of Exclusion
Functional Hypothalamic Amenorrhea (FHA) accounts for approximately 30-35% of secondary amenorrhea cases. It is a state of reversible hypogonadism caused by the suppression of the hypothalamic GnRH pulse generator.
4.1.1 Pathophysiology of Energy Availability
The core mechanism of FHA is often energy deficiency. The concept of "Energy Availability" (EA) is defined as dietary energy intake minus exercise energy expenditure, normalized to fat-free mass (FFM).
● Thresholds: Optimal physiological function occurs at an EA of >45 kcal/kg FFM/day. Menstrual disturbances begin when EA drops below 30 kcal/kg FFM/day.
● Metabolic Mediators: In states of low EA (starvation, anorexia, excessive exercise), adipose tissue mass decreases, leading to a drop in leptin. Low leptin signals the hypothalamus to conserve energy by shutting down non-essential functions, primarily reproduction. Concurrently, ghrelin (hunger hormone) and peptide YY increase.
● Stress Response: Psychological or physical stress activates the HPA axis, increasing CRH and cortisol. Cortisol directly inhibits GnRH neurons. Furthermore, increased beta-endorphin and dopaminergic tone contribute to the suppression.12
4.1.2 Clinical Presentation and "The Female Athlete Triad"
The classic presentation is the "Female Athlete Triad," now expanded to Relative Energy Deficiency in Sport (RED-S) to acknowledge the broader systemic effects. The triad components are:
1. Low Energy Availability (with or without an eating disorder).
2. Menstrual Dysfunction (ranging from luteal phase defects to amenorrhea).
3. Low Bone Mineral Density (osteopenia/osteoporosis).11
4.1.3 Diagnosis
FHA is a diagnosis of exclusion. The Endocrine Society Clinical Practice Guideline (2017) emphasizes excluding organic causes.
● Hormonal Profile: Low or low-normal FSH and LH (hypogonadotropic hypogonadism). Estradiol is low (<50 pg/mL). Prolactin and TSH are typically normal, though TSH may be slightly suppressed in severe starvation (euthyroid sick syndrome).17
● Neuroimaging: An MRI of the pituitary is recommended if there is a history of severe headaches, persistent vomiting, or if gonadotropins are extremely low, to rule out infiltrative diseases or tumors.17
4.1.4 Bone Health in FHA: A Critical Sequela
The most devastating consequence of FHA is bone loss. The mechanism is distinct from postmenopausal osteoporosis.
● Uncoupling of Remodeling: Estrogen deficiency leads to an increase in osteoclast activity via the RANKL pathway (discussed in Section 8). However, the nutritional deficit in FHA also causes osteoblast dysfunction. Low levels of IGF-1 (insulin-like growth factor 1) result in reduced bone formation. This "uncoupling" leads to rapid trabecular bone loss.
● Irreversibility: Unlike muscle mass, bone density lost during adolescence and early adulthood (the window of peak bone mass accrual) may never be fully recovered, even with the resumption of menses.16
4.2 Hyperprolactinemia
Prolactin is unique among anterior pituitary hormones because its primary control is inhibitory. Dopamine, secreted by the hypothalamus, travels down the pituitary stalk to bind D2 receptors on lactotrophs, suppressing prolactin release.
4.2.1 Etiology
● Prolactinomas: These are benign adenomas of the lactotrophs. They are the most common functional pituitary tumor.
○ Microadenomas: <10 mm in diameter.
○ Macroadenomas: >10 mm. These can impinge on the optic chiasm, causing bitemporal hemianopsia.21
● The "Stalk Effect": Any mass (e.g., craniopharyngioma, non-functioning adenoma) that compresses the pituitary stalk can block dopamine transport. This results in disinhibition of lactotrophs and modest hyperprolactinemia (usually <100 ng/mL).
● Medications: Antipsychotics (risperidone, haloperidol), anti-emetics (metoclopramide), and verapamil can cause hyperprolactinemia by blocking dopamine receptors.
