Diagnostics in Tuberculosis
1. Kurmanaliev Nurlan K.
2. Akre Sakshi Sanjay
Pawan Anant Gayakwad
Aslam Mustehasin
Daniyal Mohammad
Kumar Shubham
(1. Teacher, International Medical Faculty, Osh State University, Kyrgyzstan)
https://orcid.org/0000-0002-5952-1463
(2. Students, International Medical Faculty, Osh State University, Kyrgyzstan)
Abstract
Tuberculosis (TB), caused by the bacillus Mycobacterium tuberculosis, remains one of the leading causes of infectious mortality worldwide. The diagnosis of TB poses a unique challenge to the clinician because the pathogen is slow-growing, paucibacillary in early stages, and capable of mimicking various other pathologies. This article provides a comprehensive review of the current diagnostic landscape. We examine the foundational role of anamnesis and physical examination, recognizing the "classic" constitutional symptoms. We then explore the evolution of laboratory methods, from the century-old Ziehl-Neelsen smear to the gold-standard liquid culture and the revolutionary GeneXpert MTB/RIF molecular assay. Furthermore, the role of radiological imaging, immunodiagnostics (IGRA and TST), and invasive surgical procedures for extrapulmonary cases is discussed. The integration of these modalities is essential for early detection, breaking the chain of transmission, and preventing the emergence of drug-resistant strains.
Keywords: Tuberculosis, Mycobacterium tuberculosis, GeneXpert, Ziehl-Neelsen Staining, IGRA, Chest Radiography, Latent TB Infection.
1. Introduction
Despite being a curable disease, Tuberculosis (TB) continues to claim over 1.3 million lives annually [1]. The causative agent, Mycobacterium tuberculosis (M.tb), is a master of evasion. Its thick, lipid-rich cell wall not only makes it resistant to many antibiotics but also allows it to survive within the harsh environment of human macrophages.
For the clinician, diagnosing TB is often a race against time. A delayed diagnosis allows the patient to remain infectious, spreading the bacteria to family and community members. Historically, diagnosis relied heavily on the stethoscope and the microscope. Today, however, the field has been transformed by molecular diagnostics that can identify genetic material and drug resistance profiles in less than two hours. Yet, technology cannot replace clinical judgment [2]. Understanding the nuances of each diagnostic modality—from the humble skin test to advanced bronchoscopy—is critical for accurate patient management.
2. Clinical Assessment: Anamnesis and Physical Examination
Before any sample reaches the laboratory, the diagnosis begins at the bedside. The value of a thorough history (anamnesis) cannot be overstated, particularly in distinguishing active TB from other respiratory conditions like pneumonia or malignancy.
2.1 Anamnesis: The Narrative of Disease
The presentation of pulmonary TB is often insidious (slow-onset). Patients rarely present with acute sepsis; instead, they describe a "wasting away" process—historically termed "consumption."
Systemic "B-Symptoms": The hallmark of active TB includes low-grade fever (often rising in the late afternoon), drenching night sweats that require changing bedclothes, and significant, unintentional weight loss [2].
Respiratory Symptoms: A chronic cough lasting more than 2-3 weeks is the primary red flag. Initially dry, it may become productive with purulent sputum. Hemoptysis (coughing up blood) is a late and alarming sign, indicating tissue destruction and cavitation into pulmonary blood vessels.
Epidemiological Risk: A crucial part of the history is social context. Clinicians must ask about contacts with known TB patients, recent incarceration, homelessness, or travel to high-burden countries [1].
2.2 Physical Examination
Physical findings in TB can be deceptively subtle. In early disease, the chest exam may be entirely normal. As the disease progresses, auscultation may reveal apical crackles (rales) or bronchial breathing sounds, particularly in the upper lobes where the high oxygen tension favors bacterial growth. In cases of pleural effusion, one may find dullness to percussion and decreased breath sounds. Importantly, the exam must extend beyond the lungs. Lymphadenopathy (swollen lymph nodes), particularly in the cervical chain (scrofula), is a common manifestation of extrapulmonary TB [2].
3. Laboratory Diagnosis: The Microbiological Pillars
Demonstrating the presence of the organism remains the cornerstone of diagnosis.
3.1 Microscopy: The First Line of Defense
For over 100 years, the Ziehl-Neelsen (ZN) stain has been the primary method for detecting acid-fast bacilli (AFB). Because the waxy cell wall of M.tb retains dye even after being washed with acid-alcohol, the bacteria appear as bright red rods against a blue background [5].
