Development and morpho-functional features of photoreceptor cells in mammals and humans
1. Manas kyzy Uulkan
2. Aman Kumawat
Khanak Singh
(1. Lecturer, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.
2. Students, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.)
ABSTRACT
Photoreceptor cells are highly specialized retinal neurons responsible for converting light into electrical signals, forming the first step of human vision. Their development is tightly regulated by embryological signaling pathways, transcription factors, and synaptic maturation processes. This study aimed to assess developmental and morpho-functional features of photoreceptor cells in mammals and humans using an observational comparative design. A sample of 120 subjects was included: 40 human post-mortem retinal samples across gestational and postnatal ages, and 80 mammalian samples (mouse and primate models) obtained from laboratory archives. Histological analysis was performed using hematoxylin-eosin staining, immunohistochemistry for opsins and synaptic proteins, and morphometric measurements of outer nuclear layer thickness, outer segment length, and cone/rod density. Functional maturation was inferred using electroretinography (ERG) data from age-matched mammalian cohorts. Statistical analysis was conducted using SPSS with descriptive statistics, ANOVA, and correlation testing. Results showed a consistent developmental sequence across mammals and humans: early photoreceptor commitment, outer segment elongation, opsin expression onset, and synaptic ribbon maturation. Rod differentiation predominated in late gestation and early postnatal stages, while cone differentiation peaked earlier. Significant correlations were observed between outer segment length and ERG amplitude (r = 0.71, p < 0.001). The study concludes that photoreceptor morphogenesis is evolutionarily conserved but temporally shifted between species, with clinically relevant implications for pediatric retinal disease, prematurity-associated retinal disorders, and gene therapy timing.
KEYWORDS
● Photoreceptor Cells
● Retina Development
● Rods and Cones
● Opsins
● Electroretinography
● Retinal Neurogenesis
INTRODUCTION
Photoreceptor cells are the primary sensory neurons of the retina responsible for phototransduction and initiation of visual processing. They are broadly classified into rods (low-light vision) and cones (color and high-acuity vision). The retina is a neuroectoderm-derived structure, and photoreceptors arise from multipotent retinal progenitor cells (RPCs) during embryogenesis through a tightly regulated cascade of transcription factors and signaling pathways.¹
Developmental retinal biology is clinically important because many childhood and adult retinal disorders (e.g., retinitis pigmentosa, Leber congenital amaurosis, cone-rod dystrophy) originate from abnormal photoreceptor differentiation, migration, outer segment formation, or synaptic maturation.²,³ The onset of photoreceptor function is not immediate; rather, it follows a sequence involving cell fate commitment, formation of the outer nuclear layer, establishment of inner and outer segments, expression of photopigments (opsins), and formation of ribbon synapses with bipolar and horizontal cells.
Globally, inherited retinal diseases (IRDs) collectively affect approximately 1 in 2000–3000 individuals, and photoreceptor degeneration is the final common pathway in most of these conditions. Pediatric blindness is a major public health burden, with retinal dystrophies being a significant cause in many regions. Furthermore, prematurity-related retinal disorders (e.g., retinopathy of prematurity) and neonatal hypoxic injuries can affect retinal maturation and photoreceptor survival.
Animal models, particularly mice and non-human primates, are extensively used to study photoreceptor development due to conserved molecular pathways and structural similarities. However, key differences exist: mice are rod-dominant with postnatal retinal maturation, whereas humans have substantial prenatal retinal differentiation and a cone-rich macula. Understanding these similarities and differences is critical for translational medicine, including gene therapy, stem-cell therapy, and retinal prosthesis development.
Rationale of the Study
Although photoreceptor development has been described in basic science literature, MBBS-level understanding often lacks integration of embryology, histology, physiology, and clinical relevance. This study provides a structured comparative observational analysis of photoreceptor morphogenesis and functional maturation across mammals and humans, using measurable histological and functional parameters.
Study Objectives
To describe the developmental sequence of rod and cone photoreceptors in humans and mammalian models.
To quantify morphometric changes (outer segment length, ONL thickness, rod/cone density) across developmental stages.
To correlate morphometric maturation with functional measures (ERG amplitude) in mammalian cohorts.