● Hypothyroidism: Severe primary hypothyroidism leads to elevated TRH (Thyrotropin-Releasing Hormone). TRH acts as a stimulator of prolactin release, leading to secondary hyperprolactinemia.21
4.2.2 Diagnostic Nuances
● Macroprolactinemia: In 10-25% of patients with elevated prolactin, the hormone is bound to an IgG antibody, forming a large complex ("Big-Big Prolactin"). This complex is biologically inactive but is detected by immunoassays. If a patient is asymptomatic (no galactorrhea, regular menses) but has high prolactin, screening for macroprolactin with polyethylene glycol precipitation is mandatory to avoid unnecessary treatment.22
● The Hook Effect: In cases of giant prolactinomas (>4 cm) with extremely high prolactin levels (>5,000 ng/mL), the assay antibodies may become saturated, leading to a falsely low or "normal" result. If a large tumor is seen on MRI but prolactin levels are only mildly elevated, the sample must be diluted (1:100) to reveal the true value.22
4.3 Polycystic Ovary Syndrome (PCOS): The 2023 Paradigm Shift
PCOS is the most common endocrine cause of amenorrhea, affecting 8-13% of reproductive-age women. It is a heterogeneous metabolic-reproductive disorder.
4.3.1 Evolution of Diagnostic Criteria
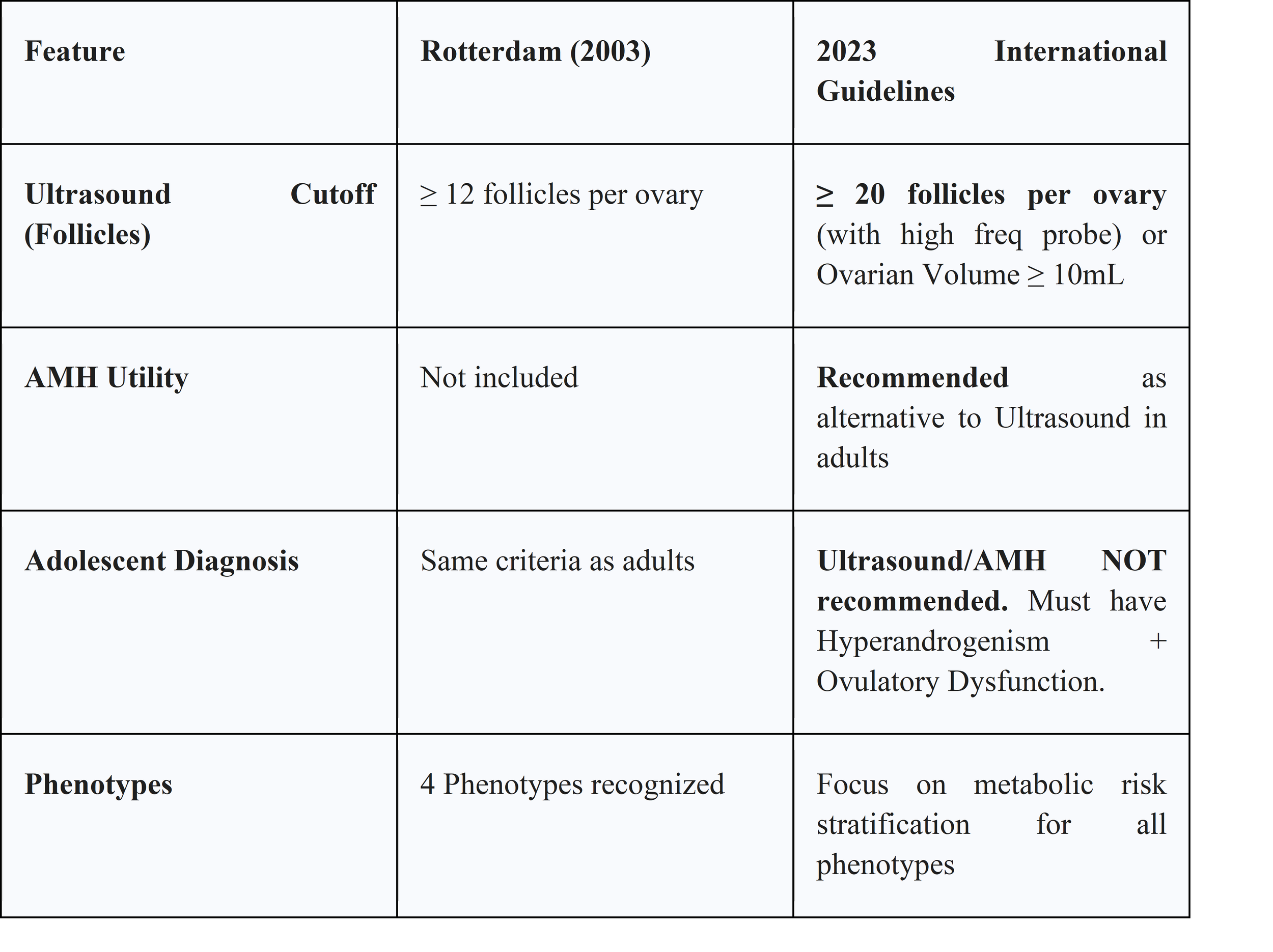
The diagnosis of PCOS has evolved from the NIH criteria (1990) to the Rotterdam criteria (2003) and most recently to the 2023 International Evidence-Based Guidelines.
