Zones of the Adrenal Cortex
1. Manas kyzy Uulkan
2. Dharaniya Sree Kumaresan
(1. Lecturer, Dept. of Histology, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.
2. Student, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.)
ABSTRACT:
The adrenal cortex is a highly organized endocrine tissue that surrounds the adrenal medulla and is responsible for the synthesis and secretion of steroid hormones essential for homeostasis. Its structural and functional zonation into the zona glomerulosa, zona fasciculata and zona reticularis reflects distinct steroidogenic capacities critical for mineral balance, metabolic regulation, stress response, and androgen production.
This article reviews adrenal cortical morphology, cellular differentiation, steroidogenic pathways, regulatory mechanisms, and the clinical implications of dysregulated hormone synthesis. Comparative insights from animal models, molecular controls of steroidogenesis, and emerging perspectives in adrenal biology are also discussed. A thorough understanding of adrenal cortex zonation is fundamental for clinicians and researchers addressing endocrine disorders, metabolic disease and stress physiology.
KEYWORDS:
Adrenal cortex, steroidogenesis, zona glomerulosa, zona fasciculata, zona reticularis, corticosteroids, mineralocorticoids, glucocorticoids, adrenal androgens, regulation, and endocrine pathology.
FIGURE:1 ADRENAL GLAND-INTERNAL STRUCTURE
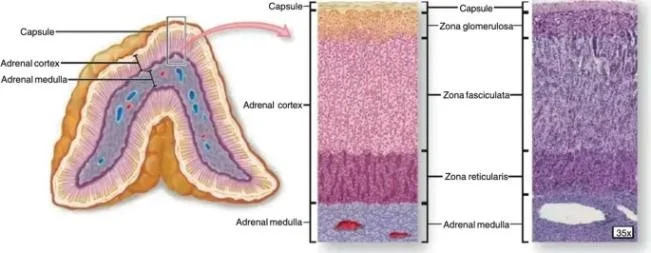
INTRODUCTION:
The adrenal glands are paired endocrine organs located at the superior poles of the kidneys. Each gland consists of an outer cortex and an inner medulla that arise from different embryological origins and perform distinct endocrine functions. The adrenal cortex, derived from mesodermal tissue, is the primary site of steroid hormone production, which regulates critical physiological processes including electrolyte homeostasis, blood pressure control, carbohydrate metabolism, immune modulation, and secondary sexual characteristics.
Anatomically, the adrenal cortex is divided into three concentric zones: the zona glomerulosa (outermost), the zona fasciculata (middle), and the zona reticularis (innermost). Although contiguous in structure, each zone expresses unique enzymes, transcription factors, and regulatory responses that confer specific steroidogenic functions. This zonal specialization exemplifies the principle of structure–function correlation in endocrine physiology.
Understanding the precise organization and regulation of adrenal cortical zones is indispensable for appreciating how the endocrine system adapts to physiological demands and why its dysfunction leads to significant clinical diseases such as hyperaldosteronism, Cushing’s syndrome, and congenital adrenal hyperplasia. This article synthesizes current knowledge on adrenal cortex zones with an emphasis on morphology, molecular controls, hormonal outputs, regulatory feedback mechanisms, and clinical relevance.
METHODS:
This article is a comprehensive narrative review of established textbooks, primary research articles, and authoritative endocrine reviews. Sources were selected to integrate morphological, biochemical, molecular, and clinical perspectives on adrenal cortex zonation. Key databases (e.g., PubMed, Endocrine Reviews) and standard references in physiology, histology, and endocrinology were consulted to compile up-to-date information on steroidogenic pathways, gene expression patterns, and regulatory feedback loops. Emphasis was placed on correlating cellular structure with functional output and clinical phenotype.
RESULTS:
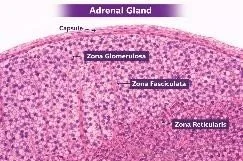
Anatomical and Histological Overview of the Adrenal Cortex
The adrenal cortex forms the outermost functional portion of the adrenal gland and accounts for approximately 80–90% of the gland’s volume. Under light microscopy, the cortex displays three distinct zones:
1. Zona Glomerulosa (ZG)—Beneath the capsule, cells appear in rounded clusters.
2. Zona Fasciculata (ZF)—Thickest layer; cells arranged in long cords or fascicles radiating toward the medulla.
3. Zona Reticularis (ZR)—Innermost cortical layer; cells form anastomosing networks.
Each zone’s cellular morphology reflects its functional specialization. ZG cells are smaller with dense cytoplasm, ZF cells contain abundant lipid droplets that appear foamy, and ZR cells are compact with lipofuscin pigment.