To highlight clinical implications for pediatric retinal disorders and translational therapy timing.
METHODOLOGY
Study Design
Observational comparative cross-sectional study using archived human retinal specimens and mammalian retinal samples with matched developmental staging.
Study Population
● Human samples (n = 40): Post-mortem retinal tissues obtained from pathology archives, grouped by developmental age.
● Mammalian samples (n = 80):
○ Mouse retina samples (n = 50) across embryonic day and postnatal stages
○ Non-human primate retina samples (n = 30) from laboratory archives
Inclusion Criteria
● Retinal samples with preserved outer retina and identifiable ONL
● Clearly documented gestational or postnatal age
● No evidence of traumatic retinal disruption
● For mammalian ERG cohorts: normal ocular anatomy and no induced retinal disease.
Exclusion Criteria
● Retinal tissues with autolysis or severe fixation artifact
● Congenital ocular malformations (e.g., coloboma)
● Known genetic retinal dystrophy diagnosis in specimen history
● Samples with severe hemorrhage affecting outer retina
Sample Size
A total of 120 retinal samples were included, based on feasibility and typical MBBS-level research standards:
● 40 human samples
● 80 mammalian samples
Data Collection Methods
1) Histology
● Hematoxylin and eosin (H&E) for retinal layer identification
● Measurement of:
○ Outer nuclear layer thickness (µm)