● Rotterdam Criteria (2003): Required two of three:
1. Oligo-anovulation.
2. Hyperandrogenism (clinical or biochemical).
3. Polycystic Ovarian Morphology (PCOM) on ultrasound.
● 2023 Updates and AMH: The most significant change in the 2023 guidelines is the inclusion of Anti-Müllerian Hormone (AMH) as a diagnostic marker.
○ Adults: Elevated serum AMH can now replace ultrasound to fulfill the PCOM criterion. This simplifies diagnosis, particularly in telemedicine or resource-limited settings.24
○ Adolescents: The guidelines specifically recommend against using ultrasound or AMH for diagnosis in adolescents (<8 years post-menarche). This is because multi-follicular ovaries and irregular cycles are physiological during pubertal maturation. Diagnosis in adolescents requires both hyperandrogenism and ovulatory dysfunction.24
4.3.2 Ultrasound Thresholds
The definition of PCOM has been refined. The older threshold of ≥12 follicles per ovary resulted in overdiagnosis as ultrasound resolution improved. The 2023 guidelines recommend a threshold of ≥20 follicles per ovary (FNPO) and/or an ovarian volume ≥10 mL, using a high-frequency transvaginal transducer. If older technology is used, ovarian volume is the preferred metric.26
4.3.3 Pathophysiology of Anovulation in PCOS
PCOS is characterized by "follicular arrest." Small antral follicles are recruited but fail to reach dominance.
● Insulin Resistance: Hyperinsulinemia acts on the ovarian theca cells to synergize with LH, driving excess androgen production. It also suppresses Sex Hormone Binding Globulin (SHBG) production in the liver, increasing free testosterone.
● LH Hypersecretion: Women with PCOS often have high-frequency LH pulses. This constant LH drive favors androgen production over follicular maturation, preventing the LH surge required for ovulation.28
4.4 Primary Ovarian Insufficiency (POI)
POI differs clinically from menopause. Menopause is the irreversible cessation of function due to follicular depletion. POI, defined as loss of function before age 40, is a variable state. Up to 50% of women with POI have intermittent ovarian function, and 5-10% may conceive spontaneously.
4.4.1 Etiologies and Genetics
● Genetic:
○ Fragile X Premutation: The FMR1 gene (Xq27.3) normally has <45 CGG repeats. A premutation (55-200 repeats) leads to the production of toxic mRNA that damages ovarian follicles. Approximately 13-15% of familial POI cases are due to FMR1 premutations. This has profound implications for future offspring (risk of full Fragile X syndrome causing intellectual disability).10
○ Turner Mosaicism: As discussed in Primary Amenorrhea.
● Autoimmune: Autoimmune oophoritis is associated with lymphocytic infiltration of the theca cells. It is strongly linked to adrenal autoimmunity (Addison's disease). ACOG recommends screening all POI patients for 21-hydroxylase antibodies (adrenal antibodies) and TSH.10
● Iatrogenic: Chemotherapy (alkylating agents like cyclophosphamide) and radiation are major causes of POI.