ZONA GLOMERULOSA:
Structure and Steroidogenic Pathways:
The zona glomerulosa lies beneath the adrenal capsule and contributes nearly 5–15% of total cortical volume. Its cells are arranged in rounded clusters resembling glomeruli. These cells express aldosterone synthase (CYP11B2)—an enzyme that catalyzes the final steps in aldosterone biosynthesis from deoxycorticosterone. Unlike deeper cortical zones, ZG cells lack significant expression of enzymes such as 17α-hydroxylase (CYP17), limiting their capacity to produce glucocorticoids.
Hormonal Function and Regulation:
Aldosterone is the primary mineralocorticoid secreted by the ZG. It plays a critical role in maintaining sodium balance, potassium homeostasis, and blood pressure. Aldosterone increases sodium reabsorption and potassium excretion in renal distal tubules and collecting ducts, thereby influencing extracellular fluid volume and arterial pressure.
Regulation of aldosterone secretion is primarily driven by the renin–angiotensin–aldosterone system (RAAS) and plasma potassium levels. Renin release from juxtaglomerular cells in the kidney, stimulated by hypovolemia or low sodium, leads to the generation of angiotensin II, which directly stimulates aldosterone synthesis. High extracellular potassium also stimulates ZG cells. In contrast, adrenocorticotropic hormone (ACTH) plays a minor and transient role in ZG regulation.
HISTOLOGY OF ZONA GLOMERULOSA:
FIGURE:3 HISTOLOGICAL STRUCTURE OF ZONA GLOMERULOSA
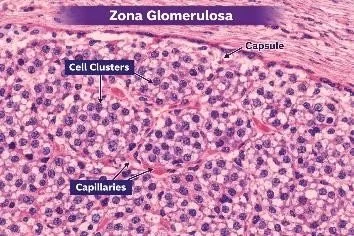
Zona glomerulosa is the outermost layer of the adrenal cortex and is located immediately beneath the connective tissue capsule. It is separated from the capsule by a thin layer of connective tissue and forms the superficial cortical zone of the adrenal gland.
Histologically, the cells of the zona glomerulosa are arranged in characteristic rounded clusters, arcs, or whorl-like structures, giving a glomerulus-like appearance from which the name “glomerulosa” is derived. These cell clusters are separated by delicate connective tissue septa that support the tissue framework.
The cells are small, compact, and columnar to pyramidal in shape, with centrally placed round nuclei. Their cytoplasm is lightly eosinophilic and contains fewer lipid droplets compared to the deeper cortical layers, especially the zona fasciculata. Because of the reduced lipid content, the cells appear darker and more densely packed in routine H&E sections.
A rich network of fenestrated sinusoidal capillaries runs between the cell clusters, providing an efficient blood supply for hormone secretion and transport. These capillaries allow rapid diffusion of mineralocorticoids into the circulation.
Functionally, the zona glomerulosa is specialized for the synthesis and secretion of mineralocorticoids, mainly aldosterone, which regulates sodium and potassium balance and helps maintain blood pressure. The histological structure of compact cells with fewer lipid vacuoles correlates with its specific role in aldosterone production.
Clinical Correlation
Dysregulation of ZG function leads to disorders of electrolyte balance and blood pressure:
Primary hyperaldosteronism (Conn’s syndrome): Autonomous aldosterone production causes hypertension, hypokalemia, and metabolic alkalosis.
Hypoaldosteronism: Seen in conditions like Addison’s disease or isolated aldosterone deficiency, leading to hyperkalemia and hypotension.
ZONA FASICULATA:
Morphology and Steroidogenic Machinery
Occupying 60–80% of the cortex, the zona fasciculata contains large cells with abundant lipid droplets that store cholesterol esters. These cells express key steroidogenic enzymes, including cholesterol side-chain cleavage enzyme (CYP11A1) and 17α-hydroxylase/17,20-lyase (CYP17), enabling the synthesis of glucocorticoids. The extensive smooth endoplasmic reticulum and mitochondria with tubular cristae facilitate steroid hormone production.
