Sleep Paralysis: When the Brain Wakes Before the Body – Mechanisms of REM Cycle Dissociation
1. Arunagiri Johnjebas
2. Palanisamsy Nithen
3. Tashimbetova Umut Kaparovna
(1. Student, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic
2. Student, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic
3. Teacher, International Medical Faculty, Osh State University, Osh, Kyrgyz Republic.)
Abstract
Sleep paralysis (SP) is a transient parasomnia defined by the persistence of rapid eye movement (REM) sleep-associated skeletal muscle atonia into the wakeful state, creating a dissociative neurological condition in which conscious awareness coexists with complete voluntary motor inhibition. Occurring at the boundary between REM sleep and wakefulness — whether at sleep onset in its hypnagogic form or upon awakening in its hypnopompic form — its clinical presentation ranges from brief, unremarkable episodes of immobility to profoundly distressing experiences accompanied by vivid multisensory hallucinations. This narrative review synthesizes evidence from six peer-reviewed medical publications to examine the neurophysiological underpinnings of the REM cycle, the specific neural and neurochemical mechanisms through which its dysregulation gives rise to sleep paralysis, the epidemiological landscape of the condition across global populations, and current evidence-based approaches to its clinical management.
The subcoeruleus nucleus, GABAergic and glycinergic neurotransmitter systems, orexinergic projections, and the broader brainstem-hypothalamic regulatory network emerge as central players in understanding why, in some individuals, the brain regains consciousness while the body remains paralyzed. Epidemiologically, sleep paralysis demonstrates a global lifetime prevalence of approximately 30% in broad estimates, with dramatically elevated rates in patients with post-traumatic stress disorder and panic disorder. Cultural interpretations of the phenomenon vary substantially across human societies, yet the underlying neuroscience is strikingly universal. This review concludes that while isolated episodes are generally benign, recurrent sleep paralysis demands structured clinical evaluation for comorbid sleep disorders, psychiatric illness, and circadian disruption, and calls for further investigation into targeted pharmacological and psychobehavioral interventions. An improved understanding of the neurophysiology of REM sleep dysregulation at the sleep-wake boundary is essential for translating emerging scientific knowledge into meaningful clinical benefit.
Keywords: sleep paralysis, REM sleep, muscle atonia, hypnagogic hallucinations, parasomnia, subcoeruleus nucleus, GABA, glycine, orexin, isolated sleep paralysis, narcolepsy
INTRODUCTION
There is something deeply unsettling about the experience — awakening in the dark and finding that the mind has arrived but the body has not. The eyes may open, the room comes into focus, and the clear awareness of being awake settles in with startling precision. And yet the arms will not move. The legs lie as still and heavy as stone. The voice does not come. In some episodes, a malevolent weight seems to press against the chest, making each breath feel labored and shallow. A shadow occupies the periphery of vision. The certainty of a presence — human or otherwise — presses against the edges of consciousness. This is sleep paralysis, a phenomenon that has haunted human experience across cultures and centuries, attributed in various traditions to night-demons, witches, supernatural visitors, and pressing spirits, yet now understood through the precise language of neuroscience as a dissociation between brain awakening and the cessation of REM sleep-associated motor inhibition.
Sleep paralysis (SP) is formally classified under the International Classification of Sleep Disorders, Third Edition (ICSD-3) as a parasomnia associated with rapid eye movement sleep. It may arise as an isolated phenomenon — the form most commonly encountered in the general population — or in direct association with narcolepsy, where it constitutes one component of the diagnostic tetrad alongside excessive daytime sleepiness, cataplexy, and hypnagogic hallucinations. At its neurophysiological core, SP represents a failure of the smooth temporal coordination between the two principal states of neural organization that alternate throughout a night of sleep — specifically at the moment of REM sleep termination or, less commonly, at its onset. During normal REM sleep, a sophisticated and actively maintained neural apparatus ensures that the vivid, emotionally charged theater of dreaming is effectively decoupled from voluntary motor output. The brainstem, through the coordinated activity of the subcoeruleus nucleus and downstream pathways utilizing gamma-aminobutyric acid (GABA) and glycine as inhibitory neurotransmitters, suppresses spinal and cranial motor neuron activity with remarkable precision. When awakening occurs before this suppression is fully withdrawn, the sleeper enters a state of conscious paralysis, aware of their environment but physiologically unable to act within it (Bhalerao et al., 2024).