5. Diagnostic Evaluation Strategies
The evaluation of amenorrhea must be systematic, moving from non-invasive screening to complex imaging.
5.1 The Initial Workup
Every patient with amenorrhea requires:
1. Pregnancy Test (hCG): To exclude the most common cause.
2. FSH, LH, Estradiol: To categorize into Compartments (Hypergonadotropic vs. Hypogonadotropic).
3. Prolactin and TSH: To rule out pituitary and thyroid disease.
4. Total Testosterone/DHEAS: Only if there are clinical signs of hyperandrogenism (hirsutism, virilization).
5.2 Critique of the Progestin Challenge Test (PCT)
Historically, the PCT (administering medroxyprogesterone 10mg for 10 days) was used to assess estrogen status. A withdrawal bleed was interpreted as "positive" (adequate estrogen, anovulation), while no bleed was "negative" (hypoestrogenism or outflow obstruction).
● Current Consensus: The ASRM and most experts now consider the PCT unnecessary and potentially misleading.
● Limitations:
○ False Positives: In FHA, the endometrium may be primed just enough to bleed, masking the diagnosis of hypothalamic dysfunction.31
○ False Negatives: A patient with PCOS may have high androgens causing endometrial atrophy, failing to bleed despite having estrogen.
● Modern Approach: Direct measurement of serum estradiol and endometrial thickness on ultrasound provides more accurate information than the PCT. If outflow obstruction is suspected (e.g., Asherman's), an Estrogen-Progestin Challenge (giving estrogen first to build the lining) is the appropriate diagnostic step.32
5.3 Advanced Imaging Protocols
● Pelvic Ultrasound: Transvaginal ultrasound is the gold standard for assessing ovarian morphology (PCOS) and endometrial thickness. In adolescents or virilized patients, transabdominal views may be needed.
● MRI of the Sella Turcica: If prolactin is elevated, a dedicated pituitary MRI is required.
○ Protocol: Standard brain MRI is insufficient. The protocol must include thin-section (2-3mm) T1 and T2 weighted images through the sella.
