The Development of Cerebral Cortex in Human and Mammals
1. Manas kyzy Uulkan
2. Vadivel Divya
(1. Lecturer, Dept. of Histology, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.
2. Student, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.)
ABSTRACT:
Background: The cerebral cortex is the most highly developed region of the mammalian brain and is responsible for higher cognitive functions such as perception, learning, memory, language, and voluntary motor control. The development of the cerebral cortex is a complex and tightly regulated process that involves coordinated events including neural proliferation, migration, differentiation, synaptogenesis, and cortical organization. While fundamental mechanisms of cortical development are conserved across mammals, the human cerebral cortex exhibits unique features such as increased size, surface area, and structural complexity that underlie advanced cognitive abilities.
Objective: This review aims to describe the developmental processes of the cerebral cortex in humans and other mammals, highlighting key cellular and molecular mechanisms, stages of cortical formation, and evolutionary differences that contribute to species-specific brain functions.
Methods: A descriptive review approach was used based on standard neuroanatomy, neuroembryology, and developmental neuroscience literature. Information related to cortical development, neuronal migration patterns, cortical layering, and comparative aspects between humans and other mammals was synthesized to provide a comprehensive overview.
KEYWORDS:
Cerebral cortex, Brain development, Neurogenesis, Neuronal migration, Mammalian brain
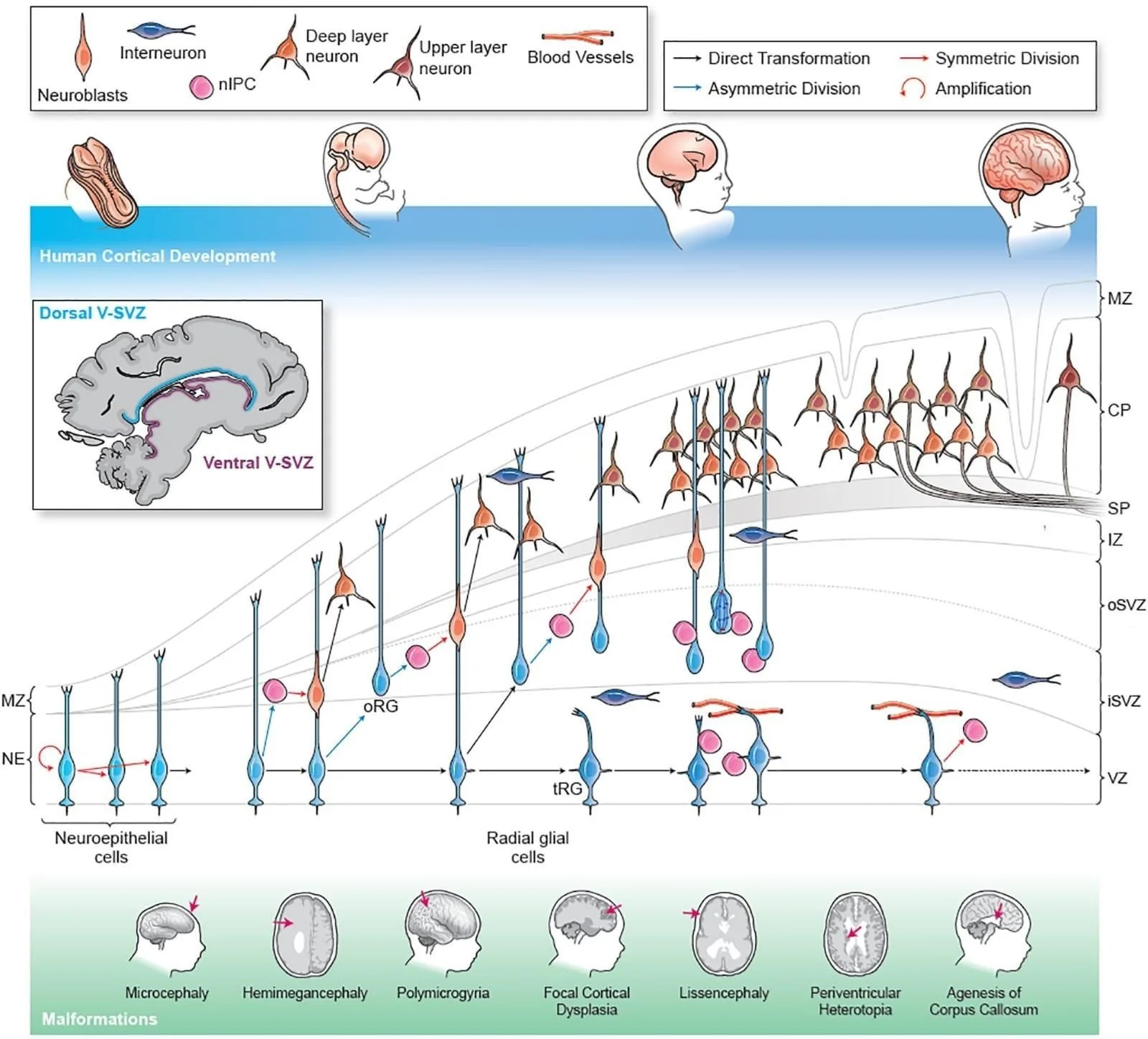
FIGURE:1 THE DEVELOPMENT OF CEREBRAL CORTEX
INTRODUCTION:
The cerebral cortex represents the outermost layer of the brain and is responsible for the most sophisticated functions of the nervous system. It enables perception, voluntary motor control, learning, memory, reasoning, language, and conscious awareness. The remarkable functional capacity of the cerebral cortex is directly related to its structural complexity and developmental organization.