The phenomenological landscape of sleep paralysis encompasses not only motor incapacitation but also an array of multisensory hallucinations that can be extraordinarily vivid and deeply frightening. What has been aptly described in the literature as 'a nightmare spilling into the real world' captures the essential quality of the experience — the perceptual content of dreaming, still vivid and dynamically present, persisting against the backdrop of a room that is simultaneously, undeniably real (Ableidinger and Holzinger, as cited in Bhalerao et al., 2024). The hallucinations of sleep paralysis have been categorized into three principal types: the intruder hallucination, involving the sensed or directly perceived presence of a threatening figure in the room; the incubus hallucination, characterized by pressure on the chest, difficulty breathing, and the felt sense of being physically restrained or attacked; and vestibular-motor hallucinations, involving sensations of floating, flight, or bodily displacement. These categories correspond, as will be discussed in detail, to identifiable neurophysiological substrates rather than merely anecdotal experience.
From an epidemiological standpoint, sleep paralysis presents a genuine clinical paradox — simultaneously common and underrecognized. A 2024 systematic review and meta-analysis by Hefnawy and colleagues, drawing on data from 167,133 participants across 25 countries, found a global prevalence of approximately 30%, with particularly elevated rates among individuals with post-traumatic stress disorder (PTSD) and panic disorder (Hefnawy et al., 2024). Despite this frequency, the condition remains poorly understood by many clinicians and even more inadequately explained to patients, who may interpret their experiences through cultural frameworks that delay appropriate help-seeking or, conversely, generate unnecessary shame and diagnostic confusion. The gap between the prevalence of sleep paralysis and the sophistication of available clinical interventions represents one of the more urgent unresolved questions in contemporary sleep medicine.
This review draws upon six peer-reviewed, published medical and neuroscientific articles to synthesize current understanding of the REM cycle mechanisms that underpin sleep paralysis, the neurochemical and anatomical circuitry implicated in its pathogenesis, the clinical and epidemiological characteristics of affected populations, the cultural dimensions of the condition's expression, and the therapeutic strategies that have shown promise in managing it. The ultimate aim is to provide clinicians, researchers, and medical students with a coherent, evidence-based account of why the brain sometimes wakes before the body — and what can be done when it does.
METHODS
This article constitutes a narrative review of the peer-reviewed literature pertaining to sleep paralysis and the neurophysiological mechanisms of REM sleep. A systematic search was conducted across PubMed, PubMed Central (PMC), Google Scholar, and the Cochrane Library using the following search terms and their Boolean combinations: 'sleep paralysis,' 'REM sleep atonia,' 'isolated sleep paralysis,' 'subcoeruleus nucleus,' 'motor atonia REM,' 'hypnagogic hallucinations,' 'parasomnia REM,' 'REM sleep neurophysiology,' 'GABA glycine motoneuron,' and 'sleep paralysis epidemiology.' Searches were restricted to peer-reviewed publications in English, with preference given to studies published between 2012 and 2024 to ensure contemporaneity of the evidence base while retaining access to foundational mechanistic work.
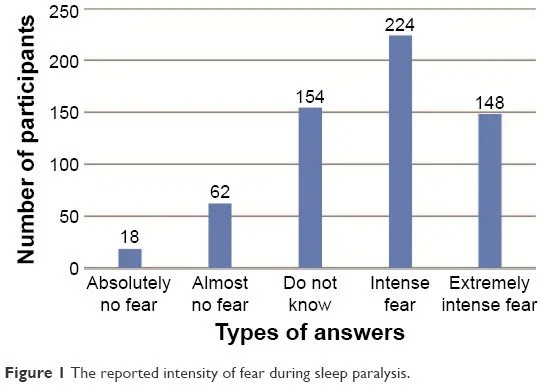
Reference lists of identified articles were reviewed for additional relevant sources through backward citation searching. Six primary source publications were selected for inclusion in this review on the basis of four criteria: methodological rigor, clinical and scientific relevance, contribution to mechanistic, epidemiological, or therapeutic understanding of sleep paralysis, and recency of publication. The selected articles include a systematic review and meta-analysis of global prevalence (Hefnawy et al., 2024), a narrative review of mechanisms and management from a community medicine perspective (Bhalerao et al., 2024), a neurochemical experimental study identifying the transmitter mechanisms responsible for REM paralysis (Brooks and Peever, 2012), a narrative review from a psychiatric residency journal addressing neurophysiology and cultural intersections (McShane et al., 2023), a spectral EEG analysis comparing sleep paralysis to standard and lucid REM sleep (Mainieri et al., 2021), and a comprehensive review of neural control of REM sleep and motor atonia (Vetrivelan and Bandaru, 2023). Articles primarily addressing sleep disorders other than sleep paralysis, or those lacking direct relevance to the REM mechanistic or clinical focus of this review, were excluded.