○ Dynamic Contrast: The critical sequence is the dynamic contrast-enhanced T1 sequence. Pituitary adenomas enhance slower than the normal pituitary gland. Rapid sequence imaging immediately after gadolinium injection allows the detection of microadenomas that would otherwise be isointense to the gland on delayed images.34
6. Therapeutic Management and Pharmacology
Treatment is strictly etiology-dependent, aiming to restore fertility (if desired) and prevent long-term sequelae.
6.1 Management of Hyperprolactinemia
Dopamine agonists (DAs) are the first-line therapy, even for macroadenomas with visual field defects, as they cause rapid tumor shrinkage.
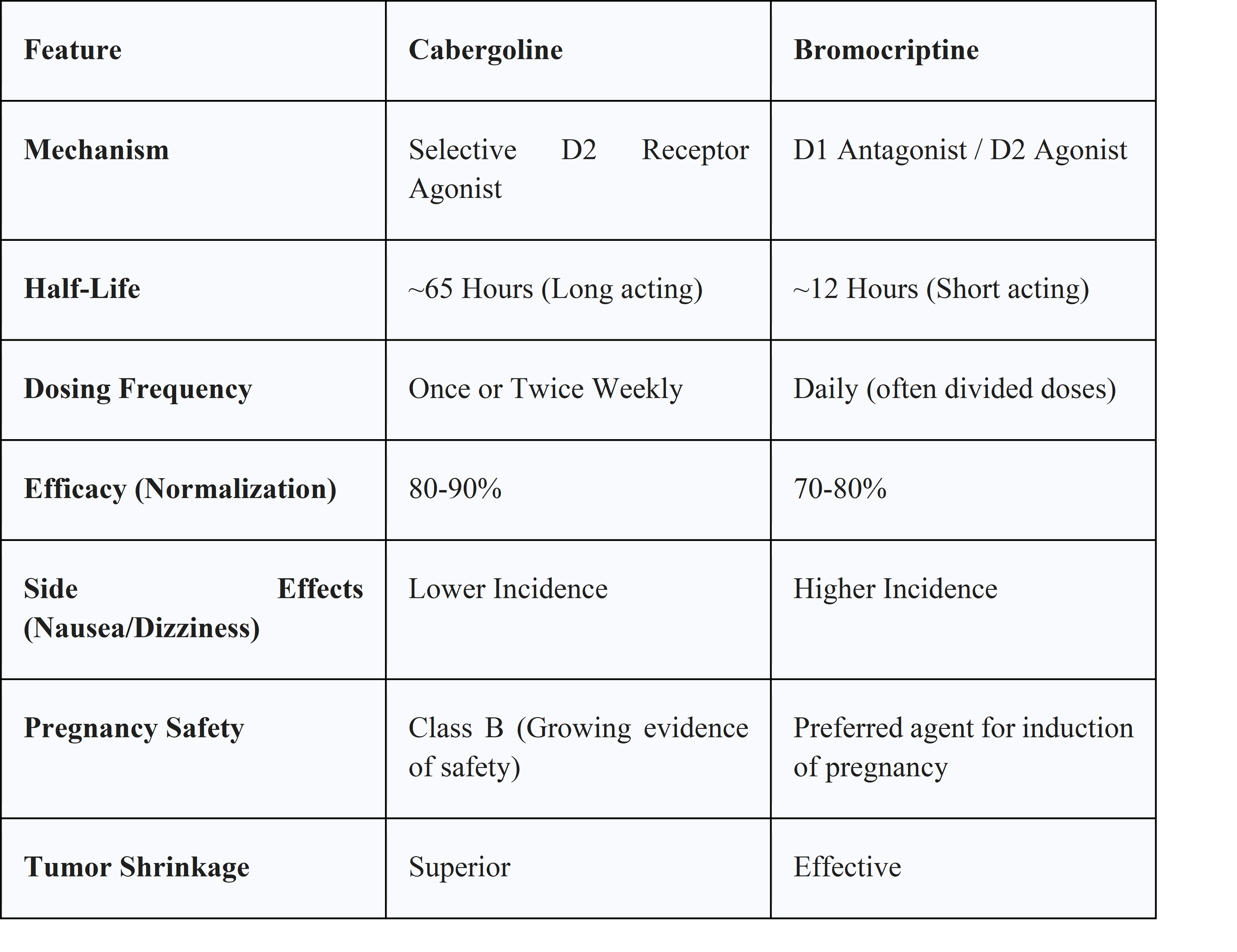
● Cabergoline:
○ Pharmacology: A selective D2 receptor agonist with a very long half-life (~65 hours), allowing for once or twice-weekly dosing.
○ Efficacy: Superior to bromocriptine in normalizing prolactin (90% vs 70%) and shrinking tumors.
○ Adverse Effects: Nausea, dizziness. High cumulative doses (used in Parkinson's, rarely in prolactinoma) are associated with cardiac valvulopathy, though this risk is negligible at standard endocrine doses.36
● Bromocriptine:
○ Pharmacology: Non-selective dopamine agonist with a short half-life requiring daily dosing.
○ Use Case: While less effective, it has a longer safety record in pregnancy and is often the drug of choice for ovulation induction in women attempting to conceive.36
6.2 Management of FHA and RED-S
● Behavioral: The primary treatment is increasing Energy Availability. This requires a multidisciplinary team (dietitian, therapist). Cognitive Behavioral Therapy (CBT) has been shown to restore ovulation in FHA patients even without significant weight gain by reducing the stress-cortisol drive.17
● Hormonal: Oral contraceptives (OCPs) are not recommended for bone health in FHA. The hepatic first-pass effect of oral ethinyl estradiol suppresses IGF-1, which is crucial for bone formation. This exacerbates the bone deficits in FHA.