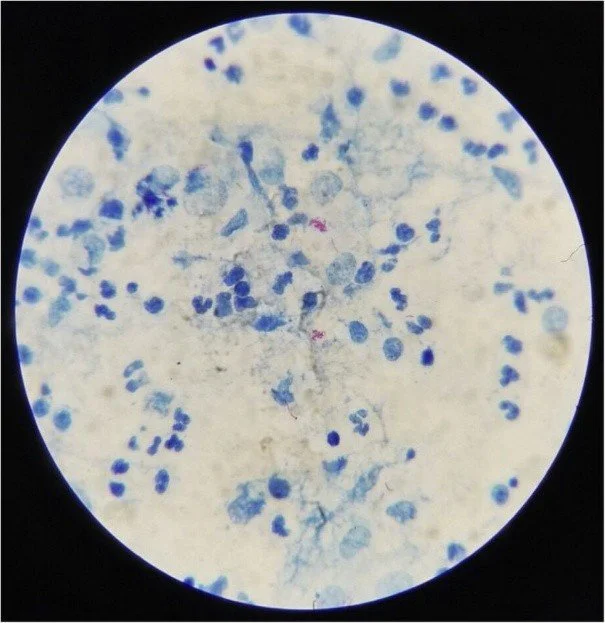
Figure 1. Photomicrograph of a sputum smear stained with Ziehl-Neelsen stain. The acid-fast bacilli (Mycobacterium tuberculosis) appear as bright red rods against a methylene blue background. This "red snapper" appearance is diagnostic for active TB.
(Source: Centers for Disease Control and Prevention (CDC), Public Health Image Library (PHIL), ID #5789).
3.2 Culture: The Gold Standard
Culturing the bacteria is the only way to definitively confirm diagnosis and test for drug susceptibility.
Solid Media (Lowenstein-Jensen): The classic "egg-based" medium. M.tb grows as rough, buff-colored colonies. However, because the bacteria divide so slowly (every 18-24 hours), it can take 4-8 weeks to see growth [2].
Liquid Media (MGIT - Mycobacteria Growth Indicator Tube): This is the modern standard. It uses automated systems to detect oxygen consumption by growing bacteria. It is significantly faster (1-3 weeks) and more sensitive than solid media [3].
3.3 Molecular Diagnostics (PCR): The Game Changer
The introduction of GeneXpert MTB/RIF has revolutionized TB care. This is a cartridge-based nucleic acid amplification test (NAAT).
Mechanism: It detects the specific DNA of M.tb and, crucially, detects mutations in the rpoB gene that confer resistance to Rifampicin (a marker for Multidrug-Resistant TB or MDR-TB).
Impact: It provides results in 2 hours with high sensitivity, allowing patients to be isolated and started on the correct regimen immediately. WHO now recommends this as the initial diagnostic test for all suspected cases [3].
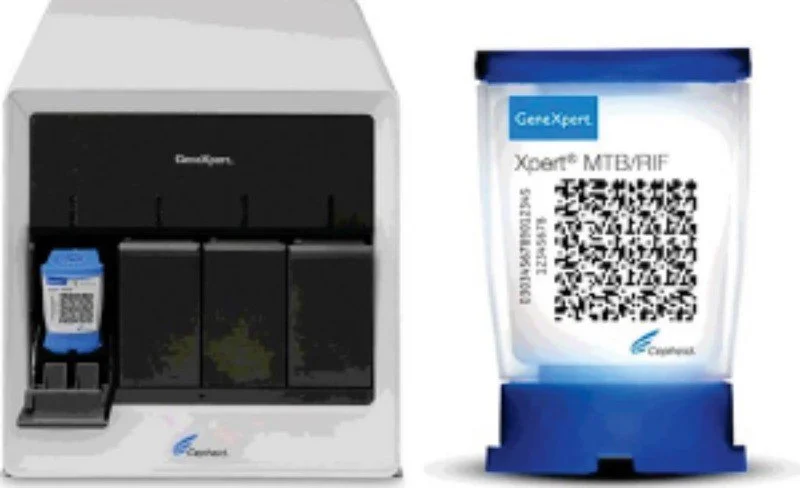
Figure 2. The GeneXpert MTB/RIF system and cartridge. This automated nucleic acid amplification test (NAAT) detects specific DNA sequences of Mycobacterium tuberculosis and mutations associated with rifampicin resistance within 2 hours.
(Source: World Health Organization (WHO), Automated Real-Time Nucleic Acid Amplification Technology for Rapid and Simultaneous Detection of Tuberculosis and Rifampicin Resistance, 2011).
