Lymphogranuloma Venereum
1. Chaitanya Vasant Gondkar
2. Kaveri Santosh Batwal
3. Samatbek Turdaliev
(1. Student, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.
2. Student, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.
3. Teacher, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.)
Abstract
Lymphogranuloma venereum is a sexually transmitted disease caused by L1, L2, and L3 serovars of Chlamydia trachomatis. In the last 10 years outbreaks have appeared in North America, Europe, and Australia in the form of proctitis among men who have sex with men.
Three stages of disease have been described. The disease in primary stage may go undetected when only a painless papule, pustule, or ulceration appears. The diagnosis is difficult to establish on clinical grounds alone and frequently relies upon either serologic testing, culture, or more recently, nucleic acid amplification testing of direct specimens. A proper treatment regimen cures the infection and prevents further damage to tissues. Lymphogranuloma venereum causes potentially severe infections with possibly irreversible sequels if adequate treatment is not begun promptly. Early and accurate diagnosis is essential.
Doxycycline is the drug of choice. Pregnant and lactating women should be treated with
erythromycin or azithromycin. Patient must be followed up during the treatment, until disease signs and symptoms have resolved. Repeated testing for syphilis, hepatitis B and C, and HIV to detect early infection should be performed.
Keywords: sexually transmitted disease, Chlamydia trachomatis, early and accurate diagnosis
INTRODUCTION
Lymphogranuloma venereum (LGV) is a sexually transmitted disease (STD) caused byL1, L2, and L3 serovars of Chlamydia trachomatis that primarily infects the lymphatics and can be transmitted through unprotected vaginal, anal, or oral sexual contact.C. trachomatis is the most common cause of bacterial STDs in both men and women.
C. trachomatis is an obligate intracellular gram-negative bacterium that during its developmental cycle alternates between two forms: the infectious elementary body (EB) and the noninfectious replicating form, the reticulate body. The attachment process of the elementary body to a host cell is the event most crucial to a successful infection. Elementary bodies attach to columnar epithelial cells followed by endocytosis and inhibition of lysosomal fusion. A number of chlamydial ligands have been identified and characterized. These include the major outer membrane protein (MOMP) as well as the cysteine-rich OmcB (Omp2) protein, hsp70, the polymorphic outer membrane proteins, and the thermolabile 34-kDa membrane protein. In addition to these proposed ligands, there is a considerable amount of experimental evidence to suggest that the glycosaminoglycan, heparan sulfate, is involved in the chlamydial attachment- infectivity process. C. trachomatis does not produce heparan sulfate. Heparan sulfate acts as a host cell receptor for MOMP.2 The disease was described in 1833 by W allace, but it was defined as a clinical and pathological entity in 1913 by Durand, Nicolas, and Favre.
EPIDEMIOLOGY
LGV probably affects both sexes equally, although it is more commonly reported in men because early manifestations of LGV are more apparent in men. Men typically present with the acute form of the disease, whereas women often present when they develop complications from later stages of the disease.
LGV may appear at any age, but the highest incidence is between 15 and 40 years (sexually active population).Most cases in Europe and North America have been identified among white, frequently HIV-positive MSM patients presenting with proctitis. Since 2003, there have been a series of LGV outbreaks reported across Europe.10–13 Since formal surveillance was launched in 2004, the United Kingdom has seen the highest number of confirmed MSM cases globally.13 Between April 2003 and June 2012, more than 2,000 cases of LGV had been confirmed in the United Kingdom.12 A large increase in diagnosed cases occurred in late 2009, peaking at 150 cases per quarter in mid-2010; since then, UK rates have remained steady at approximately 80 cases per quarter until 2012.
Seventy-seven percent of cases have been diagnosed in London, Brighton, and Manchester. In one report from the United Kingdom (outbreak of 327 new cases of LGV), 76% MSM were also HIV positive, 39% had a diagnosis of 40 submit your manuscript another STD, and also infected with hepatitis C virus. This is a major public health concern because enhanced shedding of HIV during clinical proctitis could increase the risk of HIV transmission to uninfected men.