In mammals, the cerebral cortex has undergone significant evolutionary expansion, with the most dramatic enlargement observed in primates, particularly humans. While smaller mammals such as rodents possess relatively smooth cortices with limited surface area, humans exhibit an extensively folded cortex that allows for increased surface area within the limited volume of the skull. This structural expansion supports enhanced connectivity and functional specialization.
Understanding how the cerebral cortex develops is crucial for appreciating both normal brain function and the origins of neurological and psychiatric disorders. Abnormalities in cortical development can lead to severe conditions such as microcephaly, lissencephaly, epilepsy, autism spectrum disorders, and schizophrenia. Therefore, a detailed study of cortical development provides insight into brain evolution, function, and disease.
METHODS:
A descriptive review approach was used based on standard neuroanatomy, neuroembryology, and developmental neuroscience literature. Information related to cortical development, neuronal migration patterns, cortical layering, and comparative aspects between humans and other mammals was synthesized to provide a comprehensive overview.
RESULTS:
The cerebral cortex represents the most highly evolved region of the mammalian brain and is responsible for higher cognitive functions, sensory perception, voluntary motor control, language, and behavior. In humans, the cerebral cortex is exceptionally expanded and complex compared to other mammals, reflecting advanced intellectual and social capabilities. Development of the cerebral cortex is a highly regulated, multistep process involving neurogenesis, neuronal migration, differentiation, synaptogenesis, and cortical maturation. These processes are conserved across mammals but exhibit species-specific variations in timing, scale, and complexity.
ANATOMICAL OVERVIEW OF THE DEVELOPING CEREBRAL CORTEX
The cerebral cortex develops from the dorsal telencephalon, which arises from the anterior neural tube during early embryogenesis. Initially, the telencephalic wall consists of a simple neuroepithelium that undergoes rapid proliferation. As development proceeds, this wall differentiates into distinct layers known as the ventricular zone (VZ), subventricular zone (SVZ), intermediate zone (IZ), cortical plate (CP), and marginal zone (MZ).
In mature mammals, the cerebral cortex is organized into six distinct layers (neocortex), arranged in an inside-out pattern during development. Older neurons occupy deeper layers, while younger neurons migrate past them to form superficial layers. This laminar organization is a defining feature of mammalian cortical architecture and is especially pronounced in primates and humans.
VENTRICULAR ZONE: NEURAL PROGENITOR POOL
The ventricular zone is the innermost germinal layer lining the lateral ventricles and is the primary site of neural progenitor cell proliferation. It consists mainly of neuroepithelial cells and radial glial cells, which serve as both neural stem cells and scaffolds for migrating neurons. Early in development, symmetric divisions expand the progenitor pool, while later asymmetric divisions generate neurons and glial cells.
Radial glial cells express transcription factors such as Pax6 and Sox2, which maintain progenitor identity and regulate cortical growth. In humans and higher mammals, the prolonged activity of radial glial cells contributes significantly to cortical expansion and increased neuronal output.
SUBVENTRICULAR ZONE: AMPLIFICATION AND EVOLUTIONARY EXPANSION
The subventricular zone lies superficial to the ventricular zone and is particularly prominent in primates. It contains intermediate progenitor cells and outer radial glial cells, which further amplify neuron production. The outer subventricular zone (oSVZ), well developed in humans, is considered a major evolutionary innovation that supports cortical folding (gyrification) and increased surface area.
Cells in the SVZ predominantly express transcription factors such as Tbr2 (Eomes), which promote neuronal differentiation. The expansion of the SVZ in humans and other gyrencephalic mammals correlates with increased cortical thickness and complexity.
NEURONAL MIGRATION AND CORTICAL PLATE FORMATION
Newly generated neurons migrate from the VZ and SVZ to their final destinations in the cortical plate. Radial migration is the dominant mode in excitatory pyramidal neurons and occurs along radial glial fibers. Inhibitory interneurons, by contrast, originate in the ganglionic eminences of the ventral telencephalon and migrate tangentially into the cortex.