4. Immunodiagnostics and Tuberculin Diagnostics
These tests detect the host's immune response to the bacteria, rather than the bacteria itself. They are primarily used to diagnose Latent TB Infection (LTBI) but can support the diagnosis of active TB in culture-negative cases.
4.1 The Tuberculin Skin Test (TST / Mantoux)
This method involves the intradermal injection of Purified Protein Derivative (PPD). If the patient has been sensitized to TB antigens, a T-cell mediated hypersensitivity reaction (Type IV) occurs, causing induration (hardening) at the site after 48-72 hours.
Limitation: It has poor specificity. False positives are common in patients vaccinated with BCG (Bacille Calmette-Guérin) or those exposed to non-tuberculous mycobacteria [4].
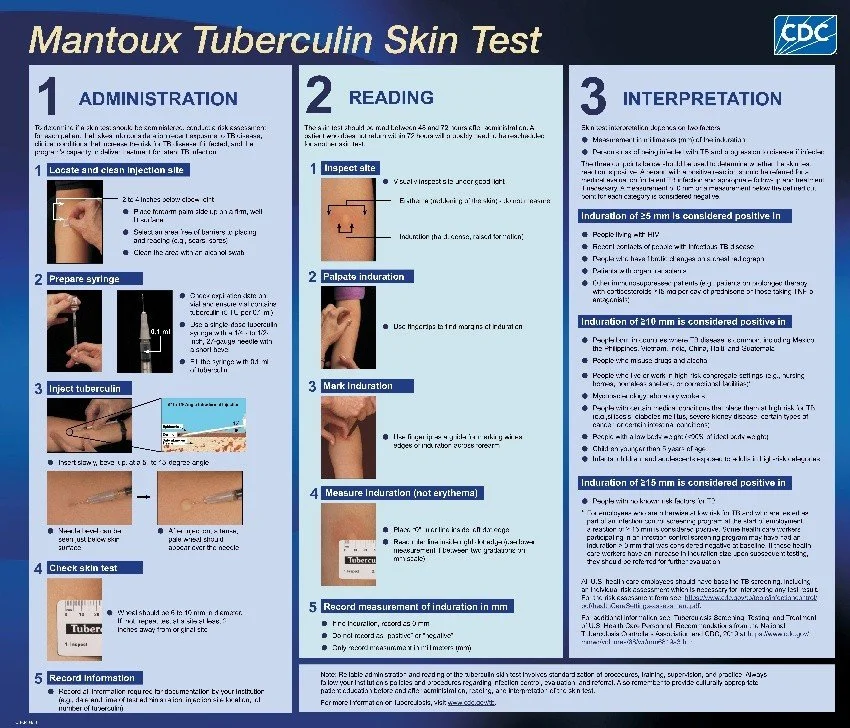
Figure 3. Interpretation of the Mantoux Tuberculin Skin Test (TST). The reaction is read 48–72 hours after injection. The clinician must measure the diameter of induration (palpable raised hardening) transverse to the long axis of the forearm, ignoring erythema (redness).
(Source: CDC Division of Tuberculosis Elimination, Core Curriculum on Tuberculosis: What the Clinician Should Know, 2021).
4.2 Interferon-Gamma Release Assays (IGRA)
Commercial tests like QuantiFERON-TB Gold are blood tests that measure the release of interferon-gamma by white blood cells when exposed to specific M.tb antigens.
Superiority: Unlike the TST, these antigens are not found in the BCG vaccine. Therefore, IGRA is not affected by prior vaccination, making it far more specific and the preferred test in many developed nations [4].
5. Instrumental and Imaging Methods
While microbiology confirms the diagnosis, imaging reveals the extent of the damage.
5.1 Chest Radiography (CXR)
The chest X-ray is the primary screening tool.
Primary TB: Often presents with hilar lymphadenopathy and lower lobe infiltrates (Ghon complex).
Post-Primary (Reactivation) TB: Classically involves the upper lobes (apical or posterior segments). The hallmark finding is cavitation—a thick-walled hollow space formed by caseous necrosis. This is highly suggestive of infectiousness [2].