RESULTS
The Neurophysiology of REM Sleep: Architecture, Oscillations, and the Generation of Atonia
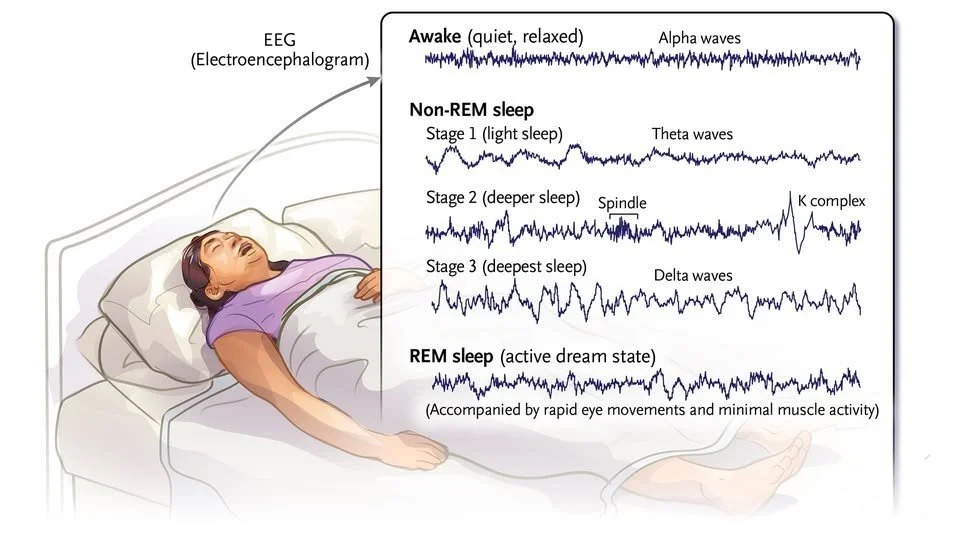
To understand sleep paralysis in its full depth, it is necessary first to appreciate the remarkable neural architecture of REM sleep itself. Human sleep does not proceed as a uniform or monolithic state but as a precisely cycling alternation between non-rapid eye movement (NREM) sleep and REM sleep, with each complete cycle lasting approximately 90 minutes and repeating four to six times over the course of a normal night. NREM sleep encompasses three progressive stages of increasing depth, characterized electroencephalographically by progressively increasing amplitude and decreasing frequency — from N1 and N2 sleep, marked by sleep spindles and K-complexes, through to N3 slow-wave sleep, in which delta oscillations predominate. REM sleep, in contrast, is characterized by a desynchronized, low-amplitude, mixed-frequency EEG that bears a striking resemblance to the waking state, giving rise to its alternate designation as 'paradoxical sleep.' This apparent contradiction — an EEG suggesting arousal occurring alongside behavioral quiescence — lies at the heart of REM sleep's physiological identity, and it is the resolution of this paradox that makes sleep paralysis mechanistically intelligible.
The defining physiological features of REM sleep are three: a desynchronized, activated cortical EEG; rapid conjugate eye movements occurring in phasic bursts; and near-total suppression of skeletal muscle tone, referred to as REM atonia. It is the third of these features — the active, neurally maintained suppression of voluntary motor activity — that is directly relevant to the pathogenesis of sleep paralysis. REM atonia is not a passive absence of motor drive but a vigorously maintained state of motor inhibition, generated and sustained by dedicated neural circuits that must be actively withdrawn at the moment of awakening. As Vetrivelan and Bandaru (2023) detail in their comprehensive review of neural control of REM sleep, the primary generator of motor atonia during REM sleep is the subcoeruleus nucleus (SLD), located in the pontine brainstem, which acts through descending projections to spinal cord interneurons and, ultimately, alpha-motoneurons.
The subcoeruleus nucleus achieves motor inhibition through a dual mechanism that has been elegantly characterized at the neurochemical level. Brooks and Peever (2012), in a landmark study published in the Journal of Neuroscience, demonstrated through a series of pharmacological experiments in rodent models that a powerful, combined GABA and glycine drive is responsible for triggering REM paralysis by switching off motoneuron activity. Critically, this inhibitory drive targets both metabotropic GABA-B receptors and ionotropic GABA-A and glycine receptors simultaneously, providing a robust and redundant system of motor suppression. The co-activation of multiple receptor types ensures that the inhibition of motor neurons during REM sleep is both powerful and resistant to disruption — a design feature that underscores the physiological importance of preventing motor activity during dreaming. The elegant molecular architecture of this dual inhibitory drive means that complete failure of REM atonia, as occurs in REM sleep behavior disorder (RBD), requires the disruption of multiple receptor systems simultaneously, while partial or timing-related dysregulation — as occurs in sleep paralysis — may arise from subtler disturbances in the synchronization of this inhibitory circuit's withdrawal at awakening.