The cortical plate forms between the marginal zone and the intermediate zone and gradually develops into the six-layered neocortex. The marginal zone later becomes layer I of the cortex and contains Cajal–Retzius cells, which secrete Reelin, a crucial glycoprotein that regulates neuronal positioning and laminar organization.
HISTOLOGY OF THE DEVELOPING CEREBRAL CORTEX
Histologically, the developing cerebral cortex displays distinct cellular zones corresponding to different stages of neuronal maturation. The ventricular zone is densely packed with elongated, mitotically active cells. The subventricular zone contains loosely arranged progenitors with round nuclei. The intermediate zone appears relatively cell-poor and later differentiates into the white matter. The cortical plate is densely cellular and shows progressive stratification as neurons accumulate.
In humans, the cortical plate becomes thicker and more complex over a prolonged gestational period, reflecting extended neurogenesis and maturation compared to other mammals such as rodents.
CORTICAL LAYER DIFFERENTIATION AND MATURATION
As development proceeds, neurons differentiate into specific cortical layers (I–VI), each characterized by distinct neuronal types, connectivity, and gene expression profiles. For example:
• Layer I (Molecular layer): Contains few neurons and abundant horizontal fibers.
• Layers II–III: Rich in small and medium pyramidal neurons involved in corticocortical connections.
• Layer IV: Prominent in sensory cortices and receives thalamocortical input.
• Layers V–VI: Contain large pyramidal neurons projecting to subcortical targets.
Transcription factors such as Tbr1, Ctip2, and Satb2 regulate layer-specific neuronal identity. These molecular mechanisms are highly conserved across mammals but are more elaborated in humans.
GLIOGENESIS AND SYNAPTOGENESIS
Following neurogenesis, radial glial cells give rise to astrocytes and oligodendrocytes. Gliogenesis occurs later in development and continues postnatally. Astrocytes support synapse formation, neurotransmitter regulation, and metabolic homeostasis, while oligodendrocytes myelinate axons, enhancing signal transmission.
Synaptogenesis begins prenatally but peaks after birth, especially in humans. This is followed by synaptic pruning, a process that refines neural circuits based on activity and experience. The extended period of synaptic plasticity in humans underlies learning, memory, and behavioral adaptation.
COMPARATIVE DEVELOPMENT: HUMANS VS OTHER MAMMALS
Although the basic principles of cortical development are conserved among mammals, significant differences exist:
• Humans exhibit prolonged neurogenesis and delayed cortical maturation.
• The oSVZ is markedly expanded in primates compared to rodents.
• Gyrification is prominent in humans and higher mammals but absent in lissencephalic species like mice.
• Human cortex shows greater neuronal diversity and synaptic complexity.
These differences reflect evolutionary adaptations that support advanced cognitive functions.
CLINICAL CORRELATION
Disruption of cortical development leads to severe neurodevelopmental disorders:
• Lissencephaly: Defective neuronal migration results in a smooth cerebral surface.
• Microcephaly: Reduced progenitor proliferation leads to decreased brain size.
• Polymicrogyria: Abnormal cortical organization causes excessive small gyri.
• Autism spectrum disorders and schizophrenia have been linked to altered synaptogenesis and cortical connectivity.
MOLECULAR REGULATION OF CORTICAL DEVELOPMENT
Cortical development is controlled by complex genetic and molecular networks, including:
•Sonic hedgehog (Shh): Regulates progenitor proliferation and patterning.
•Wnt/β-catenin signaling: Promotes cortical progenitor expansion.
•Notch signaling: Maintains neural stem cell populations.
• Reelin signaling: Ensures proper neuronal layering.
These pathways interact with environmental factors to shape cortical structure and function.
CONCLUSION
The development of the cerebral cortex in humans and mammals is a highly coordinated and evolutionarily conserved process that underlies higher brain function. While the fundamental mechanisms are shared across species, humans exhibit prolonged development, expanded progenitor zones, and increased structural complexity. Understanding cortical development is essential for elucidating the basis of cognition and for diagnosing and treating neurodevelopmental disorders. Ongoing research into molecular and cellular mechanisms continues to provide insight into both normal brain development and disease.
REFERENCES
1. Carlson, B. M. Human Embryology and Developmental Biology. Elsevier.
2. Kandel, E. R., et al. Principles of Neural Science. McGraw-Hill.
3. Sadler, T. W. Langman’s Medical Embryology. Wolters Kluwer.
4. Rakic, P. (2009). Evolution of the neocortex: a perspective from developmental biology. Nature Reviews Neuroscience.
5. Kriegstein, A., & Alvarez-Buylla, A. (2009). The glial nature of embryonic and adult neural stem cells. Annual Review of Neuroscience.
6. Bystron, I., Blakemore, C., & Rakic, P. (2008). Development of the human cerebral cortex: Boulder Committee revisited. Nature Reviews Neuroscience.
7. Molnár, Z., et al. (2019). Development of the cerebral cortex: evolution, development, and plasticity. Nature Reviews Neuroscience.