Latent Tuberculosis Infection
1. Kurmanaliev Nurlanbek Kambaralyevich
2. Rajwardhan Mohan Punekar
Pratap Paritosh Sarkar
(1. Teacher, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.
https://orcid.org/0000-0002-5952-1463
2. Students, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.)
Abstract
Latent Tuberculosis Infection (LTBI) represents a clinical paradox: the presence of a virulent pathogen within a host who shows no evidence of disease. It is estimated that one-quarter of the global population is infected with Mycobacterium tuberculosis, serving as a massive reservoir for future active cases. This article explores the immunopathogenesis of latency, challenging the binary view of "active vs. latent" in favour of a spectrum of infection. We review the diagnostic challenges, comparing the traditional Tuberculin Skin Test (TST) with modern Interferon-Gamma Release Assays (IGRA), and discuss the risk factors that drive reactivation. Finally, we analyse the evolving treatment landscape, highlighting the shift from long-course isoniazid monotherapy to short-course rifamycin-based regimens (3HP, 4R), which are essential for global TB elimination strategies.
Keywords: Latent Tuberculosis Infection (LTBI), Granuloma, Interferon-Gamma Release Assay (IGRA), Tuberculin Skin Test, Isoniazid, Rifapentine.
1. Introduction
Tuberculosis (TB) is often visualized as a disease of coughing, wasting, and hemoptysis. However, for the vast majority of infected individuals (approximately 90–95%), the interaction with Mycobacterium tuberculosis (M.tb) is silent. This state, known as Latent Tuberculosis Infection (LTBI), is defined as a state of persistent immune response to stimulation by M.tb antigens without evidence of clinically manifested active TB [1].
From a public health perspective, LTBI is the "iceberg beneath the water." While these individuals are not infectious, they carry a lifelong risk of reactivation. This risk is not uniform; it is dramatically amplified by conditions that compromise cellular immunity, such as HIV infection, diabetes mellitus, and immunosuppressive therapies. Therefore, the identification and treatment of LTBI in high-risk groups is a cornerstone of the World Health Organization’s End TB Strategy [2].
2. Pathophysiology: The Granuloma
Latency is not a passive state where the bacteria are simply "asleep." It is an active, dynamic standoff between the pathogen and the host's immune system.
2.1 Formation of the Granuloma
Upon inhalation, M.tb is phagocytosed by alveolar macrophages. If the innate immune system fails to kill the bacteria, an adaptive immune response is triggered. T-lymphocytes (CD4+ and CD8+) migrate to the site of infection and surround the infected macrophages, forming a structured aggregate called a granuloma [3]. Inside this structure, the bacteria are contained but not necessarily eradicated. They enter a state of dormancy, downregulating their metabolic activity to survive in the hypoxic, nutrient-poor environment of the granuloma's core.
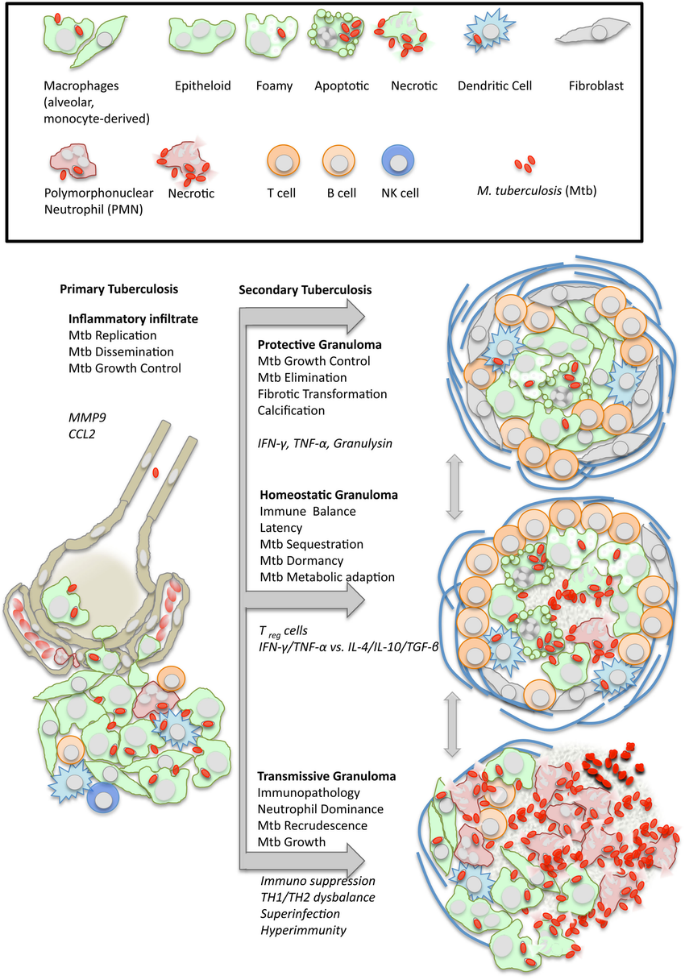
Figure 1. Diagram of a tuberculous granuloma. This organized structure consists of infected macrophages (often fusing to form giant cells) surrounded by a cuff of lymphocytes. It serves to contain the infection, preventing dissemination while allowing the bacteria to persist in a latent state.
