Prevention of Tuberculosis
1. Kurmanaliev Nurlan K.
2. Mohd Mazid Khan
Mohammad Sajid
Salma Parween
Shah Mairaj Ali
Siddiqui Mohd. Zeeshan
(1. Teacher, International Medical Faculty, Osh State University, Kyrgyzstan)
https://orcid.org/0000-0002-5952-1463
(2. Students, International Medical Faculty, Osh State University, Kyrgyzstan)
Abstract
The prevention of Tuberculosis (TB) is a complex, multi-layered challenge that extends far beyond the administration of a single vaccine. While the Bacille Calmette-Guérin (BCG) vaccine remains the most widely used immunization in history, its efficacy is variable, necessitating a broader preventive strategy. This article reviews the three pillars of TB prevention: specific immunoprophylaxis via BCG, chemoprophylaxis (preventive treatment) for latent infection, and social prevention through infection control and socioeconomic improvement. We critically analyze the complications of BCG vaccination, ranging from common local reactions to rare disseminated disease, and discuss the modern shift towards short-course preventive regimens (e.g., 3HP) for high-risk populations. Ultimately, we argue that while biological tools are essential, the eradication of TB requires addressing the social determinants—poverty, overcrowding, and malnutrition—that fuel the epidemic.
Keywords: BCG Vaccine, Chemoprophylaxis, Latent Tuberculosis Infection (LTBI), BCG Lymphadenitis, Social Determinants of Health, Infection Control.
1. Introduction
Tuberculosis is unique among infectious diseases in that infection does not equal disease. Of the estimated 2 billion people infected with Mycobacterium tuberculosis worldwide, only 5–10% will develop active disease. The goal of prevention, therefore, is twofold: first, to prevent transmission from infectious patients to healthy individuals (breaking the chain of transmission); and second, to prevent the progression of latent infection to active disease in those already infected [1].