Glucocorticoid Biosynthesis and Function:
The primary glucocorticoid in humans is cortisol, which exerts metabolic, immunomodulatory, and stress-responsive effects. Cortisol increases gluconeogenesis, mobilizes amino acids from peripheral tissues, promotes lipolysis, and elevates blood glucose. It also has potent anti-inflammatory effects by modulating immune cell function and cytokine production.
Cortisol secretion is regulated by the hypothalamic–pituitary–adrenal (HPA) axis: hypothalamic corticotropin-releasing hormone (CRH) stimulates pituitary ACTH release, which in turn promotes cortisol synthesis in ZF cells. A negative feedback loop exists whereby elevated cortisol inhibits CRH and ACTH secretion.
HISTOLOGY OF ZONA FASICULATA:
FIGURE:4 HISTOLOGICAL STRUCTURE OF ZONA FASICULATA
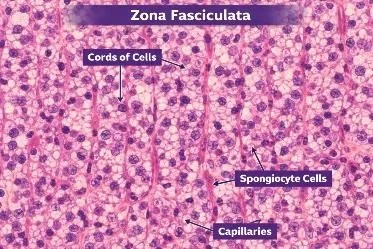
Zona fasciculata is the middle and widest layer of the adrenal cortex, located between the zona glomerulosa and the zona reticularis. It forms the bulk of the adrenal cortex and is easily identifiable in histological sections due to its distinctive pale appearance and organized structure.
Histologically, the cells are arranged in long, straight, parallel cords (fascicles) that run perpendicular to the surface of the gland. These cords are separated by sinusoidal capillaries, which form a rich vascular network for efficient transport of hormones into the bloodstream. This regular, column-like arrangement gives the layer its name fasciculata (meaning “bundle-like”).
The cells are large, polyhedral and have round centrally placed nuclei. Their cytoplasm appears pale and vacuolated because it contains a large amount of lipid droplets (cholesterol and steroid precursors). During tissue processing, these lipids are dissolved, leaving clear spaces, which gives the cells a characteristic spongy appearance—hence they are also called spongiocytes.
The sinusoidal capillaries between the cell cords are fenestrated, allowing easy diffusion of steroid hormones into the blood. This extensive vascularization supports the high secretory activity of this layer.
Functionally, the zona fasciculata is responsible for the secretion of glucocorticoids, mainly cortisol, which regulates metabolism, stress response, and immune functions. Its histological features—large lipid-rich cells arranged in straight cords with rich blood supply—are directly related to its role in steroid hormone synthesis.
Physiological and Pathological Implications:
Appropriate glucocorticoid action is essential for adaptation to stress, metabolic balance, and immune regulation.
Dysregulated cortisol levels contribute to clinical syndromes:
Cushing’s syndrome: Excess cortisol resulting from adrenal tumors, pituitary adenomas (Cushing’s disease), or exogenous glucocorticoid use, characterized by central obesity, insulin resistance, hypertension, and immunosuppression.
Adrenal insufficiency (Addison’s disease): Cortisol deficiency leads to fatigue, hypoglycemia, weight loss, and hypotension. Secondary adrenal insufficiency from pituitary dysfunction also impairs cortisol secretion.
ZONA RETICULARIS:
Cellular Characteristics and Steroidogenesis:
The innermost cortical layer, the zona reticularis, constitutes 5–10% of the cortex. Cells are smaller than those of ZF and have a network arrangement. ZR cells exhibit lipofuscin pigment and express a steroidogenic profile that favours production of androgen precursors. Enzymes such as CYP17 and limited expression of 3β-hydroxysteroid dehydrogenase (3β-HSD) enable synthesis of dehydroepiandrosterone (DHEA) and androstenedione.
Androgen Production and Biological Roles:
Adrenal androgens are weak androgens that serve as precursors to more potent sex steroids in peripheral tissues. In adults, adrenal androgens contribute modestly to circulating androgen levels, with greater physiological relevance in females, where they influence pubic and axillary hair development, libido, and other secondary sexual traits. In prepubertal children, adrenal androgens contribute to adrenarche.
Regulation of ZR androgen secretion is partly ACTH-dependent, though additional paracrine and autocrine factors modulate activity. Emerging evidence suggests intra-adrenal factors, including local growth factors and transcription regulators, influence ZR function.