● Recommendation: The Endocrine Society advises transdermal estradiol (patch) with cyclic oral progesterone. This route bypasses the liver, does not suppress IGF-1, and provides physiological estrogen replacement.16
6.3 Management of PCOS
● Combined Oral Contraceptives (COCs): First-line for menstrual regulation and hyperandrogenism. They work by:
1. Suppressing LH (reducing ovarian androgen production).
2. Increasing SHBG (binding free testosterone).
3. Thinned endometrium (preventing hyperplasia).28
● Metformin: An insulin sensitizer. Recommended for patients with metabolic syndrome or glucose intolerance. It has modest effects on restoring ovulation but is less effective than COCs for hirsutism.
● Fertility:
○ Letrozole: An aromatase inhibitor. It blocks estrogen production, reducing negative feedback and increasing FSH secretion. It is now the first-line ovulation induction agent for PCOS, showing higher live birth rates than clomiphene citrate.28
○ Gonadotropins/IVF: Second-line therapies.
6.4 Management of POI
● Hormone Replacement Therapy (HRT): Unlike menopausal women where the goal is the lowest dose for the shortest time, women with POI need full physiological replacement until the average age of natural menopause (age 50-51).
○ Regimen: Transdermal estradiol (100 mcg patch) or oral estradiol (2 mg) daily, combined with cyclic micronized progesterone (200 mg for 12 days/month) to mimic a natural cycle.
○ Rationale: This high dose is necessary to maintain bone density and cardiovascular health. OCPs are often used for convenience but contain ethinyl estradiol, which has higher thrombotic risk than bioidentical estradiol.10
7. Long-Term Health Sequelae and Pathophysiology of Bone Loss
The most significant non-reproductive consequence of amenorrhea is the loss of skeletal integrity.
7.1 The RANKL/OPG Pathway
Bone remodeling relies on a balance between osteoclasts (resorption) and osteoblasts (formation).
● Mechanism: Osteoblasts secrete RANKL (Receptor Activator of Nuclear Factor Kappa-B Ligand), which binds to RANK receptors on osteoclast precursors, stimulating their differentiation and activity. Osteoblasts also secrete Osteoprotegerin (OPG), a decoy receptor that binds RANKL, preventing it from activating osteoclasts.
● Estrogen's Role: Estrogen stimulates the production of OPG and suppresses RANKL. In amenorrhea (hypoestrogenism), OPG levels plummet and RANKL levels rise. This leads to unchecked osteoclast activity and rapid bone resorption.
● Clinical Impact: Women with amenorrhea lose trabecular bone (spine, hip) at a rate of 2-5% per year. Early intervention is critical because the microarchitectural damage (disconnection of trabeculae) is often irreversible.39
7.2 Cardiovascular Disease
Estrogen promotes vasodilation via nitric oxide synthase and maintains a favorable lipid profile (high HDL, low LDL). Premature loss of estrogen in POI is associated with endothelial dysfunction and a twofold increase in cardiovascular mortality. This underscores the necessity of aggressive HRT in this population.10
8. Tables and Data Summaries
Table 1: Differential Diagnosis of Amenorrhea by Compartment
9. Conclusion
Amenorrhea is a sentinel symptom that requires a rigorous, evidence-based evaluation. The clinical landscape has shifted significantly in the last decade. We have moved away from the Progestin Challenge Test and toward direct hormonal assessment. We have refined the diagnosis of PCOS to avoid over-labeling adolescents while utilizing AMH for easier adult diagnosis. We have recognized FHA not just as a reproductive nuisance but as a systemic state of energy deficiency with profound skeletal consequences requiring transdermal, not oral, estrogen.
Whether the etiology is the "suppressed" axis of FHA, the "hyperactive" axis of POI, or the "disrupted" axis of PCOS, the clinician’s role extends beyond the restoration of menses. It encompasses the preservation of fertility, the protection of bone density, and the mitigation of cardiovascular risk. Management must be holistic, addressing the genetic, autoimmune, metabolic, and psychological dimensions of the patient's health.