In high-burden settings like Kyrgyzstan and India, this requires a strategy that integrates pediatric vaccination, aggressive management of contacts, and broad social interventions [3].
2. Specific Prevention: The BCG Vaccine
The Bacille Calmette-Guérin (BCG) vaccine, developed a century ago from an attenuated strain of Mycobacterium bovis, remains the only licensed vaccine for TB.
2.1 Mechanism and Efficacy
BCG does not prevent primary infection. A child vaccinated with BCG can still be infected if they breathe in the bacteria. However, BCG is highly effective (60–80%) at preventing the hematogenous spread of the bacteria. This means it acts as a "shield" against the most lethal forms of pediatric TB: Miliary Tuberculosis and TB Meningitis [1].
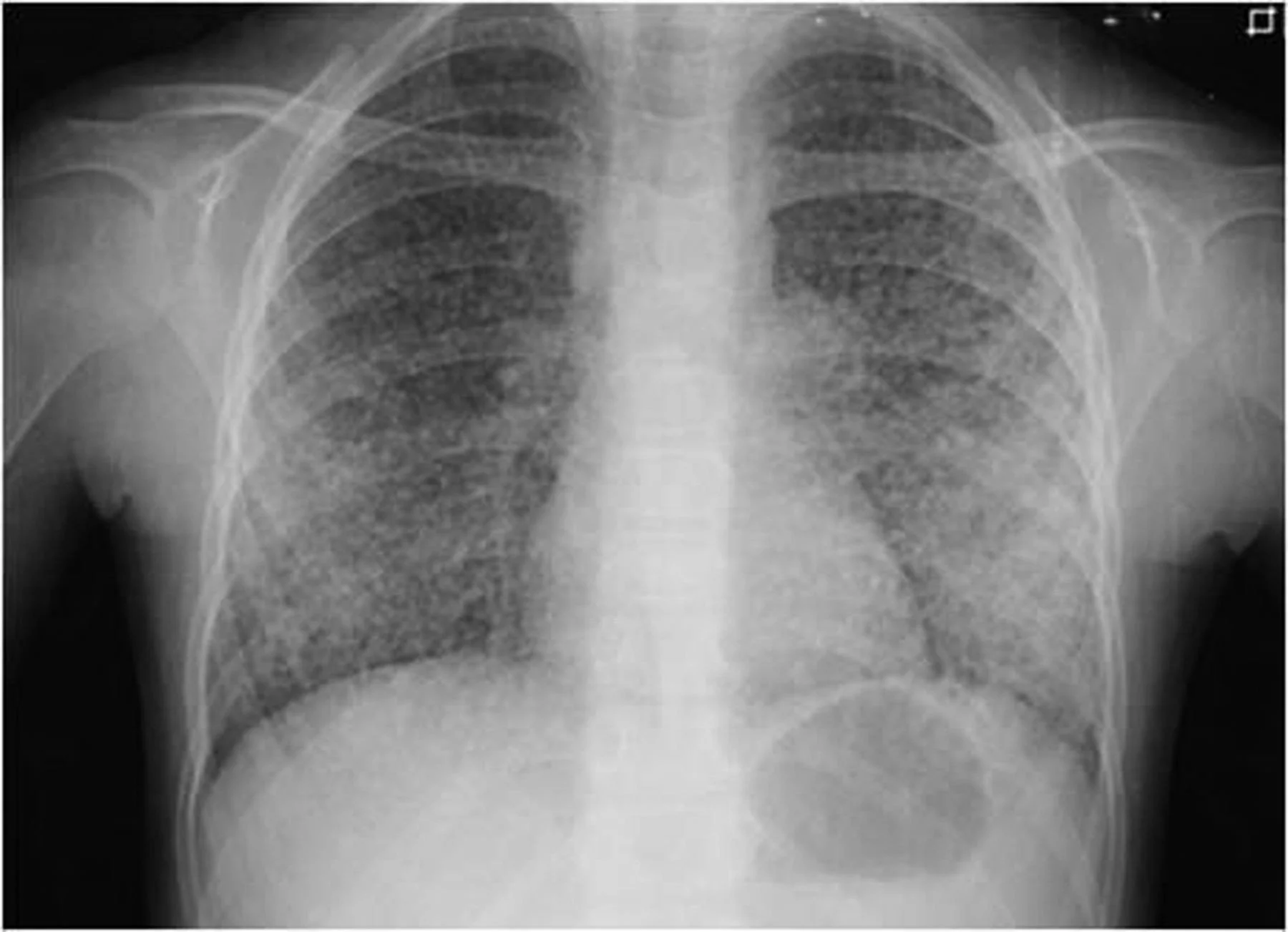
Figure 1. Chest radiograph of a pediatric patient with Miliary Tuberculosis. Note the diffuse, millet-seed-sized nodules throughout both lung fields. This is the specific lethal form of the disease that the BCG vaccine effectively prevents.
(Source: Centers for Disease Control and Prevention (CDC), Public Health Image Library, ID #3428).
2.2 Current Recommendations
Because its protection wanes in adolescence and is variable in adults (ranging from 0% to 80% efficacy depending on geography), the WHO recommends BCG primarily for neonates in high-burden countries. It is given as a single intradermal dose, typically on the left deltoid, as soon as possible after birth [3].
3. Complications of BCG Vaccination
While generally safe, BCG is a live bacterial vaccine, and complications can occur. These are classified into local, regional, and disseminated.
3.1 Normal Reaction vs. Local Complications
A "normal" reaction follows a predictable timeline: a papule appears at 2–3 weeks, ulcerates at 6 weeks, and heals with a scar by 3 months.
BCG Abscess: If the vaccine is injected subcutaneously rather than intradermally, or if the dose is too high, a "cold abscess" may form. It is a painless, fluctuant swelling that may persist for months [2].
Keloid Formation: An exaggerated scar tissue response, often causing cosmetic concern but no medical danger.