Before the outbreaks in MSM, LGV was primarily endemic in heterosexuals in areas of East and West Africa, India, parts of Southeast Asia, and the Caribbean where it is manifested as the classic form of disease with genital ulcers and lymphadenopathy (without proctitis). In these regions, a survey from Madagascar showed that C. trachomatis is not the main cause of genital ulcers. In that survey, 76% of 196 patients with genital ulcers had chlamydial antibodies, but only 8% of patients had LGV confirmed by multiplex polymerase chain reaction (PCR).
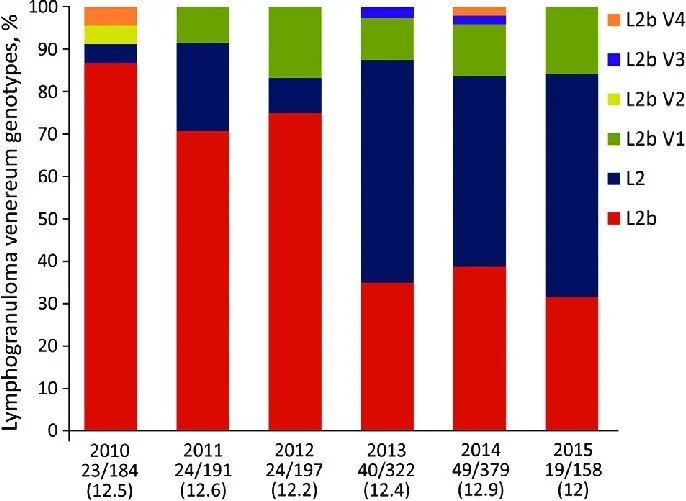
PATHOPHYSIOLOGY
C. trachomatis is divided into 15 serovars (A, B, Ba, C–K, and L1–L3) based on analysis of the MOMP. Serovars L1–L3 cause LGV. The L2 serovar can be further separated into L2,L2′, L2a, or L2b according to minor differences in their component amino acids. LGV is predominantly a disease of the lymphatic tissue.6 In contrast to serovars A–K, which remain confined to the mucosa, the LGV serovars C. trachomatis induce a lymphoproliferative reaction, it gains entrance through skin breaks and abrasions or it crosses the epithelial cells of mucous membranes. LGV serovars and other strains of C. trachomatis appear to bind to epithelial cells via heparan sulfate receptors and then it travels via lymphatics to multiply within mononuclear phagocytes in regional lymph nodes. After lymphangitis, areas of necrosis occur within the nodes, followed by the formation of abscess.
In one study of 12 patients, lymph node macrophages contained organisms that stained black
with W arthin–Starry stain, the organisms were clustered within vacuoles and both reticulate and elementary bodies were visible by electron microscopy. PCRtesting of the lymph nodes for LGV serovars of C. trachomatis was positive in 9/12 cases. The pathologic findings are compatible with LGV or Bartonella henselae (cat scratch disease), but are not diagnostic of either entity. The lymph node reaction may take several weeks to develop and can result in substantial inflammation and subsequent fibrosis.
In a small number of cases, dissemination and systemic disease can occur. Neither the degree of infectiousness nor the reservoir of disease has been accurately defined, but heterosexual transmission has been attributed largely to asymptomatic female carriers and in the MSM population; asymptomatic rectal infection and/or penile infection is the likely source of onward transmission.
CLINICAL FEATURES
First stage (primary LGV)
The incubation period lasts 3–30 days, after which a primary lesion occurs in the form of a small painless papule, pustule, nodule, shallow erosion, or herpetiform ulcer.
The initial lesions may be differentiated from the more common herpetic lesions by the lack of pain associated with the lesion. Differentiation from a syphilitic chancre requires serologic testing. The primary lesion of LGV is most commonly located on the coronal sulcus of men and on the posterior vaginal wall, fourchette (known as the frenulum of labia minora/posterior commissure of the labia minora) or vulva and on the cervix of women. The lesion usually heals within 1 week and may go unnoticed in the urethra, vagina, or rectum. Mucopurulent discharge from urethra, cervix, or rectum may be
present regarding the inoculation site. Some ulcers in the recent MSM outbreak have been described as indurated and of variable tenderness; their duration has been as long as several weeks. Extra-genital lesions have been reported such as ulcers and fissures in the perianal area in MSM, the lip or oral cavity (tonsil), and extra-genital lymph nodes. The new clinical picture is mostly seen among the MSM.