The SLD does not act in isolation but receives regulatory input from a broader network of brainstem, hypothalamic, and forebrain structures that govern the timing and stability of REM sleep as a whole. Cholinergic neurons of the laterodorsal tegmentum (LDT) and pedunculopontine tegmentum (PPT) provide excitatory drive to the SLD, promoting REM sleep initiation and maintenance. In opposition, monoaminergic neurons of the locus coeruleus (noradrenergic), dorsal raphe nucleus (serotonergic), and tuberomammillary nucleus (histaminergic) are actively and profoundly inhibited during REM sleep — a state-dependent quiescence that disinhibits REM-generating circuits and allows the paradoxical combination of cortical activation and motor suppression to proceed. The flip-flop nature of the switch between REM and waking, dependent on mutual inhibition between REM-active and wake-active populations, means that the transition out of REM sleep during awakening involves the coordinated re-engagement of these wake-promoting systems while simultaneously withdrawing the inhibitory drive to motoneurons — a synchronization that, when imperfect, creates the conditions for sleep paralysis.
The orexin (hypocretin) system, projecting widely from the lateral hypothalamus to arousal-promoting brainstem and diencephalic structures, plays a critically important stabilizing role in this regulatory framework. Orexinergic neurons function, in essence, as a stabilizing bias in the sleep-wake flip-flop switch, preventing inappropriate state transitions by reinforcing the dominance of whichever state is currently active. In individuals with intact orexin signaling, transitions between sleep and wakefulness occur in a temporally organized and physiologically smooth fashion. In narcolepsy type 1, characterized by the selective autoimmune destruction of orexin-producing neurons in the lateral hypothalamus, the stabilizing influence of orexin is lost, and the boundaries between states become profoundly unstable. The result is a clinical syndrome in which fragments of REM sleep intrude into wakefulness — as cataplexy and hypnagogic hallucinations — and in which the complete dissociation of brain awakening from body paralysis that characterizes sleep paralysis occurs with greatly elevated frequency, affecting between 20% and 50% of narcoleptic patients (Bhalerao et al., 2024).
Pathogenesis of Sleep Paralysis: The Dissociative State at the Sleep-Wake Interface
Sleep paralysis arises when elements of REM sleep — specifically skeletal muscle atonia and, in many cases, the associated dreaming mentation — persist into or intrude upon the waking state, producing a mixed neurophysiological condition in which conscious awareness and complete voluntary motor inhibition coexist. This intrusion may occur at sleep onset, producing the hypnagogic form of sleep paralysis, or upon awakening, producing the more common hypnopompic form. Regardless of timing, the essential mechanism is the same: the brain's arousal systems re-engage — generating awareness, sensory processing, and environmental orientation — while the brainstem motor inhibitory circuits responsible for REM atonia remain active, sustaining paralysis beyond the point at which it is neurophysiologically appropriate.
Mainieri and colleagues (2021), in a carefully conducted spectral EEG analysis published in the Journal of Clinical Sleep Medicine, provided direct neurophysiological evidence that sleep paralysis represents a genuinely distinct brain state rather than merely a delayed or protracted awakening from standard REM sleep. By recording overnight polysomnography in individuals with documented sleep paralysis episodes and analyzing the spectral power characteristics of the EEG during SP compared to standard REM sleep, wakefulness, and lucid REM sleep, Mainieri et al. demonstrated significant differences in the distribution of oscillatory activity across states — with sleep paralysis showing a pattern intermediate between REM sleep and wakefulness, consistent with a mixed or transitional state. This finding has important implications for understanding not only the subjective peculiarity of the sleep paralysis experience but also its potential relationship to other anomalous states at the sleep-wake boundary, including lucid dreaming and out-of-body experiences.
The hallucinations accompanying sleep paralysis arise directly from the continuation of REM dream processes against the backdrop of environmental awareness. These are not mere mental images or vague impressions but neurophysiologically complete sensory experiences generated by the same cortical and subcortical circuits responsible for normal dreaming, now rendered vivid and spatially located because the dreamer is simultaneously aware of the real physical environment in which they lie. The neurochemical basis of the hallucinatory component has been substantially illuminated by research into serotonergic neurotransmission. As McShane and colleagues (2023) review in the American Journal of Psychiatry Residents' Journal, altered activity at serotonin-2A receptors — particularly in areas responsible for visual processing such as the inferolateral temporal cortex and in regions involved in body representation and self-referential processing — is centrally involved in generating the hallucinatory content of SP episodes. The close phenomenological resemblance between the hallucinations of sleep paralysis and those induced by the serotonin-2A receptor agonists LSD and psilocybin provides a compelling pharmacological parallel: both have the dreamlike, dissociative quality of preserved insight, the attribution of heightened personal significance to perceptions, and the vivid sensory realism that distinguishes them from the more lifelike, insight-absent character of dopaminergic hallucinations.