References
1. Current evaluation of amenorrhea: a committee opinion (2024) - ASRM, https://www.asrm.org/practice-guidance/practice-committee-documents/current-evaluation-of-amenorrhea/
2. Current evaluation of amenorrhea, https://obgyn.med.jax.ufl.edu/wordpress/files/2025/01/amenorrhea.pdf
3. Williams Gynecology, Fourth Edition|Hardcover - Barnes & Noble, https://www.barnesandnoble.com/w/williams-gynecology-fourth-edition-lisa-m-halvorson/1147594418
4. Amenorrhea | Williams Gynecology, 3e - AccessMedicine - McGraw Hill Medical, https://accessmedicine.mhmedical.com/Content.aspx?bookid=1758§ionid=118169857
5. Speroff's Clinical Gynecologic Endocrinology and Infertility|Hardcover - Barnes & Noble, https://www.barnesandnoble.com/w/speroffs-clinical-gynecologic-endocrinology-and-infertility-hugh-taylor-md/1123884927
6. Speroff's Clinical Gynecologic Endocrinology and Infertility - Hugh S. Taylor, Lubna Pal, Emre Seli - Google Books, https://books.google.com/books/about/Speroff_s_Clinical_Gynecologic_Endocrino.html?id=yhGHEQAAQBAJ
7. Berek & Novak's Gynecology - ResearchGate, https://www.researchgate.net/publication/262098292_Berek_Novak's_Gynecology
8. Berek & Novak's Gynecology - Wolters Kluwer, https://shop.lww.com/Berek---Novak-s-Gynecology/p/9781975238636
9. Amenorrhea - StatPearls - NCBI Bookshelf, https://www.ncbi.nlm.nih.gov/books/NBK482168/
10. Primary Ovarian Insufficiency - Gynecology and Obstetrics - Merck Manuals, https://www.merckmanuals.com/professional/gynecology-and-obstetrics/abnormal-uterine-bleeding/primary-ovarian-insufficiency
11. Amenorrhea - Gynecology and Obstetrics - MSD Manual Professional Edition, https://www.msdmanuals.com/professional/gynecology-and-obstetrics/abnormal-uterine-bleeding/amenorrhea
12. Functional Hypothalamic Amenorrhea: Recognition and Management of a Challenging Diagnosis - PMC, https://pmc.ncbi.nlm.nih.gov/articles/PMC10491417/
13. Amenorrhea - Gynecology and Obstetrics - Merck Manual Professional Edition, https://www.merckmanuals.com/professional/gynecology-and-obstetrics/abnormal-uterine-bleeding/amenorrhea
14. Current evaluation of amenorrhea (2008) | American Society for Reproductive Medicine, https://integration.asrm.org/practice-guidance/practice-committee-documents/current-evaluation-of-amenorrhea/
15. Amenorrhea | Choose the Right Test - ARUP Consult, https://arupconsult.com/content/amenorrhea
16. HORMONES FOR BONE HEALTH IN FUNCTIONAL HYPOTHALAMIC AMENORRHOEA, https://www.endocrinology.org/endocrinologist/151-spring-24/features/hormones-for-bone-health-in-functional-hypothalamic-amenorrhoea/
17. Hypothalamic Amenorrhea Guideline Resources - Endocrine Society, https://www.endocrine.org/clinical-practice-guidelines/hypothalamic-amenorrhea
18. Functional Hypothalamic Amenorrhea: An Endocrine Society Clinical Practice Guideline - Oxford Academic, https://academic.oup.com/jcem/article/102/5/1413/3077281
19. Functional hypothalamic amenorrhea: An endocrine society clinical practice guideline, https://mayoclinic.elsevierpure.com/en/publications/functional-hypothalamic-amenorrhea-an-endocrine-society-clinical-/
20. Bone health in functional hypothalamic amenorrhea: What the endocrinologist needs to know - Frontiers, https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2022.946695/full
21. Hyperprolactinemia | Endocrine Society, https://www.endocrine.org/patient-engagement/endocrine-library/hyperprolactinemia
22. Diagnosis and management of hyperprolactinemia - PMC - NIH, https://pmc.ncbi.nlm.nih.gov/articles/PMC191295/
23. Correlation between MRI findings of pituitary gland and prolactin level among hyperprolactinemia adult female Saudi patients in rural areas: A retrospective multicentric study - PMC, https://pmc.ncbi.nlm.nih.gov/articles/PMC11730409/