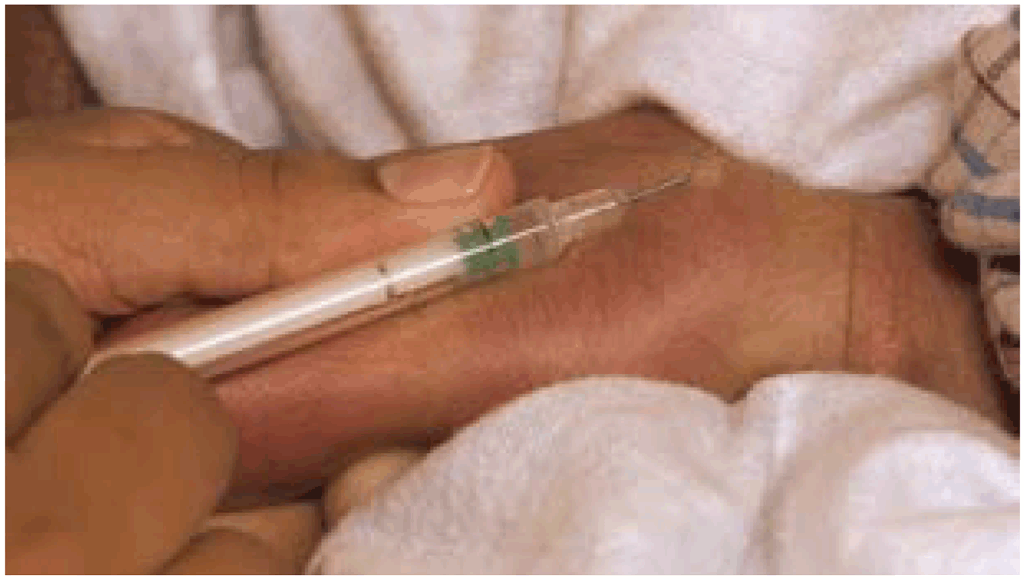
Figure 2. Proper technique for intradermal administration of the BCG vaccine. The formation of a "wheal" (a raised, blanched bump like an orange peel) confirms correct placement. Deep subcutaneous injection is a common cause of abscess formation.
(Source: CDC Public Health Image Library (PHIL), ID #1228).
3.2 Regional Lymphadenitis (BCGitis)
This is the most common complication, occurring in <1% of vaccinations. It involves the enlargement of the ipsilateral (same side) axillary or supraclavicular lymph nodes.
Non-suppurative: The node is hard but not red. This usually resolves spontaneously and should not be treated with antibiotics or surgery.
Suppurative: The node softens, turns red, and may rupture (sinus formation). Needle aspiration may be required to prevent spontaneous rupture [2].
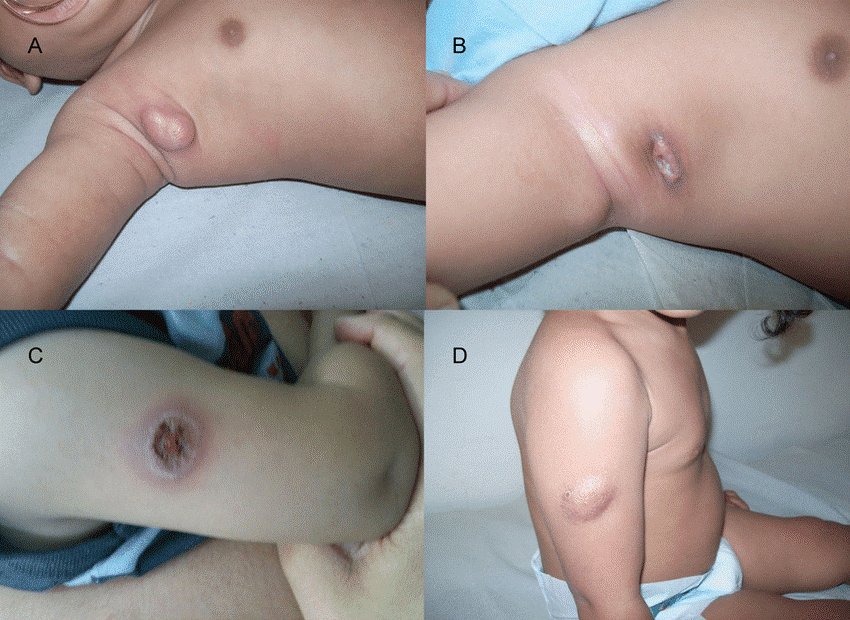
Figure 3. BCG Lymphadenitis in an infant. Note the enlarged, erythematous axillary lymph node on the same side as the vaccination. This complication, often called "BCGitis," can progress to suppuration (pus formation).
(Source: Wikimedia Commons, Case courtesy of Dr. James Heilman, MD).