The activation of threat-detection circuitry — particularly amygdaloid and anterior cingulate networks — during sleep paralysis is considered the primary neural basis for the profound fear that characterizes most episodes. Unable to move, unable to call for help, and perceiving the vivid intrusion of threatening imagery into the waking environment, the individual's threat-response systems activate maximally, generating the intense fear, sympathetic arousal, and sense of helplessness that are the defining emotional qualities of the experience. This fear response may itself perpetuate the episode by maintaining a state of arousal that, paradoxically, prolongs the mixed brain state rather than fully completing the transition to wakefulness — a positive feedback dynamic that may explain why conscious attempts to struggle against the paralysis are often ineffective or even counterproductive.
Epidemiology and Population Risk Factors
Establishing the precise prevalence of sleep paralysis across populations has proved methodologically challenging, a reflection of genuine heterogeneity in diagnostic criteria, measurement instruments, and population sampling strategies across the existing literature. Prevalence estimates have ranged from as low as 2% to as high as 60% in different studies, a spread that reflects both methodological variation and true population-level differences. The systematic review by Bhalerao and colleagues (2024) in Cureus, drawing on a prior systematic review involving 36,533 participants, reported a lifetime prevalence of approximately 7.6% in the general population under more conservative criteria, with substantially elevated rates among student populations (approximately 28%) and psychiatric inpatients (approximately 32%). The most comprehensive contemporary estimate derives from the systematic review and meta-analysis by Hefnawy et al. (2024), encompassing 76 studies, 167,133 participants, and 25 countries, which yielded a global prevalence figure of 30% (95% CI: 22%–39%). The wide confidence interval reflects the genuine between-study heterogeneity, but the central estimate nonetheless clearly situates sleep paralysis as a common human experience rather than a rare clinical curiosity.
The psychiatric risk profile of sleep paralysis is among its most clinically significant epidemiological features, and one that deserves prominent attention in clinical practice. The Hefnawy meta-analysis demonstrated that the highest rates of sleep paralysis are observed in patients with PTSD and panic disorder, where prevalence rates of approximately 60% have been documented — substantially higher than the rates seen in other psychiatric populations (35%) and in student cohorts (34%). The association with PTSD is mechanistically compelling: PTSD is characterized by hyperactivation of threat-detection circuits, disrupted REM sleep architecture with excessive REM fragmentation, and exaggerated threat-appraisal responses that persist into REM sleep. These neurophysiological features may collectively lower the threshold for sleep-wake state dissociation and amplify the fear response during paralytic episodes, creating a mutually reinforcing relationship between PTSD pathophysiology and sleep paralysis phenomenology.

Beyond psychiatric comorbidity, a well-characterized constellation of behavioral, physiological, and situational risk factors predisposes individuals to sleep paralysis episodes. Acute sleep deprivation is among the most consistently replicated precipitants, acting primarily by increasing REM sleep pressure at recovery sleep — the subsequent sleep after deprivation is enriched in REM sleep and contains more frequent, longer, and more intense REM cycles, during which the likelihood of an imperfect state transition is correspondingly elevated. Irregular sleep-wake schedules, including those imposed by shift work, transmeridian travel and jet lag, and voluntary sleep restriction in student and professional populations, similarly disrupt the circadian regulation of REM sleep timing and amplify the risk of state dissociation. The supine sleeping position has been identified as a specific positional risk factor in multiple studies, with sleep paralysis episodes significantly more frequent when individuals sleep on their backs — a finding attributed to the influence of supine positioning on pharyngeal muscle tone, arousal threshold, and REM sleep architecture (Dahmen and Kasten, as cited in Hefnawy et al., 2024). A heritable component to sleep paralysis vulnerability has been suggested by twin studies demonstrating moderate heritability estimates, consistent with a polygenic predisposition modulated substantially by environmental and behavioral factors.