Hemorrhagic proctitis in MSM is the primary manifestation of infection following direct transmission to the rectal mucosa. It might also occur in women with rectal exposure. In the recent outbreaks of LGV in MSM in Western Europe, approximately 96% of all cases presented with proctitis; symptoms included rectal pain, anorectal bleeding, mucoid and/or hemopurulent rectal discharge, tenesmus, constipation, and other symptoms of lower gastrointestinal inflammation. However, patients may present to gastroenterologists or colorectal surgeons for persisting symptoms.
Endoscopic features are nonspecific, with a wide range of differential diagnoses including Crohn’s disease, lymphoma, anorectal carcinoma, and other sexually transmitted ulcerative infections (eg, syphilis, herpes).LGV proctitis mimics chronic inflammatory bowel disease, both clinically and in the pathological substrate. These cases may present with an incomplete or undisclosed history of proctosigmoiditis, without the characteristic adenopathy syndrome. During the initial evaluation and colonoscopy, there is a strong clinical and endoscopic suspicion of inflammatory bowel disease by virtue of presentation and endoscopic and histological findings.
The diagnosis of IBD is subsequently modified to LGV proctosigmoiditis when one or more of the following transpire: 1) there is a failure of response to inflammatory bowel disease therapy; 2) additional components of history are identified (MSM/travel); 3) initially performed chlamydia antibody test shows seroconversion in the follow-up sample; and response to antibiotics effective against chlamydia. Some patients reported
systemic symptoms (fever and malaise). Genital ulcers and inguinal symptoms were less common.
Secondary lesions (secondary LGV)
Secondary LGV begins within 2–6 weeks after the onset of primary lesion. Depending on the site of inoculation, LGV can cause inguinal syndrome (after primary lesion of the anterior vulva, penis, or urethra) or anorectal syndrome (usually after primary lesion of the posterior vulva, vagina, or anus).Inguinal syndrome presents with painful inflammation of the inguinal (superficial and deep) lymph nodes and occurs mostly in men (occurs in only 20% of women with LGV). It is the most common clinical manifestation of genital LGV among heterosexuals. In two-thirds of cases, this produces unilateral enlargement, inflammation, suppuration, and abscesses. The disease process may involve one or many lymph nodes, and if adjacent to one another, they may coalesce. The central areas of such lymph nodes may then undergo necrosis. Fluctuant and suppurative lymph nodesthen develop, causing the classic “bubo” of LGV. These “buboes” may rupture in one-third of patients, which may lead to sinus tract formation. In other cases, they may develop into hard, nonsuppurative masses. When both inguinal and femoral lymph nodes are involved, they can be separated by the inguinal (Poupart’s) ligament, “Groove sign”. This sign is pathognomonic of LGV, but occurs in only 15%–20% of cases.
DIAGNOSTIC APPROACH
Modern techniques now rely on nucleic acid amplification tests (NAATs) in well-equipped laboratories. The assays have high sensitivity and specificity. C.
trachomatis is an intracellular organism, so samples should contain cellular material. For the detection of LGV serovars of C. trachomatis, different DNA samples can be used:
1) primary anogenital lesion swab (ulcer base exudate),
2) rectal mucosa swab (when anorectal LGV is suspected)
3) enlarged or fluctuant lymph nodes or buboes aspirate(when inguinal LGV is suspected).After topical disinfection, a 21-gauge needle should be inserted into the lymph node through healthy adjacent tissue and the pus aspirated into a syringe; a small volume (,0.5 mL) of saline solution may be injected and re-aspirated for nonfluctuant nodes.Urethral swab or first-catch urine specimen can be used as a sample when urethritis and/or inguinal lymphadenopathy is present and LGV is suspected as the cause. Urine specimen usually shows negative PCR results in case of anorectal manifestation of LGV . A two-step procedure is usually followed. The first step includes C. trachomatis NAAT test (test only confirms presence of C. trachomatis, and does not allow serogroup identification). NAAT s are not approved for testing samples from extra-genital sites,but studies have demonstrated high sensitivity and specificity in rectal chlamydial infections.