(Source: Ramakrishnan L. (2012). Revisiting the role of the granuloma in tuberculosis. Nature Reviews Immunology, 12(5), 352–366).
2.2 The Spectrum of Infection
Recent research suggests that "latency" and "active disease" are not binary opposites but rather ends of a continuous spectrum. Some individuals may have "incipient TB" or "subclinical TB," where there is low-level bacterial replication and tissue damage without overt symptoms. This nuance explains why some "latent" patients may still progress to active disease despite having a robust immune response [3].
3. Diagnosis of LTBI
Diagnosing LTBI is challenging because there is no "gold standard" test that can directly detect the dormant bacteria. Instead, we must rely on indirect tests that measure the host's immune sensitization to mycobacterial antigens.
3.1 The Tuberculin Skin Test (TST)
The Mantoux TST has been used for over a century. It involves the intradermal injection of Purified Protein Derivative (PPD).
v Mechanism: In a sensitized person, memory T-cells migrate to the site of injection and release cytokines, causing induration (hardening) at 48–72 hours [4].
v Limitations: The PPD contains antigens shared with the BCG vaccine and non-tuberculous mycobacteria. This leads to frequent false positives in BCG-vaccinated populations (cross-reactivity). Conversely, false negatives (anergy) can occur in severely immunosuppressed patients who cannot mount an immune response [2].
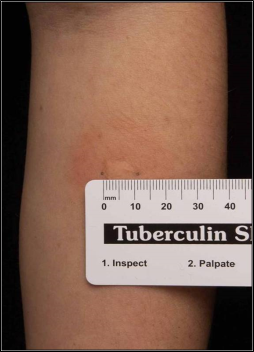
Figure 2. A positive Mantoux Tuberculin Skin Test (TST). The reaction is characterized by induration (palpable raised area) at the injection site. The size of induration required for a positive result varies by risk group (e.g., ≥5mm for HIV patients, ≥10mm for recent immigrants).
(Source: CDC Public Health Image Library, ID #6722).
3.2 Interferon-Gamma Release Assays (IGRA)
IGRAs (e.g., QuantiFERON-TB Gold, T-SPOT.TB) are in vitro blood tests.
v Mechanism: Patient blood is mixed with specific M.tb antigens (ESAT-6 and CFP-10). If the patient has been infected, their T-cells will release Interferon-Gamma (IFN-γ) in response.
v Advantage: These specific antigens are absent from the BCG vaccine. Therefore, IGRAs are highly specific and do not show false positives in vaccinated individuals. They are the preferred test in most high-income settings and for anyone with a history of BCG vaccination [4].
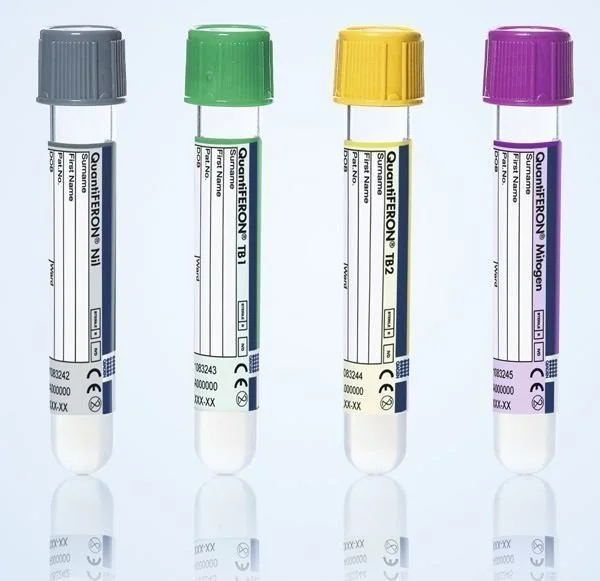
Figure 3. The QuantiFERON-TB Gold Plus collection tubes. This IGRA test measures the cell-mediated immune response to specific TB antigens. Unlike the skin test, it requires a single patient visit and is not affected by prior BCG vaccination.
(Source: Qiagen, QuantiFERON-TB Gold Plus Product Guide).