Clinical Presentation, Differential Diagnosis, and Cultural Context
A typical episode of sleep paralysis begins abruptly either at sleep onset or, more commonly, upon awakening from REM sleep, with the individual becoming aware of their immediate environment while finding themselves wholly unable to initiate voluntary movement. Breathing continues unimpaired, as do conjugate eye movements and blinking, and many individuals describe opening their eyes and scanning the room with full situational awareness while being otherwise completely immobile. Episodes range in duration from a few seconds to several minutes, typically resolving spontaneously and completely, often precipitated by external sensory stimulation — being touched by a bed partner, a sudden sound, a change in light — or, occasionally, by an intense voluntary effort to move a distal extremity such as a finger. For the duration of the episode, however, the subjective experience is frequently described by those affected as among the most frightening events of their lives, its terror magnified by the paradoxical clarity of consciousness during a state of complete helplessness.
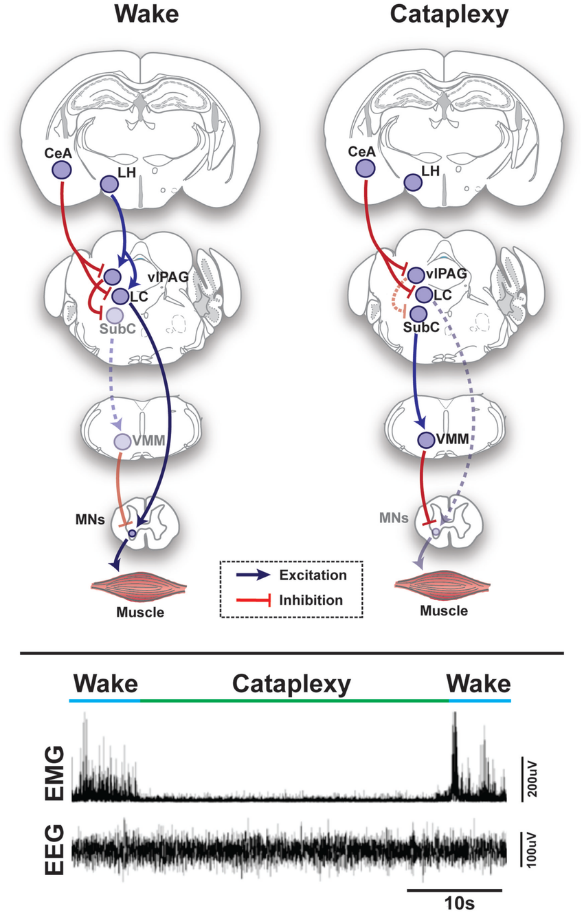
The differential diagnosis of sleep paralysis requires careful exclusion of conditions that may superficially mimic its presentation. Cataplexy, the sudden bilateral loss of voluntary muscle tone triggered by strong emotion that characterizes narcolepsy, differs from sleep paralysis in its association with emotional precipitants, its preservation of consciousness, and its typical absence of hallucinations. Hypnic jerks and other NREM parasomnias present with distinct polysomnographic signatures and lack the conscious awareness of surroundings that defines sleep paralysis. Nocturnal seizure disorders — particularly nocturnal frontal lobe epilepsy — may produce unusual nighttime behavioral episodes, but these are typically accompanied by altered or impaired consciousness, post-ictal confusion, and characteristic abnormalities on electroencephalography. In cases presenting with recurrent distressing episodes, polysomnography combined with the Multiple Sleep Latency Test (MSLT) is recommended as the definitive diagnostic workup, with particular attention to the presence of sleep-onset REM periods (SOREMPs) that would indicate underlying narcolepsy.
The cultural dimension of sleep paralysis deserves explicit clinical attention, as it profoundly shapes both the patient's experience of the phenomenon and their likelihood of seeking and accepting medical care. Across human societies, the hallucinatory experiences of sleep paralysis have been interpreted through locally available symbolic systems, giving rise to remarkably consistent cross-cultural accounts of nocturnal visitation, assault, or supernatural oppression. In Newfoundland, the entity is called the Old Hag; in Turkey, the Karabasan; in Italy, the Pandafeche; in Japanese tradition, Kanashibari. In West African and Caribbean traditions, the experience is attributed to visiting spirits or the recently dead. As McShane et al. (2023) observe, a phenomenon they identify as cultural priming means that culturally shared narratives of sleep paralysis can predispose individuals who have been exposed to those narratives to experience hallucinations that conform to them — a striking illustration of the interaction between neurophysiological substrate and cultural construction in shaping subjective experience. Clinicians working with diverse populations should approach these accounts with both anthropological sensitivity and diagnostic precision, recognizing that a patient describing a supernatural assault may be describing a neurophysiological event that is both entirely explicable and genuinely treatable.