3.3 Disseminated BCG Disease (BCGosis)
This is a rare but life-threatening complication (occurring in ~1 per million doses) where the vaccine strain spreads throughout the body. It occurs almost exclusively in infants with Severe Combined Immunodeficiency (SCID) or HIV. This risk is why BCG is contraindicated in symptomatic HIV-positive infants [2].
4. Chemoprophylaxis: Treating Latent Infection
The second pillar of prevention is Tuberculosis Preventive Treatment (TPT). The logic is simple: if we kill the dormant bacteria in a person with Latent TB Infection (LTBI), they can never develop active contagious TB.
4.1 Target Populations
We do not treat everyone. Priority is given to those at highest risk of progression:
People Living with HIV (PLHIV): The risk of progression is 10% per year (compared to 10% per lifetime in HIV-negative people) [3].
Child Contacts (<5 years): Children living with a smear-positive adult are at extremely high risk of disseminated disease. They receive "Window Prophylaxis" regardless of test results until infection is ruled out.
Clinical Risk Groups: Patients starting anti-TNF therapy (e.g., for rheumatoid arthritis), dialysis patients, and organ transplant recipients [3].
4.2 Regimens
Isoniazid Monotherapy (IPT): The traditional "gold standard." Daily Isoniazid for 6 or 9 months (6H or 9H). It is effective but has poor compliance due to the long duration.
Short-Course Rifamycin Regimens: The modern standard.
3HP: Isoniazid + Rifapentine taken once weekly for 3 months (12 doses total).
4R: Rifampicin daily for 4 months.
3HR: Isoniazid + Rifampicin daily for 3 months (often used in children) [3].
5. Social Prevention: The "Social Vaccine"
TB is famously a disease of poverty. Virchow, the father of pathology, famously stated that "Medicine is a social science, and politics is nothing else but medicine on a large scale."
5.1 Breaking the Cycle at the Source
The most effective way to prevent TB is to cure infectious cases quickly. An untreated smear-positive patient can infect 10–15 people in a year. Early diagnosis (via GeneXpert) acts as prevention for the community [4].
5.2 Infection Control in Healthcare
Hospitals are major amplification sites. Prevention relies on administrative controls (triage), environmental controls, and respiratory protection.
Ventilation: TB bacteria are airborne. High air exchange rates (natural ventilation or negative pressure rooms) dilute the concentration of infectious droplet nuclei.
UVGI: Ultraviolet Germicidal Irradiation lights can kill airborne bacteria in the upper room air [4].
Figure 4. Signage for an Airborne Infection Isolation Room (AIIR). Social and environmental prevention relies on engineering controls like negative pressure ventilation to prevent the escape of infectious droplet nuclei into corridors.
(Source: CDC/National Institute for Occupational Safety and Health (NIOSH)).
5.3 Social Determinants
Overcrowding and poor ventilation in housing are the primary drivers of transmission. Malnutrition weakens the immune system, allowing latent infection to become active. Therefore, poverty alleviation, food security programs, and improved urban housing standards are, in reality, powerful "anti-TB vaccines" [4].
6. Conclusion
Preventing tuberculosis requires a delicate balance between biological interventions and social reform. The BCG vaccine protects the most vulnerable children from death, while chemoprophylaxis sterilizes the reservoir of latent infection in high-risk adults. However, neither of these medical tools can succeed in isolation. True prevention implies a social commitment to identifying infectious cases early, protecting the uninfected through environmental control, and lifting populations out of the conditions of poverty that allow the bacillus to thrive.
7. References
[1] Trunz, B. B., Fine, P., & Dye, C. (2006). Effect of BCG vaccination on childhood tuberculous meningitis and miliary tuberculosis worldwide: a meta-analysis and assessment of cost-effectiveness. The Lancet, 367(9517), 1173–1180.
[2] Ritz, N., & Curtis, N. (2018). The susceptibility of infants to complications of BCG vaccination. Frontiers in Immunology, 9, 1318. https://doi.org/10.3389/fimmu.2018.01318
[3] World Health Organization. (2020). WHO consolidated guidelines on tuberculosis: Module 1: prevention - tuberculosis preventive treatment. Geneva: WHO.
[4] Hargreaves, J. R., Boccia, D., Evans, C. A., et al. (2011). The social determinants of tuberculosis: from evidence to action. American Journal of Public Health, 101(4), 654–662.