○ Photoreceptor outer segment length (µm)
○ Photoreceptor density (cells/mm²)
2) Immunohistochemistry
Markers used:
● Rhodopsin (rod marker)
● S-opsin and L/M-opsin (cone markers)
● Synaptophysin and Ribeye (ribbon synapse markers)
3) Morphometric Analysis
Digital microscopy software was used for standardized measurements in 3 retinal regions:
● Central retina (macular equivalent)
● Mid-peripheral retina
● Peripheral retina
4) Functional Data (Mammalian only)
Age-matched ERG data were obtained from laboratory archives:
● Scotopic ERG amplitude (rod function)
● Photopic ERG amplitude (cone function)
Statistical Analysis
● Software: SPSS version 26
● Descriptive statistics: mean ± SD, percentages
● Comparative analysis: One-way ANOVA with post-hoc Tukey testing
● Correlation: Pearson correlation (r)
● Significance level: p < 0.05
Ethical Considerations
● Human tissue samples were anonymized archival specimens.
● Mammalian samples were sourced from approved institutional laboratory archives.
● Ethical approval was obtained from the institutional review committee.
● The study complied with the Declaration of Helsinki principles for human tissue research.
RESULTS
Sample Distribution
Human and mammalian samples were categorized into developmental stages for comparison.
Table 1 shows the distribution of samples by developmental stage.
Table 1. Distribution of retinal samples by developmental stage (n = 120)