C. trachomatis NAAT s can be performed using one of three methods: PCR, strand
displacement amplification, or transcription-mediated amplification. The second step is performed only if the first step test detects C. trachomatis in the sample. The second step diagnostic test is LGV biovar–specific DNA NAAT from the same sample used in first step test. T wo tests for the second step are available: a real-time PCR-based test (detects all C. trachomatis LGV biovar strains)9,41,50 and areal-time quadriplex PCR-based assay. Both PCR techniques are reliable; the assay correctly identified 100% of non-LGV chlamydial specimens, 100% with no chlamydial infection,and 96.36% LGV specimens. The identification of rectal polymorphonuclear leucocytes from rectal swabs is predictive of LGV proctitis, especially in HIV-positive MSM. In a study of 87 MSM with confirmed LGV , majority of samples (Gram-stained anorectal swabs) had more than 10 white blood (WBCs) per high-powerfield.Histology of the lymph nodes is not specific: follicular hyperplasia, abscesses, cryptitis, and crypt abscesses without distortion of crypt architecture (last two were the most common findings in 12 anorectal biopsies from LGV in MSM).
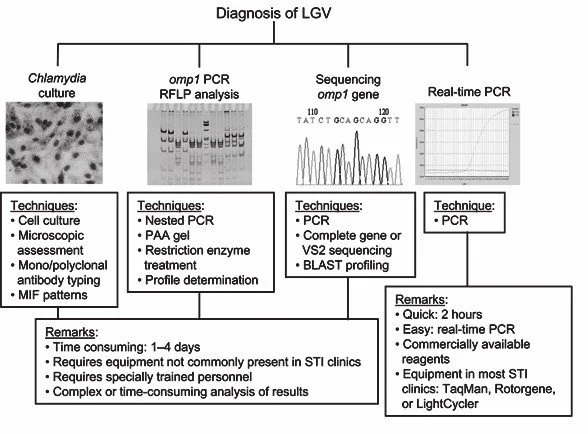
MANAGEMENT AND NEW RESEARCH
A proper treatment regimen cures the infection and prevents further damage to tissues. LGV causes potentially severe infections with possibly irreversible sequels if adequate treatment is not begun promptly. Early and accurate diagnosis is essential. The management of the ongoing epidemic LGV in industrialized Western countries caused by the C. trachomatis variant L2b still needs improvements in diagnosis, therapy, and prevention. Therefore, the rapid C. trachomatis variantL2b-specific PCR has been developed to circumvent laborious ompA gene sequencing.57 C. trachomatis should be treated with antibacterial drugs reaching high intracellular concentrations. In general, intracellular-acting agents such as doxycycline, erythromycin, and azithromycin and certain quinolones are advised. Doxycycline is the drug of choice for patients who are not pregnant.
Pregnant and lactating women should be treated with erythromycin or azithromycin.Only one case of clinical failure with extended doxycycline therapy has been reported in an HIV-negative MSM with anorectal LGV. He subsequently responded to treatment with moxifloxacin 400 mg daily for 10 days; no isolate was available for resistance testing.
Three MSMs who developed inguinal LGV failed the recommended 21-day doxycycline treatment. A recent paper suggests that inguinal LGV may require prolonged courses of doxycycline, exceeding the currently advised 21-day regimen.3 Current STD treatment guidelines for MSM in the United States recommend treatment of rectal chlamydia with a single 1-g dose of azithromycin, but there are increasing concerns about its effectiveness treatment failures reported A recent study suggests that HIV seropositivity was the strongest risk factor for LGV and that proctoscopic findings and elevated WBC counts in anorectal smear specimens were the only clinically relevant predictors forLGV infection. For all MSM reporting receptive anorectal intercourse, rectal chlamydia screening is recommended.