DISCUSSION
The literature synthesized in this review reveals sleep paralysis to be a condition of considerable neurophysiological complexity and substantial clinical significance, with implications that extend far beyond the boundaries of sleep medicine into psychiatry, neurology, and the cultural study of illness experience. Several themes merit particular reflection and contextualization.
The mechanistic elegance of REM atonia's dual GABA/glycine inhibitory architecture, as described by Brooks and Peever (2012), is simultaneously a strength and a vulnerability. The redundancy of the system simultaneously targeting GABA-B, GABA-A, and glycine receptors — ensures robust protection against inadvertent motor activity during dreaming under normal physiological conditions. However, this very redundancy means that partial functional disruption of the system's temporal coordination, rather than its complete failure, may be sufficient to generate sleep paralysis without producing the full syndrome of REM sleep behavior disorder. The transitional nature of sleep paralysis as a mixed brain state, documented by Mainieri et al. (2021) through spectral EEG analysis, is entirely consistent with this mechanistic picture: the arousal systems have re-engaged and restored perceptual awareness, while the descending inhibitory drive to motoneurons has not yet been fully withdrawn, producing a state in which neither full REM sleep nor full wakefulness accurately describes the individual's neurophysiological condition.
The epidemiological findings reviewed here raise important questions about the relationship between sleep paralysis and mental health that remain incompletely resolved. The dramatically elevated rates of sleep paralysis in patients with PTSD and panic disorder — approaching 60% in meta-analytic estimates suggest a bidirectional relationship in which psychiatric illness and sleep dysregulation mutually amplify one another. Whether sleep paralysis in these populations reflects primarily the disrupted REM sleep architecture characteristic of PTSD and anxiety disorders, or whether the fear and helplessness of SP episodes themselves contribute to the maintenance and severity of those conditions through their impact on sleep quality and fear generalization, is a question with direct therapeutic implications. If the former, treating the underlying psychiatric condition may secondarily reduce SP frequency; if the latter, specific intervention for SP may contribute to improved psychiatric outcomes. The evidence reviewed suggests both mechanisms are likely operative, and an integrated approach addressing both the sleep disturbance and the psychiatric comorbidity is clinically warranted.
The pharmacological management of sleep paralysis remains an underdeveloped area with no approved therapies specifically indicated for the condition. The current pharmacological strategies are largely borrowed from related conditions and are based more on mechanistic reasoning than on evidence from large randomized controlled trials. Tricyclic antidepressants — particularly clomipramine — and selective serotonin reuptake inhibitors (SSRIs) have been employed with some reported clinical success, rationalized through their potent REM-suppressive effects: by reducing the frequency, duration, and depth of REM sleep, these agents decrease the frequency of the state transitions during which sleep paralysis arises. The mechanistic specificity of serotonin-2A receptor involvement in SP hallucinations, as reviewed by McShane et al. (2023), raises the intriguing possibility that serotonin-2A receptor antagonism might selectively attenuate the hallucinatory component of SP episodes without necessarily altering sleep architecture broadly — a hypothesis that warrants systematic pharmacological investigation. Sodium oxybate, which is approved for use in narcolepsy through its consolidation of nocturnal sleep and reduction of REM fragmentation, may have particular applicability in narcolepsy-associated sleep paralysis.
Non-pharmacological interventions represent the most evidence-consistent approach currently available for managing recurrent isolated sleep paralysis. Cognitive-behavioral therapy adapted specifically for sleep paralysis, incorporating psychoeducation about the neurophysiological basis of the experience, cognitive restructuring of catastrophic interpretations of episodes, and behavioral modification targeting the precipitating factors — particularly sleep deprivation, irregular sleep schedules, and supine sleeping position — has demonstrated effectiveness in controlled studies. Psychoeducation is especially powerful in this context: the knowledge that one's experience of paralysis and hallucination has a precise, benign, and well-understood neurological explanation fundamentally transforms the subjective experience of episodes for many patients, converting terror into curiosity and helplessness into informed self-management. This transformation is itself a form of therapeutic mechanism — by reducing the fear response during episodes, psychoeducation may attenuate the very sympathetic activation that prolongs the mixed brain state.