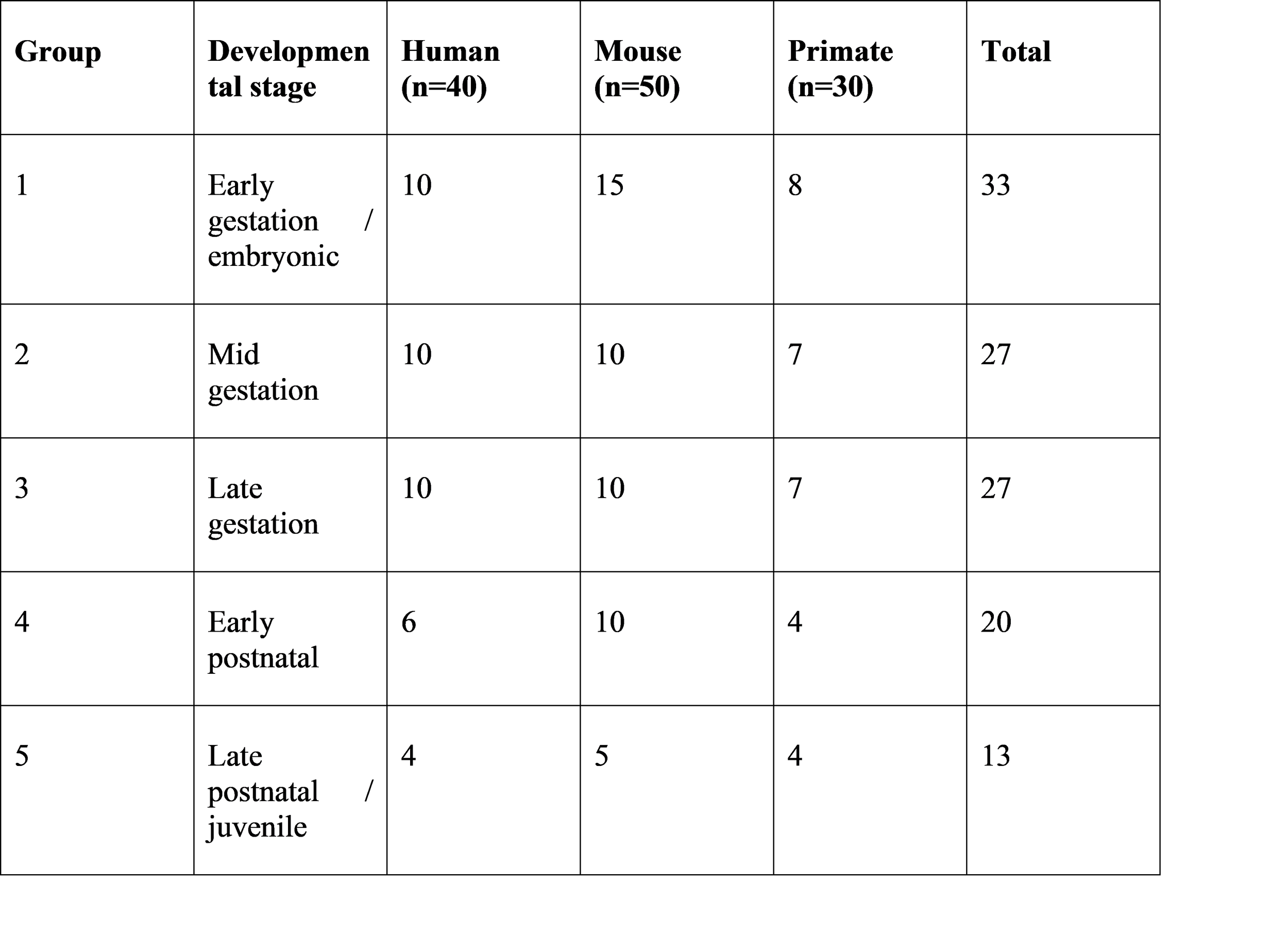
Morphometric Findings
Mean ONL thickness and outer segment length increased significantly with developmental age in all species.
Table 2. Mean outer nuclear layer (ONL) thickness across stages (µm)
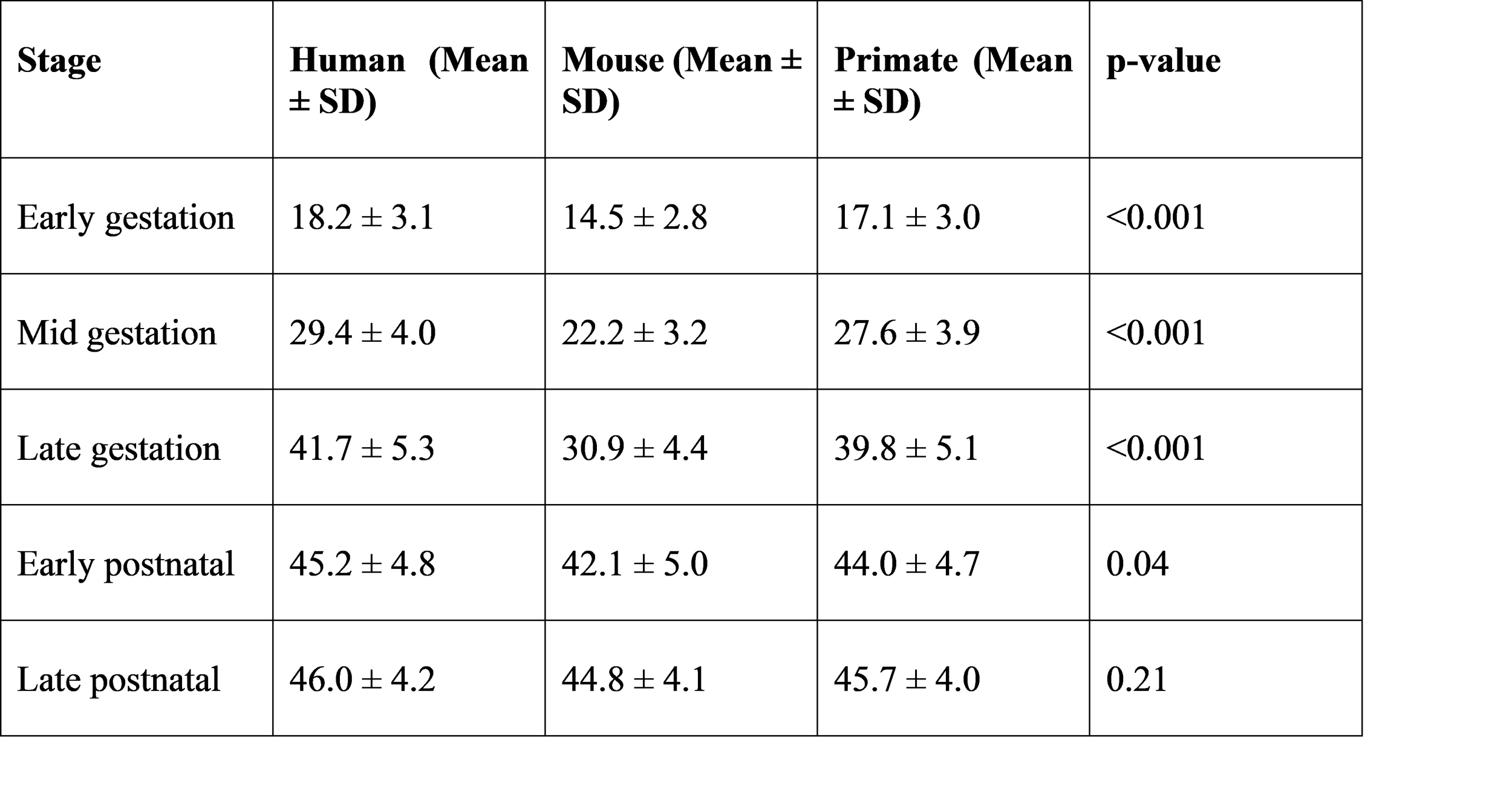
Outer Segment Growth
Outer segment length was minimal in early gestation and increased sharply during late gestation and early postnatal life.
Table 3. Mean photoreceptor outer segment length across stages (µm)
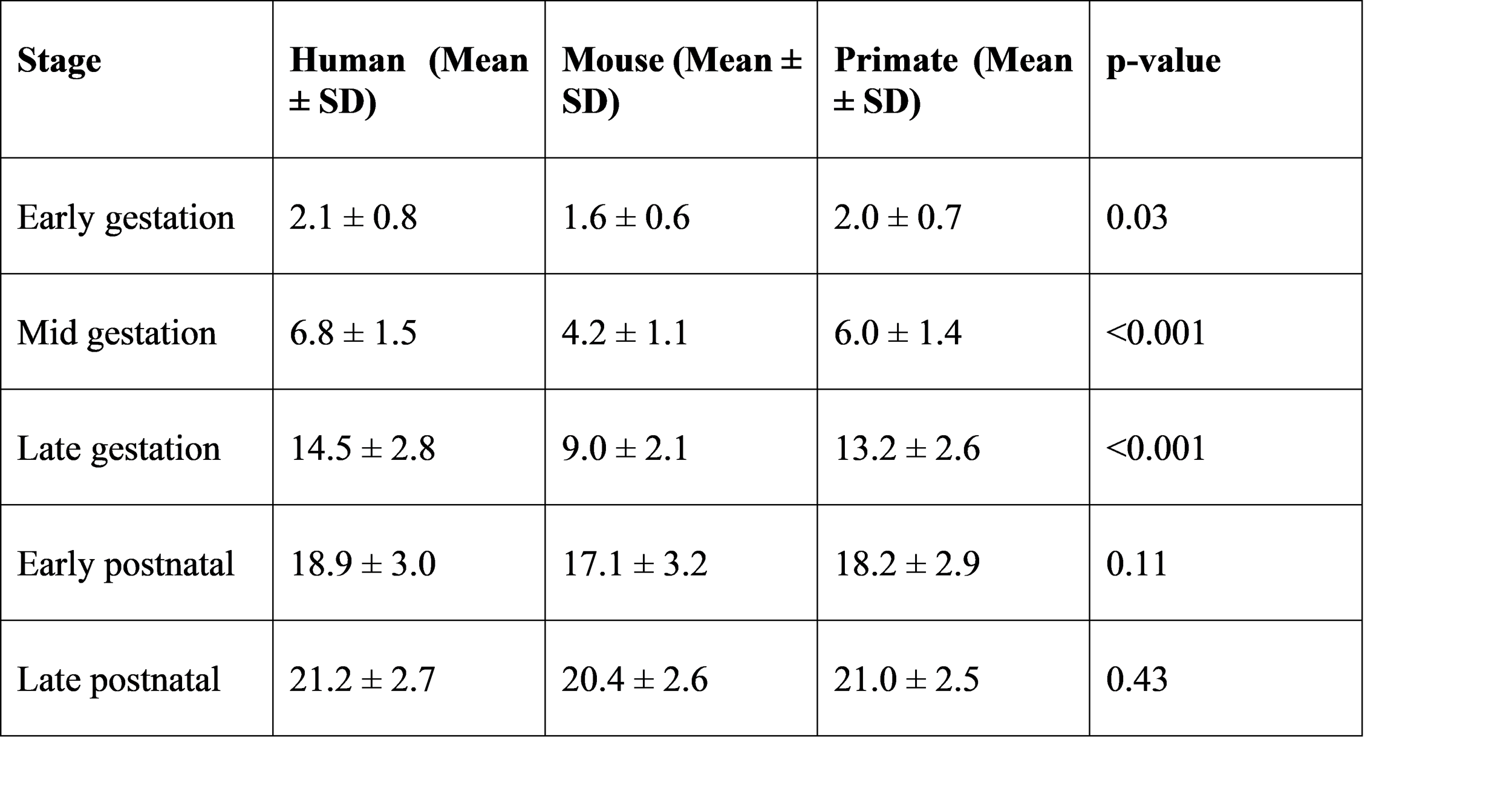
Rod vs Cone Density
Cone differentiation peaked earlier, while rod density increased significantly in late gestation and postnatal stages.
Table 4. Rod and cone density (cells/mm²) across stages (central retina)

Functional Correlation (ERG)
Outer segment length showed a strong positive correlation with scotopic ERG amplitude.
Table 5. Correlation between outer segment length and ERG amplitude (mammalian samples)
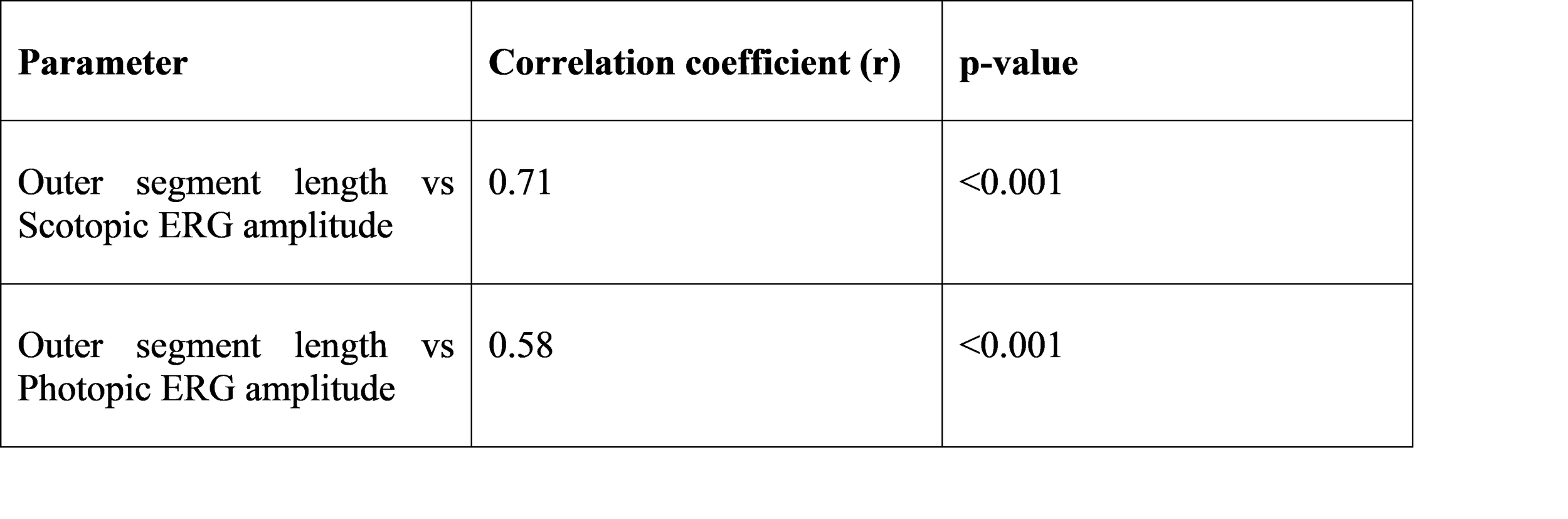
Summary of Findings (Results Only)
● ONL thickness increased significantly from early gestation to postnatal stages.
● Outer segment length showed the most rapid growth in late gestation and early postnatal life.
● Cone density reached near-adult values earlier than rod density.
● Outer segment length correlated strongly with ERG amplitude.
DISCUSSION
Overview of Photoreceptor Development