4. Treatment of Latent Infection
The decision to treat LTBI is a risk-benefit calculation. We are treating a healthy person with potentially toxic drugs to prevent a future event. Therefore, treatment is prioritized for those with the highest risk of reactivation (e.g., HIV positive, recent contacts, organ transplant recipients).
4.1 Historical Standard: Isoniazid Monotherapy
For decades, the standard of care was Isoniazid (INH) taken daily for 6 or 9 months (6H or 9H). While highly effective (90% risk reduction), the long duration leads to poor adherence. Additionally, INH carries a risk of hepatotoxicity, which increases with age [5].
4.2 The Modern Shift: Short-Course Rifamycin Regimens
To improve completion rates, guidelines now favor shorter regimens containing Rifamycins (Rifampicin or Rifapentine).
v 3HP: A combination of Isoniazid and Rifapentine taken once weekly for 3 months (12 doses total). This regimen is as effective as 9 months of INH but has much higher completion rates and less liver toxicity.
v 4R: Rifampicin taken daily for 4 months. This is a preferred alternative for patients who cannot tolerate Isoniazid or in settings with high rates of Isoniazid-resistant TB [5].
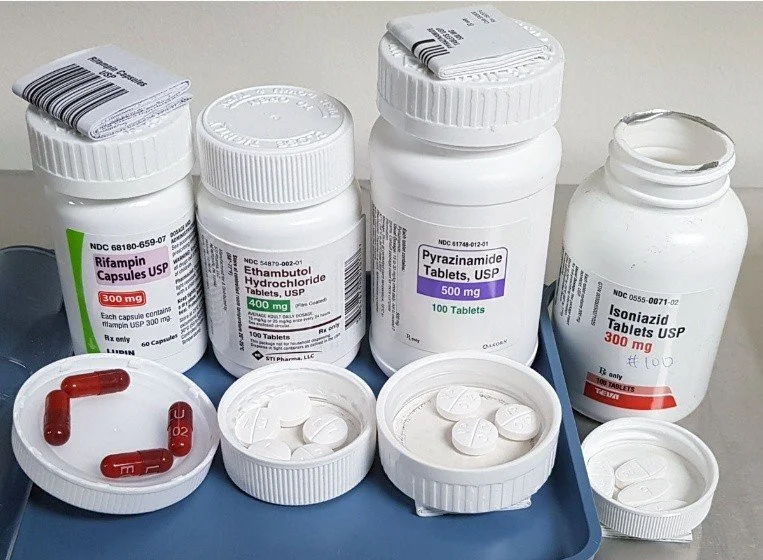
Figure 4. The components of the 3HP Regimen for Latent TB Infection. This short-course therapy requires the combination of Isoniazid (INH) and Rifapentine (RPT) taken once weekly for 12 weeks.
(Source: Centers for Disease Control and Prevention (CDC), Division of Tuberculosis Elimination, 3HP Treatment Regimen for Latent TB Infection, 2020).
5. Conclusion
Latent Tuberculosis Infection is the silent engine of the global TB epidemic. As long as billions of people harbor the dormant bacillus, the threat of reactivation remains. The transition from the non-specific Tuberculin Skin Test to the highly specific IGRA has improved our ability to accurately identify those truly at risk. Furthermore, the advent of short-course preventive therapies like 3HP has transformed LTBI treatment from a burdensome 9-month ordeal into a manageable 12-dose course. Eliminating TB will require not just treating the sick, but effectively neutralizing this vast, silent reservoir.
6. References
[1] World Health Organization. (2018). Latent tuberculosis infection: updated and consolidated guidelines for programmatic management. Geneva: WHO.
[2] Getahun, H., Mattelli, A., Abubakar, I., et al. (2015). Management of latent Mycobacterium tuberculosis infection: WHO guidelines for low tuberculosis burden countries. European Respiratory Journal, 46(6), 1563–1594.
[3] Barry, C. E., 3rd, Boshoff, H. I., Dartois, V., et al. (2009). The spectrum of latent tuberculosis: rethinking the biology and intervention strategies. Nature Reviews Microbiology, 7(12), 845–855. https://doi.org/10.1038/nrmicro2236
[4] Pai, M., Zwerling, A., & Menzies, D. (2008). Systematic review: T-cell-based assays for the diagnosis of latent tuberculosis infection: an update. Annals of Internal Medicine, 149(3), 177–184.
[5] Sterling, T. R., Njie, G., Zenner, D., et al. (2020). Guidelines for the Treatment of Latent Tuberculosis Infection: Recommendations from the National Tuberculosis Controllers Association and CDC, 2020. MMWR Recommendations and Reports, 69(1), 1–11. https://doi.org/10.15585/mmwr.rr6901a1