The relationship between sleep paralysis and the broader field of REM sleep pathology invites reflection on the spectrum of conditions arising from dysregulation of REM atonia. At one extreme lies REM sleep behavior disorder (RBD), in which atonia fails completely and individuals physically enact dream content, sometimes with injurious consequences to themselves or bed partners. At the other extreme — with perhaps too much atonia, or atonia poorly timed in relation to awakening — lies sleep paralysis. Between these poles lie a range of partial and transitional conditions including narcolepsy with its multiple state-boundary failures. The discovery that RBD is a prodromal marker of alpha-synucleinopathies including Parkinson's disease and Lewy body dementia has brought the regulation of REM atonia to the forefront of neurodegenerative research. While sleep paralysis has not been identified as a similarly predictive marker, the commonality of mechanism suggests that a deeper understanding of the neural circuitry governing REM atonia — and its vulnerability to disruption — will yield insights relevant not only to sleep medicine but to neurodegenerative disease research more broadly.
CONCLUSION
Sleep paralysis is, in its essence, a vivid illustration of the brain's extraordinary complexity and the intricate, actively maintained choreography required to navigate the transition between the sleeping and waking worlds. When the subcoeruleus nucleus, the dual GABAergic and glycinergic inhibitory systems projecting to spinal motoneurons, the orexinergic stabilizing circuits of the lateral hypothalamus, and the broader brainstem-hypothalamic network governing REM sleep fail to synchronize their activities precisely at the moment of awakening, the result is a state that has simultaneously fascinated, terrified, and inspired human beings throughout recorded history. What was once attributed to night-demons and supernatural visitation is now understood as a transient neurophysiological dissociation — real in its consequences, explicable in its mechanisms, and meaningfully manageable in its manifestations.
The evidence synthesized from six peer-reviewed medical publications underscores that sleep paralysis is more common than many clinicians appreciate, substantially more distressing to affected individuals than its technically benign reputation might suggest, and more amenable to intervention than the current paucity of specifically approved treatments implies. The strong epidemiological associations with PTSD and panic disorder call for sleep paralysis to be routinely and proactively assessed in psychiatric practice. The well-characterized precipitating role of sleep deprivation, circadian disruption, and supine positioning points toward accessible and low-cost behavioral modifications that can meaningfully reduce episode frequency in many affected individuals. And the neurochemical specificity of the mechanisms involved — particularly the dual GABA/glycine inhibitory drive at the motoneuron level and the serotonin-2A receptor mediation of hallucinatory content — identifies rational pharmacological targets that warrant systematic investigation through well-powered clinical trials.
Perhaps most importantly, the cross-cultural universality of the sleep paralysis experience, and the enduring human tendency to interpret its neurologically generated hallucinations through the frameworks of supernatural encounter, reminds clinicians that the experience of illness is always simultaneously biological and cultural. Understanding the neuroscience of why the brain sometimes wakes before the body is necessary but not sufficient; understanding the patient's own framework for making sense of that experience — and meeting them within it with both scientific clarity and genuine compassion — is what transforms neurophysiological knowledge into therapeutic benefit. As our understanding of the sleeping brain deepens, so too must our capacity to translate that understanding into care that honors both the precision of the science and the full humanity of the person lying awake, paralyzed, waiting for the body to catch up with the mind.
REFERENCES
1. Bhalerao, V., Gotarkar, S., Vishwakarma, D., & Kanchan, S. (2024). Recent insights into sleep paralysis: Mechanisms and management. Cureus, 16(7), e65413. https://doi.org/10.7759/cureus.65413
2. Brooks, P. L., & Peever, J. H. (2012). Identification of the transmitter and receptor mechanisms responsible for REM sleep paralysis. Journal of Neuroscience, 32(29), 9785–9795. https://doi.org/10.1523/JNEUROSCI.0482-12.2012
3. Hefnawy, M. T., Amer, B. E., Amer, S. A., Moghib, K., Khlidj, Y., & Elfakharany, B. (2024). Prevalence and clinical characteristics of sleeping paralysis: A systematic review and meta-analysis. Cureus, 16(1), e53212. https://doi.org/10.7759/cureus.53212
4. Mainieri, G., Maranci, J. B., Champetier, P., Leu-Semenescu, S., Gales, A., Dodet, P., & Arnulf, I. (2021). Are sleep paralysis and false awakenings different from REM sleep and from lucid REM sleep? A spectral EEG analysis. Journal of Clinical Sleep Medicine, 17(4), 719–727. https://doi.org/10.5664/jcsm.9056
5. McShane, C., Heckel, M., & Mathur, A. (2023). Sleep paralysis: A brief overview of the intersections of neurophysiology and culture. American Journal of Psychiatry Residents' Journal, 18(4). https://doi.org/10.1176/appi.ajp-rj.2023.180401
6. Vetrivelan, R., & Bandaru, S. S. (2023). Neural control of REM sleep and motor atonia: Current perspectives. Current Neurology and Neuroscience Reports, 23, 907–923. https://doi.org/10.1007/s11910-023-01322-x