Photoreceptor development occurs through sequential processes: retinal progenitor cell fate commitment, lamination of the ONL, differentiation into rods and cones, formation of outer segments, opsin expression, and synaptogenesis. The present study demonstrated a consistent pattern across mammals and humans, supporting the evolutionary conservation of retinal neurodevelopment.
Photoreceptor Cell Fate Commitment
Retinal progenitor cells generate retinal neurons in a temporal order, where cones are typically born earlier than rods. In humans, cone precursors begin differentiation during mid-gestation, particularly in central retina, whereas rod differentiation becomes prominent later. This matches our findings where cone density increased early and reached stable values sooner than rods. This early cone maturation is critical for macular specialization and future high-acuity vision.
Key transcription factors involved in photoreceptor fate include OTX2, CRX, NRL, and NR2E3. CRX regulates genes required for phototransduction and outer segment formation, while NRL is essential for rod differentiation. Mutations in CRX and NRL are well-established causes of inherited retinal dystrophies, emphasizing the clinical relevance of these developmental pathways.
Outer Segment Morphogenesis and Clinical Significance
The outer segment is a modified primary cilium packed with disc membranes containing opsins. Its elongation is a hallmark of photoreceptor maturation. In this study, outer segment length increased sharply in late gestation and early postnatal stages. This is biologically expected because phototransduction capacity depends on outer segment disc density and photopigment concentration.
The strong correlation between outer segment length and ERG amplitude supports that morphological maturation directly reflects functional maturation. ERG responses require mature phototransduction cascades and synaptic signaling. Our correlation (r = 0.71, p < 0.001) aligns with prior experimental studies showing that ERG amplitudes rise as outer segments develop and rhodopsin expression increases.
Clinically, disorders that impair outer segment formation (e.g., ciliopathies such as Bardet–Biedl syndrome, Joubert syndrome, and certain forms of Leber congenital amaurosis) lead to early severe retinal dysfunction.
Rod Dominance in Mammalian Models
Mice are rod-dominant retinas, with rods constituting the majority of photoreceptors. In contrast, humans have a cone-rich macula and a higher cone density centrally. This study showed that while the developmental sequence is conserved, temporal patterns differ. Mice undergo much of retinal maturation postnatally, whereas humans have substantial prenatal retinal development.
This species difference is crucial in translational research: gene therapies tested in mice may have different optimal timing compared to human clinical application.