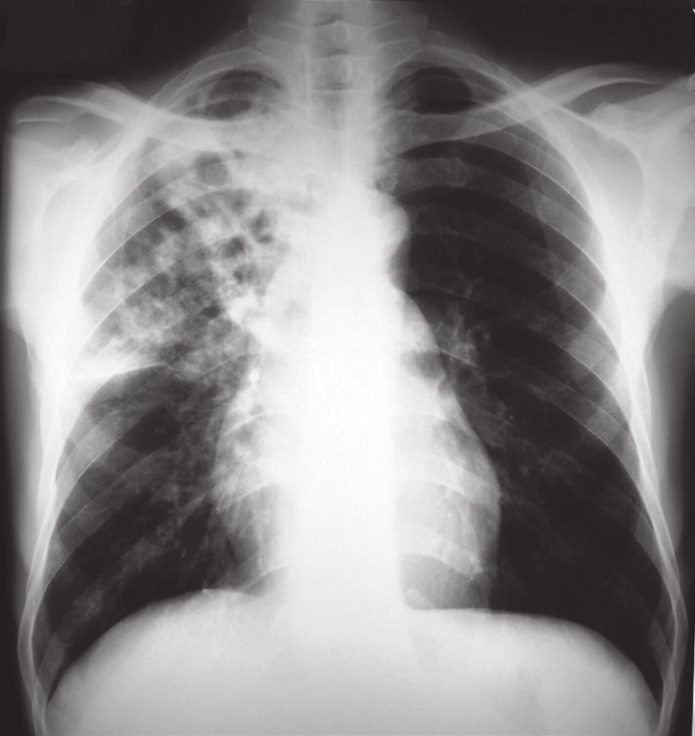
Figure 4. Anteroposterior (AP) chest radiograph of a patient with advanced pulmonary tuberculosis. The image reveals a large cavity in the right apical region (upper lung field), indicated by the arrow. This finding is characteristic of post-primary (reactivation) TB and indicates high infectivity.
(Source: Centers for Disease Control and Prevention (CDC), Public Health Image Library (PHIL), ID #14805, 1972).
5.2 Computed Tomography (CT)
A chest CT is more sensitive than an X-ray. It is particularly useful for detecting:
Subtle, early cavitation ("tree-in-bud" appearance indicative of endobronchial spread).
Miliary TB (tiny, millet-seed sized nodules spread throughout the lung).
Complications like pleural empyema or pericardial involvement [2].
6. Surgical and Invasive Methods
In complex cases—especially when patients cannot produce sputum or when the disease is extrapulmonary—invasive procedures become necessary.
6.1 Bronchoscopy and Bronchoalveolar Lavage (BAL)
If a patient is "sputum negative" but clinically suspected of having TB, a flexible bronchoscope is passed into the lungs. Saline is instilled and suctioned back (lavage) to harvest cells and bacteria from the lower respiratory tract for PCR and culture.
6.2 Biopsy (Histopathology)
For extrapulmonary TB, tissue is the issue.
Pleural Biopsy: Essential for diagnosing TB pleurisy, as pleural fluid alone often lacks bacteria.
Lymph Node Biopsy: Fine Needle Aspiration (FNA) of a swollen neck node can show caseating granulomas—the microscopic hallmark of TB [5].
VATS (Video-Assisted Thoracoscopic Surgery): In rare cases where the diagnosis remains elusive, surgical exploration of the chest may be required to obtain adequate tissue samples.
7. Conclusion
The diagnosis of tuberculosis has evolved from the era of percussion and staining to the age of molecular genetics. While the GeneXpert assay and liquid culture systems represent the pinnacle of diagnostic science, they rely on the clinician's initial suspicion. A comprehensive diagnostic approach—combining a detailed anamnesis, rigorous physical examination, appropriate imaging, and the judicious use of modern laboratory tools—is essential. Only through rapid and accurate diagnosis can we hope to interrupt the chain of transmission and turn the tide against this ancient pathogen.
8. References
[1] World Health Organization (WHO). Global Tuberculosis Report 2023. Geneva: WHO; 2023.
[2] Jameson, J. L., Fauci, A. S., Kasper, D. L., et al. Harrison's Principles of Internal Medicine. 21st ed. New York: McGraw Hill; 2022. ISBN: 978-1264268504.
[3] World Health Organization. WHO consolidated guidelines on tuberculosis: Module 3: Diagnosis - Rapid diagnostics for tuberculosis detection. Geneva: WHO; 2021.
[4] Lewinsohn, D. M., Leonard, M. K., LoBue, P. A., et al. Official American Thoracic Society/IDSA/CDC Clinical Practice Guidelines: Diagnosis of Tuberculosis in Adults and Children. Clinical Infectious Diseases, 64(2), e1–e33. https://doi.org/10.1093/cid/ciw694
[5] Murray, P. R., Rosenthal, K. S., & Pfaller, M. A. Medical Microbiology. 9th ed. Philadelphia: Elsevier; 2020. ISBN: 978-0323673228.