If chlamydia test results are not available, for MSM reporting receptive anorectal intercourse who have proctitis noted by proctoscopic examination and a WBC count of
.10 cells/high-power field in an anorectal Gram stain specimen or who have proctitis noted by proctoscopic examination and HIV seropositivity, treatment with doxycycline
(100 mg twice per day) is advised until chlamydia test results are available, with a minimum duration of treatment of 7 days. If the anorectal chlamydia test result is negative, no treatment should be administered or doxycycline treatment should be stopped after a minimum of 7 days. If the anorectal chlamydia test result is positive and LGV testing is available, doxycycline treatment (100 mg twice per day).
HEALTH PROMOTION AND PREVENTIONS
The outbreaks of LGV among MSM in developed countries support the need for careful screening of these patients. A routine screening for rectal chlamydia is recommended in asymptomatic men at risk, as this may represent an important reservoir for the onward transmission of infection.
Treatment of this group of patients is essential in the attempt to eradicate the disease. Patients should get information about safer sex behavior. Condom use may reduce the risk of LGV transmission, but uncovered ulcerated areas remaina problem. With early and accurate diagnosis and appropriate antibiotic therapy, the prognosis is excellent, but reinfection and relapses may occur.
CONCLUSION
lymphogranuloma venereum (LGV), is a significant sexually transmitted infection caused by invasive serovars of Chlamydia trachomatis that primarily affects the lymphatic system. It presents a diagnostic challenge due to its varied clinical stages and similarity to other genital ulcer diseases. Early recognition through a combination of clinical suspicion, molecular diagnostics, and exclusion of mimics is crucial for effective management.
Prolonged antibiotic therapy, primarily with doxycycline, coupled with appropriate management of lymphadenopathy and partner treatment, leads to excellent outcomes and prevention of severe chronic complications such as lymphatic obstruction and genital deformities. Ongoing research into molecular genotyping, rapid diagnostics, and evolving epidemiology underscores the need for vigilance and adaptability in clinical and public health approaches to control LGV , especially in high-risk populations.
Addressing these challenges will enhance early detection, optimize treatment strategies, and reduce transmission, ultimately improving patient prognosis and sexual health globally.
REFERENCE
1. Beigi RH. Lymphogranuloma venereum. In: Beigi RH, editor. Sexually Transmitted Diseases. W est Sussex, UK: John Wiley & Sons, L TD;
2. T araktchoglou M, Pacey AA, Turnbull JE, Eley A. Infectivity of Chlamydia trachomatis serovar LGV but not E is dependent on host cell heparansulfate. Infect Immun.
3. Oud EV , de Vrieze NH, de Meij A, de Vries HJ. Pitfalls in the diagnosis and management of inguinal lymphogranuloma venereum: important lessons from a case series. Sex T ransm Infection
4. de Vries HJ, Zingoni A, Kreuter A, Moi H, White JA. 2013 European guideline on the management of lymphogranuloma venereum. J Eur Acad Dermatol V enereol.
5. de Vrieze NH, de Vries HJ. Lymphogranuloma venereum among men who have sex with men. An epidemiological and clinical review. Expert Rev Anti Infect Ther.
6. Mabey D, Peeling R W . Lymphogranuloma venereum.
7. Behets FM, Brathwaite AR, Hylton-Kong T, et al. Genital ulcers:etiology, clinical diagnosis, and associated human immunodeficiency virus infection in Kingston, Jamaica. Clin Infect Dis
8. Ndinya-Achola JO, Kihara AN, Fisher LD, et al. Presumptive specific clinical diagnosis of genital ulcer disease (GUD) in a primary healthcare setting in Nairobi. Int J STD AIDS
9. V an der Bij AK, Spaargaren J, Morré SA, et al. Diagnostic and clinical implications of anorectal lymphogranuloma venereum in men who have sex with men: a retrospective case-control study. Clin Infect Dis