24. International evidence-based guideline for the assessment and management of polycystic ovary syndrome 2023 – Summary - Monash University, https://www.monash.edu/__data/assets/pdf_file/0003/3371133/PCOS-Guideline-Summary-2023.pdf
25. Recommendations from the 2023 International Evidence-based Guideline for the Assessment and Management of Polycystic Ovary Syndrome - ASRM, https://www.asrm.org/practice-guidance/practice-committee-documents/recommendations-from-the-2023-international-evidence-based-guideline-for-the-assessment-and-management-of-polycystic-ovary-syndrome/
26. Polycystic ovarian syndrome | Radiology Reference Article | Radiopaedia.org, https://radiopaedia.org/articles/polycystic-ovarian-syndrome-1
27. Application of a new ultrasound criterion for the diagnosis of polycystic ovary syndrome, https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2022.915245/full
28. Polycystic Ovary Syndrome | obgynboardpass, http://obgynboardpass.com/wp-content/uploads/2024/06/PB-194-Polycystic-Ovary-Syndrome.pdf
29. Current Guidelines for Diagnosing PCOS - PMC - NIH, https://pmc.ncbi.nlm.nih.gov/articles/PMC10047373/
30. Primary Ovarian Insufficiency: How to Evaluate and Make the Diagnosis - The ObG Project, https://www.obgproject.com/2023/02/13/primary-ovarian-insufficiency-how-to-evaluate-and-make-the-diagnosis/
31. Progesterone Challenge Test | Procedure & Results - Ovasave, https://www.ovasave.com/blog/progesterone-challenge-test
32. Estrogen-Progesterone Challenge Test - FPnotebook, https://mobile.fpnotebook.com/Gyn/Lab/EstrgnPrgstrnChlngTst.htm
33. Progestogen challenge test - Wikipedia, https://en.wikipedia.org/wiki/Progestogen_challenge_test
34. What type of MRI is recommended for evaluating high prolactin levels? - Dr.Oracle, https://www.droracle.ai/articles/194324/what-type-of-mri-is-recommended-for-evaluating-high
35. Pituitary gland protocol (MRI) | Radiology Reference Article | Radiopaedia.org, https://radiopaedia.org/articles/pituitary-gland-protocol-mri
36. Clinical guidelines for diagnosis and treatment of prolactinoma and hyperprolactinemia | Endocrinología y Nutrición (English Edition) - Elsevier, https://www.elsevier.es/en-revista-endocrinologia-nutricion-english-edition--412-articulo-clinical-guidelines-for-diagnosis-treatment-S2173509313001190
37. Treatment of hyperprolactinemia in women: A Position Statement from the Brazilian Federation of Gynecology and Obstetrics Associations (Febrasgo) and the Brazilian Society of Endocrinology and Metabolism (SBEM) - PMC, https://pmc.ncbi.nlm.nih.gov/articles/PMC11081053/
38. Committee Opinion No. 698: Hormone Therapy in Primary Ovarian Insufficiency, https://www.researchgate.net/publication/317782381_Committee_Opinion_No_698_Hormone_Therapy_in_Primary_Ovarian_Insufficiency
39. Osteoporosis Due to Hormone Imbalance: An Overview of the Effects of Estrogen Deficiency and Glucocorticoid Overuse on Bone Turnover - MDPI, https://www.mdpi.com/1422-0067/23/3/1376
40. Primary Osteoporosis Induced by Androgen and Estrogen Deficiency: The Molecular and Cellular Perspective on Pathophysiological Mechanisms and Treatments - ResearchGate, https://www.researchgate.net/publication/385745375_Primary_Osteoporosis_Induced_by_Androgen_and_Estrogen_Deficiency_The_Molecular_and_Cellular_Perspective_on_Pathophysiological_Mechanisms_and_Treatments
41. Premature ovarian insufficiency: Updated concepts in diagnosis and hormonal treatment strategies | Clinics - Elsevier, https://www.elsevier.es/en-revista-clinics-22-articulo-premature-ovarian-insufficiency-updated-concepts-S1807593225001644