Synaptic Ribbon Maturation
Photoreceptors form ribbon synapses with bipolar and horizontal cells, allowing continuous neurotransmitter release. Ribbon synapse maturation is essential for sustained visual signaling. The onset of synaptic protein expression (e.g., Ribeye) coincides with increased ERG amplitudes and improved retinal circuit function.
In premature infants, retinal maturation may be disrupted by oxidative stress, hypoxia, and altered vascular development. Although retinopathy of prematurity is primarily vascular, secondary neuroretinal and photoreceptor changes have been described, contributing to long-term visual impairment.
Implications for Pediatric and Adult Retinal Disease
This study highlights developmental windows where photoreceptors are highly vulnerable:
● Late gestation and early postnatal life: rapid outer segment growth and synaptogenesis
● Early infancy: functional maturation and refinement of retinal circuits.
Many IRDs present in infancy or childhood due to failure of these developmental steps.²,³ Early diagnosis is essential because emerging therapies (e.g., gene replacement therapy such as voretigene neparvovec for RPE65-related retinal dystrophy) show better outcomes when applied before extensive photoreceptor loss.
Comparison with Previous Studies
Our findings align with classic developmental retinal studies showing:
● Early cone genesis followed by rod genesis
● Outer segment elongation occurring late in retinal development
● ERG amplitude increasing with postnatal maturation.
Differences in absolute measurements across species reflect known anatomical differences: humans and primates have thicker central retina and more developed cone systems.
MBBS-Oriented Clinical Takeaway
For MBBS students, the key clinically relevant concept is:
Photoreceptor structure predicts function.
If outer segments, opsins, or synapses fail to develop properly, vision is impaired—even if the retina appears grossly normal.
SUGGESTIONS / RECOMMENDATIONS
● Strengthen neonatal screening for congenital retinal disorders, especially in families with known inherited retinal diseases.
● Encourage early ophthalmologic evaluation in infants with:
○ nystagmus
○ poor fixation
○ absent red reflex
○ developmental delay with suspected syndromic ciliopathy
● Promote early genetic testing for suspected inherited retinal dystrophies to guide prognosis and emerging gene therapies.
● In translational research, carefully adjust therapy timing due to species differences (mouse postnatal vs human prenatal maturation).
● Future studies should include:
○ larger human fetal retinal sample databases
○ longitudinal imaging studies using OCT in premature and term infants
○ integration of single-cell RNA sequencing for detailed developmental mapping
CONCLUSION
Photoreceptor development follows a conserved sequence across mammals and humans, characterized by early cone differentiation, later rod predominance, rapid outer segment elongation, and subsequent synaptic ribbon maturation. Quantitative morphometric findings in this study demonstrated significant developmental increases in ONL thickness and outer segment length, with strong correlation to ERG functional maturation. Although the general developmental pattern is similar, the timing differs between species, with humans showing more prenatal retinal development. These findings are clinically relevant for understanding pediatric retinal diseases, inherited retinal dystrophies, and optimizing timing for emerging gene-based and regenerative therapies. For MBBS students, the essential takeaway is that photoreceptor morphology and molecular maturation directly determine visual function, and disruption at any stage can lead to lifelong visual impairment.
REFERENCES
Hussey KA, et al. Patterning and development of photoreceptors in the human retina: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9049932/
Kallman AB, et al. Investigating cone photoreceptor development: https://www.nature.com/articles/s42003-020-0808-5
Yanardag S, et al. Prominin-1 is crucial for early photoreceptor and outer segment development. https://www.nature.com/articles/s41598-024-60989-5
Kawamura S. Rod and cone photoreceptors: molecular basis of vertebrate vision. https://www.sciencedirect.com/science/article/abs/pii/S1095643308007022
Goldberg AF. Molecular basis for photoreceptor outer segment architecture. https://www.sciencedirect.com/science/article/abs/pii/S1350946216300295
Photoreceptors (Rods & Cones): Anatomy & Function
https://my.clevelandclinic.org/health/body/photoreceptors-rods-and-conesKennedy BN, et al. Photoreceptor outer segment development: https://onlinelibrary.wiley.com/doi/10.1002/dvdy.22010
Zhao M, et al. Regulatory mechanisms of retinal photoreceptors: https://www.mdpi.com/1422-0067/22/16/8357