HISTOLOGY OF ZONA RETICULARIS:
FIGURE: 5 HISTOLOGICAL STRUCTURE OF ZONA RETICULARIS
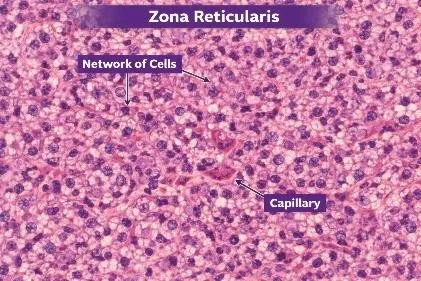
Zona reticularis is the innermost layer of the adrenal cortex, lying just above the adrenal medulla and beneath the zona fasciculata. It forms the deepest cortical zone and marks the transition between the cortex and medulla of the adrenal gland.
Histologically, the cells of the zona reticularis are arranged in an irregular network (reticular pattern) of interlacing cords and branching strands rather than in straight columns or rounded clusters. These cords are separated by numerous sinusoidal capillaries, giving the tissue a loosely arranged, mesh-like appearance.
The cells are small, polygonal, and closely packed, with round to oval centrally placed nuclei. Their cytoplasm is deeply eosinophilic and contains fewer lipid droplets than the zona fasciculata, so the cells appear darker in routine H&E-stained sections. Many cells also contain lipofuscin pigment granules, which give a brownish coloration and are characteristic of this layer.
A dense network of fenestrated sinusoidal capillaries runs between the cords of cells, ensuring efficient exchange between the blood and the hormone-secreting cells. This vascular arrangement supports the endocrine function of the layer.
Functionally, the zona reticularis is responsible for the secretion of androgens (mainly dehydroepiandrosterone – DHEA) and small amounts of glucocorticoids. Its histological features—compact, dark-staining cells arranged in a reticular pattern with rich vascularity—are closely related to its role in steroid hormone synthesis and secretion.
Clinical correlation:
Excess adrenal androgen production is associated with virilization and early pubertal changes:
Congenital adrenal hyperplasia (CAH): Genetic enzyme defects (e.g., 21-hydroxylase deficiency) lead to shunting of steroid precursors toward androgen synthesis, causing virilization.
Adrenal androgen-secreting tumors: Rare tumors can cause hirsutism, acne, and hormonal imbalance.
Molecular Regulation Across Cortical Zones
Modern research reveals that adrenal cortex zonation is controlled by transcription factors and signalling pathways.
For example:
SF-1 (Steroidogenic Factor-1): A master regulator of steroidogenic gene expression.
Wnt/β-catenin signalling: Important for cell fate determination and zonal maintenance.
ACTH receptor (MC2R): Predominantly influences ZF and ZR steroidogenesis.
These molecular cues interact with systemic regulators such as the RAAS and HPA axis to fine-tune zonal hormone output.
DIFFERENCES BETWEEN ZONES OF ADRENAL CORTEX:

CONCLUSION:
The adrenal cortex exemplifies functional specialization within endocrine organs. Its zonal architecture underlies the Each zone’s unique enzyme expression, regulatory control, and physiological role are crucial for maintaining electrolyte balance, metabolic homeostasis, stress adaptation, and sexual development. Disruption of these processes contributes to significant clinical disorders, underscoring the importance of adrenal cortical biology in health and disease. Continued research into molecular regulators of zonal identity and steroidogenesis holds promise for improved diagnosis and therapy of adrenal disorders.
REFERENCES:
1. Guyton, A. C., & Hall, J. E. Textbook of Medical Physiology. Elsevier.
2. Ross, M. H., & Pawlina, W. Histology: A Text and Atlas. Wolters Kluwer
3. Ganong, W. F. Review of Medical Physiology. McGraw-Hill.
4. Melmed, S., et al. Williams Textbook of Endocrinology. Elsevier.
5. Young, B., O’Dowd, G., & Woodford, P. Wheater’s Functional Histology. Elsevier.
6. Boron, W. F., & Boulpaep, E. L. Medical Physiology. Elsevier.
7. Jameson, J. L., et al. Harrison’s Principles of Internal Medicine. McGraw-Hill.
8. Hadley, M. E., & Levine, J. E. Endocrinology. Pearson.
9. Miller, W. L., & Auchus, R. J. (2011). The molecular biology, biochemistry, and physiology of human steroidogenesis and its disorders. Endocrine Reviews.
10. Vinson, G. P. (2003). Adrenal cortex and life. Molecular and Cellular Endocrinology.
11. Rainey, W. E., Rehman, K. S., & Carr, B. R. (2004). The human fetal adrenal: making adrenal androgens for placental estrogen. Seminars in Reproduction.
